7.3: Glycoconjugates - Proteoglycans, Glycoproteins, Glycolipids and Cell Walls
- Page ID
- 14957
\( \newcommand{\vecs}[1]{\overset { \scriptstyle \rightharpoonup} {\mathbf{#1}} } \)
\( \newcommand{\vecd}[1]{\overset{-\!-\!\rightharpoonup}{\vphantom{a}\smash {#1}}} \)
\( \newcommand{\dsum}{\displaystyle\sum\limits} \)
\( \newcommand{\dint}{\displaystyle\int\limits} \)
\( \newcommand{\dlim}{\displaystyle\lim\limits} \)
\( \newcommand{\id}{\mathrm{id}}\) \( \newcommand{\Span}{\mathrm{span}}\)
( \newcommand{\kernel}{\mathrm{null}\,}\) \( \newcommand{\range}{\mathrm{range}\,}\)
\( \newcommand{\RealPart}{\mathrm{Re}}\) \( \newcommand{\ImaginaryPart}{\mathrm{Im}}\)
\( \newcommand{\Argument}{\mathrm{Arg}}\) \( \newcommand{\norm}[1]{\| #1 \|}\)
\( \newcommand{\inner}[2]{\langle #1, #2 \rangle}\)
\( \newcommand{\Span}{\mathrm{span}}\)
\( \newcommand{\id}{\mathrm{id}}\)
\( \newcommand{\Span}{\mathrm{span}}\)
\( \newcommand{\kernel}{\mathrm{null}\,}\)
\( \newcommand{\range}{\mathrm{range}\,}\)
\( \newcommand{\RealPart}{\mathrm{Re}}\)
\( \newcommand{\ImaginaryPart}{\mathrm{Im}}\)
\( \newcommand{\Argument}{\mathrm{Arg}}\)
\( \newcommand{\norm}[1]{\| #1 \|}\)
\( \newcommand{\inner}[2]{\langle #1, #2 \rangle}\)
\( \newcommand{\Span}{\mathrm{span}}\) \( \newcommand{\AA}{\unicode[.8,0]{x212B}}\)
\( \newcommand{\vectorA}[1]{\vec{#1}} % arrow\)
\( \newcommand{\vectorAt}[1]{\vec{\text{#1}}} % arrow\)
\( \newcommand{\vectorB}[1]{\overset { \scriptstyle \rightharpoonup} {\mathbf{#1}} } \)
\( \newcommand{\vectorC}[1]{\textbf{#1}} \)
\( \newcommand{\vectorD}[1]{\overrightarrow{#1}} \)
\( \newcommand{\vectorDt}[1]{\overrightarrow{\text{#1}}} \)
\( \newcommand{\vectE}[1]{\overset{-\!-\!\rightharpoonup}{\vphantom{a}\smash{\mathbf {#1}}}} \)
\( \newcommand{\vecs}[1]{\overset { \scriptstyle \rightharpoonup} {\mathbf{#1}} } \)
\(\newcommand{\longvect}{\overrightarrow}\)
\( \newcommand{\vecd}[1]{\overset{-\!-\!\rightharpoonup}{\vphantom{a}\smash {#1}}} \)
\(\newcommand{\avec}{\mathbf a}\) \(\newcommand{\bvec}{\mathbf b}\) \(\newcommand{\cvec}{\mathbf c}\) \(\newcommand{\dvec}{\mathbf d}\) \(\newcommand{\dtil}{\widetilde{\mathbf d}}\) \(\newcommand{\evec}{\mathbf e}\) \(\newcommand{\fvec}{\mathbf f}\) \(\newcommand{\nvec}{\mathbf n}\) \(\newcommand{\pvec}{\mathbf p}\) \(\newcommand{\qvec}{\mathbf q}\) \(\newcommand{\svec}{\mathbf s}\) \(\newcommand{\tvec}{\mathbf t}\) \(\newcommand{\uvec}{\mathbf u}\) \(\newcommand{\vvec}{\mathbf v}\) \(\newcommand{\wvec}{\mathbf w}\) \(\newcommand{\xvec}{\mathbf x}\) \(\newcommand{\yvec}{\mathbf y}\) \(\newcommand{\zvec}{\mathbf z}\) \(\newcommand{\rvec}{\mathbf r}\) \(\newcommand{\mvec}{\mathbf m}\) \(\newcommand{\zerovec}{\mathbf 0}\) \(\newcommand{\onevec}{\mathbf 1}\) \(\newcommand{\real}{\mathbb R}\) \(\newcommand{\twovec}[2]{\left[\begin{array}{r}#1 \\ #2 \end{array}\right]}\) \(\newcommand{\ctwovec}[2]{\left[\begin{array}{c}#1 \\ #2 \end{array}\right]}\) \(\newcommand{\threevec}[3]{\left[\begin{array}{r}#1 \\ #2 \\ #3 \end{array}\right]}\) \(\newcommand{\cthreevec}[3]{\left[\begin{array}{c}#1 \\ #2 \\ #3 \end{array}\right]}\) \(\newcommand{\fourvec}[4]{\left[\begin{array}{r}#1 \\ #2 \\ #3 \\ #4 \end{array}\right]}\) \(\newcommand{\cfourvec}[4]{\left[\begin{array}{c}#1 \\ #2 \\ #3 \\ #4 \end{array}\right]}\) \(\newcommand{\fivevec}[5]{\left[\begin{array}{r}#1 \\ #2 \\ #3 \\ #4 \\ #5 \\ \end{array}\right]}\) \(\newcommand{\cfivevec}[5]{\left[\begin{array}{c}#1 \\ #2 \\ #3 \\ #4 \\ #5 \\ \end{array}\right]}\) \(\newcommand{\mattwo}[4]{\left[\begin{array}{rr}#1 \amp #2 \\ #3 \amp #4 \\ \end{array}\right]}\) \(\newcommand{\laspan}[1]{\text{Span}\{#1\}}\) \(\newcommand{\bcal}{\cal B}\) \(\newcommand{\ccal}{\cal C}\) \(\newcommand{\scal}{\cal S}\) \(\newcommand{\wcal}{\cal W}\) \(\newcommand{\ecal}{\cal E}\) \(\newcommand{\coords}[2]{\left\{#1\right\}_{#2}}\) \(\newcommand{\gray}[1]{\color{gray}{#1}}\) \(\newcommand{\lgray}[1]{\color{lightgray}{#1}}\) \(\newcommand{\rank}{\operatorname{rank}}\) \(\newcommand{\row}{\text{Row}}\) \(\newcommand{\col}{\text{Col}}\) \(\renewcommand{\row}{\text{Row}}\) \(\newcommand{\nul}{\text{Nul}}\) \(\newcommand{\var}{\text{Var}}\) \(\newcommand{\corr}{\text{corr}}\) \(\newcommand{\len}[1]{\left|#1\right|}\) \(\newcommand{\bbar}{\overline{\bvec}}\) \(\newcommand{\bhat}{\widehat{\bvec}}\) \(\newcommand{\bperp}{\bvec^\perp}\) \(\newcommand{\xhat}{\widehat{\xvec}}\) \(\newcommand{\vhat}{\widehat{\vvec}}\) \(\newcommand{\uhat}{\widehat{\uvec}}\) \(\newcommand{\what}{\widehat{\wvec}}\) \(\newcommand{\Sighat}{\widehat{\Sigma}}\) \(\newcommand{\lt}{<}\) \(\newcommand{\gt}{>}\) \(\newcommand{\amp}{&}\) \(\definecolor{fillinmathshade}{gray}{0.9}\)-
Define Key Terminology and Concepts:
- Differentiate among the terms “sugar,” “carbohydrate,” and “glycan” and explain how each applies to both free and protein-conjugated carbohydrates.
- Understand the structural diversity introduced by glycosylation, and why glycans are considered “information rich” compared to linear biopolymers like DNA and proteins.
-
N-linked Glycoprotein Structure and Diversity:
- Describe the core oligosaccharide structure (Man)3(GlcNAc)2(\text{Man})_3(\text{GlcNAc})_2 attached to Asn in the consensus sequence (X-Asn-X-Thr/Ser) and its significance in N-linked glycoprotein formation.
- Differentiate between high-mannose, complex, and hybrid N-linked glycans by comparing their outer sugar compositions (e.g., presence or absence of mannose, lactosamine repeats, or branching patterns) and discuss their functional implications.
- Utilize SNFG (Symbolic Nomenclature For Glycans) to interpret and represent glycan structures and variations.
-
O-linked Glycoproteins and Blood Group Antigens:
- Explain the process of O-linked glycosylation, where carbohydrates (commonly Gal(β1,3)GalNAc) attach to Ser/Thr residues, and relate this to the structure and function of blood group antigens.
- Recognize how variations in O-linked glycan structures contribute to cell–cell recognition and immune responses.
-
Proteoglycans and the Extracellular Matrix (ECM):
- Describe the structure of proteoglycans, including the linkage of glycosaminoglycans (GAGs) to core proteins, and differentiate between soluble and membrane-bound forms.
- Explain the roles of proteoglycans in the ECM, including their contribution to tissue structure, cellular adhesion, and signal transduction.
-
Carbohydrate Structures in Cell Walls and Membranes:
- Compare and contrast the composition and structure of cell walls in different organisms:
- For Gram-positive bacteria: Describe the peptidoglycan network, including the roles of NAG and NAM, pentapeptides, and teichoic acids.
- For Gram-negative bacteria: Explain the arrangement of the peptidoglycan layer, outer membrane, and the role of lipopolysaccharides (LPS) in antigenicity and antibiotic resistance.
- For plants: Summarize the components of primary and secondary cell walls (cellulose, hemicellulose, pectin, and lignin) and discuss how these polymers contribute to cell rigidity and growth.
- Discuss the unique features of archaeal cell membranes and walls, including differences in lipid composition and the absence of peptidoglycan.
- Compare and contrast the composition and structure of cell walls in different organisms:
-
Biological and Clinical Implications of Glycosylation:
- Evaluate the functional roles of glycosylation in protein folding, stability, proteolytic protection, and in modulating protein half-life.
- Analyze how specific glycan structures (e.g., Gal(α1,3)Gal) affect immune recognition and their implications in xenotransplantation and diseases such as alpha-gal syndrome.
- Discuss how differences in glycosylation patterns contribute to cell signaling and tissue-specific functions, particularly within the ECM and basement membranes.
By achieving these goals, students will gain a comprehensive understanding of the multifaceted roles that carbohydrate modifications play in protein function and cellular organization, and they will be prepared to explore further applications of glycosylation in health, disease, and biotechnology.
Many proteins, especially those destined for secretion or insertion into membranes, are post-translationally modified by the attachment of carbohydrates. They are usually attached through either Asn or Ser side chains. Carbohydrate modifications on the protein appear to be involved in recognizing other binding molecules, preventing aggregation during protein folding, protecting from proteolysis, and increasing the protein's half-life. In contrast to a protein sequence determined by a DNA template, sugars are attached to proteins by enzymes that recognize appropriate sites on proteins and attach the sugars. Since there are many sugars with many functional groups that can serve as potential attachment sites, the structures of the oligosaccharides attached to proteins are enormously varied, complex, and hence "information-rich" compared to linear or folded polymers like DNA and proteins.
N-linked Glycoproteins
These contain carbohydrates attached through either a GlcNAc or GalNAc to an Asn in an X-Asn-X-Thr sequence of the protein. There are three types of N-linked glycoproteins: high mannose, complex, and hybrid. They all contain the same core oligosaccharide - (Man)3(GlcNAc)2 attached to Asn as shown in Figure \(\PageIndex{1}\).
Table \(\PageIndex{1}\) below shows the SNFG representation for the main core and variant glycans of N-linked glycoproteins. Note that the designation of α2 implies an α(1→2) linkage. Unless otherwise stated, the linkage is presumed to start from carbon 1.
| Core | |
| High mannose | |
| Complex | |
| Mixed hybrid |
Table \(\PageIndex{1}\): SNFG representation for the main core and variant glycans in N-linked glycoproteins
Complex N-linked glycans don't contain mannose outside the core glycan and have GlcNAc attached to the branching mannoses in the core structure. The complex glycan shown above has a Gal(β1,4)GlcNAc sequence, which could be named the disaccharide lactosamine. Often, lactosamine repeats in the sequence.
Hybrid glycans have both unsubstituted terminal mannoses (as in the high-mannose type) and substituted mannoses with an N-acetylglucosamine attached (as in the complex type. GlcNAc residues added to the core in the hybrid and complex N-glycoproteins are called antennae. Figure \(\PageIndex{2}\) shows an example of a biantennary N-linked glycan with two GlcNAc branches linked to the core. The core is outlined in red, and the two GlcNAcs are labeled 1 and 2.
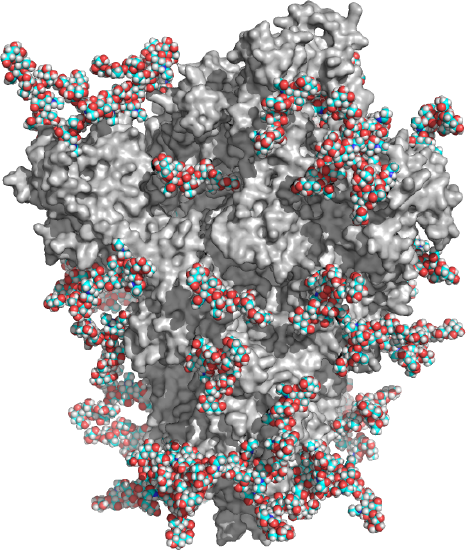
Complex glycans also have bi-, tri-, and tetraantennary forms and comprise most N-glycans. As shown in Table \(\PageIndex{1}\) above, complex N-linked glycans usually end with sialic acid residues. About 50% of the surface area of the COVID-19 SARS-CoV-2 spike protein is covered with glycans, as shown in the model structure in Figure \(\PageIndex{3}\). The protein surface is gray, and the glycans (biantennary LacNAc N-glycans) in spacefill CPK with carbon in cyan.
In the hybrid oligosaccharide shown above, one terminus contains Gal(β1,4)GlcNAc. However, in all other mammals except man, apes, and Old World monkeys, an additional Gal is often connected in an α1,3 link to the Gal to give a terminus of Gal(α1,3)Gal(β1,4)GlcNAc. These animals have an additional enzyme, an α1,3 Gal transferase. Bacteria also have this enzyme, and since we have been exposed to this link through bacterial infection, we mount an immune response against it. Why is this important? Pig hearts are similar to human hearts so that they might be good candidates for human transplantation (xenotransplants). However, the Gal-α1,3-Gal link is recognized as foreign, and we mount a significant immune response against it. Several biotech firms are trying to delete the pig α1,3 Gal transferase, which would prevent the addition of the terminal Gal and make them good donors for transplanted hearts.
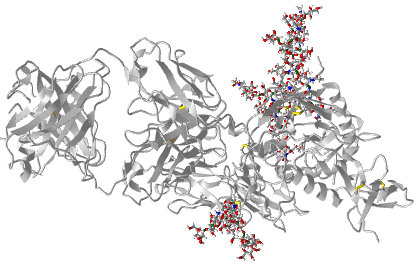
Figure \(\PageIndex{4}\) shows an interactive iCn3D model of an N-linked glycoprotein, human beta-2-glycoprotein-I (Apolipoprotein-H) (1C1Z).
_(1C1Z).png?revision=1&size=bestfit&width=443&height=226)
The glycan structures for the beta-2-glycoprotein-I are shown below. Identify the monosaccharides in each and specify to which asparagine they are linked.
- Answer
-
Here is an interactive iCn3D image showing the protein and attached glycans using SNFG notation.
Figure \(\PageIndex{5}\) shows an interactive iCn3D model of the GP120 HIV protein that contains high mannose, complex, and hybrid N-linked glycans. Most glycoproteins in the Protein Data Bank do not contain attached glycans. The glycans here were added with the program GlyProt at 3 of 17 possible Asn residues that would presumably have attached glycans. Use your mouse or keypad to hover over the monomers in the attached glycans. Abbreviations for the given residues in the model are: adm = alpha-D-Man, bdg= beta-D-Glc or Gal, adn = alpha-D-neuraminidase.
The coronavirus pandemic has been deadly (over 1.1 million deaths in the USA alone and 19-36 million around the world). However, the 1918 influenza pandemic was far worse per capita, with an estimated 650,000 deaths in the USA and 50 million around the world in a population less than 1/3 of the present. An additional 3 million deaths in the USA were probably prevented by vaccination as of December 2022. Many deaths in the developing world would have been prevented if wealthy nations had allocated more resources to produce and distribute the vaccines. The evolution of the virus in nonvaccinated areas might come back to haunt wealthy countries if present vaccines become ineffective against the mutants. A worse pandemic might await us. An avian version of the influenza virus (H5N1), presently endemic in wild birds and now found in mink populations, has infected 240 people as of January 2023 and killed 56% of them. A quick note: the 1918 pandemic affected youth the most.
Figure \(\PageIndex{6}\) shows the simple yet deadly influenza virus. It interacts with human cells through a surface protein, hemagglutinin (HA).
Figure \(\PageIndex{6}\): Alpha Influenza virus. https://viralzone.expasy.org/6. Creative Commons Attribution 4.0 International (CC BY 4.0) License.
Note the similarities and differences with the SARS-CoV-2 virus, shown below.
SARS-Covid-2 virus. https://viralzone.expasy.org/764. Creative Commons Attribution 4.0 International (CC BY 4.0) License.
The virus binds to host cells through the interaction of HA with cell surface carbohydrates. Once bound, the virus internalizes, ultimately releasing the viral RNA genome into the host cell.
The hemagglutinin protein is the most abundant protein on the viral surface. 15 avian and mammalian variants have been identified (based on antibody studies). Only three have adapted to humans in the last 100 years, giving pandemic strains H1 (1918), H2 (957), and H3 (1968). Three recent avian variants (H5, H7, and H9) have jumped directly to humans recently, but have low human-to-human transmissibility.
The influenza hemagglutinin protein has the following characteristics:
- the mature form is a homotrimer (3 identical protein subunits), MW 220,000 with multiple sites for covalent attachment of sugars. Hemagglutinin is a glycoprotein.
- each monomer is synthesized as a single polypeptide chain precursor (HA0), which is cleaved into HA1 and HA2 subunits by the protease trypsin in epithelial cells of the lung.
- structure known for human (H3), swine (H9), avian (H5) subtypes.
Figure \(\PageIndex{7}\) below shows an interactive iCn3D model of an influenza hemagglutinin trimer (6HJQ). The white backbone traces on the membrane's extracellular (red) side are antibody molecules used to stabilize the structure for crystallization.
Figure \(\PageIndex{7}\): Influenza Hemagglutinin Trimer (6HJQ). (Copyright; author via source). Click the image for a popup or use this external link: https://structure.ncbi.nlm.nih.gov/i...EhePsF4BiEjwA8
The protein is a trimer of a heterodimer, (HA1:HA2)3. Each heterodimer has two separate chains, HA1 and HA2. Together they form a globular head and stalk region, the latter crossing the membrane. HA1 interacts primarily with the sialic acid on host cells' surface proteins, while HA2 is more involved in the fusion of the virus with host cells and internalization.
Hemagglutinin binds to sialic acid (Sia) covalently attached to many cell membrane glycoproteins. The sialic acid is usually connected through an α(2,3) or α(2,6) link to galactose on N-linked glycoproteins. The subtypes found in avian (and equine) influenza isolates bind preferentially to Sia (α2,3) Gal, which predominates in the avian GI tract where viruses replicate. Human influenza isolates prefer Sia α(2,6)
Sia (α2,3) Gal predominates in the avian GI tract, where viruses replicate. Human influenza isolates prefer Sia α(2,6) Gal. The human virus of H1, H2, and H3 subtypes (causes of the 1918, 1957, and 1968 pandemics) recognizes Sia α(2,6) Gal, the major form in the human respiratory tract. The swine influenza HA binds to Sia α(2,6) Gal and some Sia (α2,3) Gal, both found in swine. The structures of the Sia-Gal disaccharide are shown in Table \(\PageIndex{2}\) below.
| Sia α(2,6) Gal (Human) | Sia α(2,3) Gal (Avian and some Swine) |
|
|
|
|
(made with Sweet, with an OH, not AcNH on sialic acid on C5) |
(made with Sweet, with an OH, not AcNH on sialic acid on C5) |
Table \(\PageIndex{2}\): Structures of Sia α(2,6) Gal (human) and Sia α(2,3) Gal (avian/swine)
The H5N1 avian flu H5N1) virus is deadly but presently lacks human-to-human transmissibility. Why? One reason is that it appears to bind deep in the lungs and is not released easily on coughing or sneezing. It appears that cell surface glycoproteins deeper in the respiratory tract have Sia (α2,3) Gal which accounts for this pathology.
Before it leaves the cell, the virus forms a bud on the intracellular side of the cell with the HA and NA in the cell membrane of the host cell. The virus in this state would not leave the cell since its HA molecules would interact with sialic acid residues in the host cell membrane, holding the virus in the membrane. Neuraminidase hydrolyzes sialic acid from cell surface glycoproteins, allowing the virus to complete the budding process and be released from the cell as new viruses. The drugs Oseltamivir (Tamiflu) and Zanamivir (Relenza) bind to and inhibit neuraminidase, whose activity is necessary for viral release from infected cells. Tamiflu appears to work against N1 of the present H5N1 avian influenza viruses. Governments across the world are hopefully stockpiling this drug in case of a pandemic caused by the avian virus jumping directly to humans and becoming transmissible from human to human.
O-linked Glycoproteins
The CHOs are usually attached from a Gal (β 1,3) GalNAc to a Ser or Thr of a protein, as shown in Figure \(\PageIndex{8}\).
Figure: O-linked Glycoproteins
The blood group antigens (CHOs on cells attached to either proteins or lipids) are examples. The sugars shown as chairs (in contrast to structures found in many texts) in Figure \(\PageIndex{9}\) are the blood group antigens. They are attached to a core heterosaccharide (a red ellipse below), which is connected to either a membrane glycoprotein or glycolipid.
Figure \(\PageIndex{10}\) shows the SNFG representation for the A antigen in the glycolipid form.
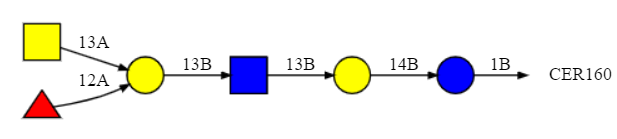
The trimeric branched residues on the left-hand side represent the A antigen shown above. The red triangle is L-fucose. Yellow represents galactose or GalNac, while blue is glucose or GlcNAc.
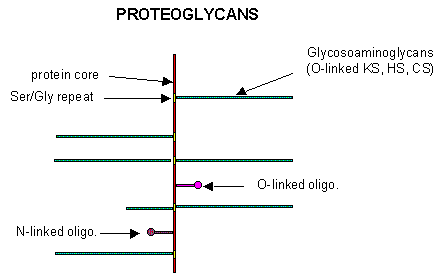
Proteoglycans
Some proteins are so modified with CHOs that they contain more CHOs than amino acids. Proteins linked to glycosaminoglycans are called proteoglycans (PGs). They consist of a core protein linked to one or more glycosaminoglycans. GAGs are linear sulfated glycans, which we described earlier. The structures of a few proteoglycans are known. The GAGs are O-linked to the protein, typically to a Ser of a Ser-Gly dipeptide, often repeated in the protein. Some of the proteoglycans also contained N-linked oligosaccharide groups. Figure \(\PageIndex{10}\) represents a proteoglycan structure.
PGs can be soluble and are found in the extracellular matrix or as integral membrane proteins. There are about 43 genes for proteoglycans. Differential splicing of the RNA transcripts gives rise to soluble and transmembrane forms. Given the diversity of sugars and the varying extent of sulfation, the CHO part of PGs provides an incredible variety of binding structures at or near the cell surface. Figure \(\PageIndex{11}\) shows the variety of proteoglycans found in mammalian cells. PGs help form the extracellular matrix, which provides a rich binding environment between cells.
One PG, syndecan, binds through its intracellular domain to the internal cytoskeleton of the cell while interacting with another protein - fibronectin - in the extracellular matrix. Fibronectin also binds other molecules, regulating cellular growth and other interactions. PGs act like glue in connecting the extracellular and intracellular functions of the cell. There are four different core syndecan proteins (SDCs 1–4), with SDC4 lacking the cytoplasmic and transmembrane domains and is a soluble form in the intracellular matrix. The glycan components of syndecans are mostly heparan sulfate, while SDC 1 and 3 also have two chondroitin sulfate chains.
Most proteins bind PGs through a PG binding motif of BBXB or BBBXXB, where B is a basic amino acid. Some proteins bind to specific sequences in specific GAGs. For instance, antithrombin 3, an inhibitor of blood clotting, binds specifically to heparin. This enhances its interaction with clotting proteins such as thrombin and Factor Xa. Figure \(\PageIndex{12}\) shows an interactive iCn3D model of a five-residue fragment of heparin interacting with the key amino acids' side chains of Factor Xa (2gd4).
.png?revision=1&size=bestfit&width=275&height=201)
The extracellular matrix (ECM) might appear to be a nondescript mess for those more chemically oriented, since chemists are used to well-defined structures. Figure \(\PageIndex{13}\) shows a cartoon of the ECM and may clarify the components. Few structure files exist for them, given the inherent flexibility of the glycan components.
Cell Walls and Glycolipids
In contrast to eukaryotic cells, bacteria, and plant cells have a cell wall in addition to a lipid bilayer membrane. These are essentially carbohydrate polymers that determine cell shape, affording protection from exterior pathogens, hypotonic conditions, and high internal osmotic pressures, preventing swelling and bursting of the cells. This is especially important in plants, which need strength and rigidity against the "turgor" pressure of the aqueous cytoplasm against the cell membrane. This prevents wilting in plants. The cell walls in plants and probably bacteria are involved in cell signaling across the cell membrane.
Bacterial Cell Walls
Two types of cell walls occur.
a. Gram-positive bacteria-
These bacteria can be stained with a Gram stain. The wall consists of a GlcNAc (β 1,4) MurNAc repeat. (GlcNAc is often abbreviated as NAG, while MurNAc is abbreviated as NAM.) This is similar to the GlcNAc (β 1,4) GlcNAc homopolymer chitin, except that every other GlcNAc contains a lactate molecule covalently attached in an ether-linkage to the C3 hydroxyl to form the monomer N-Acetylmuramic acid. A pentapeptide (Ala-D-isoGlu-Lys-D-Ala-D-Ala) is attached through an amide link to the carboxyl group of the lactate in MurNAc. A pentaglycine bridge covalently connects the GlcNAc (β 1,4) MurNAc strands through the epsilon amino group of the pentapeptide Lys on one strand and the terminal D-Ala of a pentapeptide on another strand. A small part of the structure of a gram-positive bacterial cell wall is shown in Figure \(\PageIndex{14}\). It shows one repeating GlcNAc-MurNAc disaccharide unit in front (darker) and one in the back (lighter) connected through the peptides shown.
The SNFG representation of a larger section of the gram-positive cell well is shown in Figure \(\PageIndex{15}\).
One final structure is found in Gram-positive peptidoglycan cell walls. Teichoic acids are often attached to carbon 6 of MurNAc. Teichoic acid is a polymer of glycerol or ribitol with alternative GlcNAc and D-Ala linked to the middle C of the glycerol. Multiple glycerols are linked through phosphodiester bonds. These teichoic acids often make up 50% of the dry weight of the cell wall and present a foreign (or antigenic) surface to infected hosts. These often serve as receptors for viruses that infect bacteria (called bacteriophages). Its structure is illustrated in Figure \(\PageIndex{16}\).
Notice that all monomeric units of peptidoglycan and attached teichoic acid derivatives are covalently attached to form one large molecule comprising the entire cell wall! This structure, along with the Gram-negative cell wall structures, is the largest single macromolecule in nature.
b. Gram-negative bacteria
These bacteria can NOT be stained with a Gram stain. The wall consists of the same structure as in Gram-positive bacteria. However, the GlcNAc (β 1,4) MurNAc strands are covalently connected through a direct amide bond between a derivative of Lys, meso-diaminopimelic acid (m-A2pm), on one peptide strand and to the last D-Ala of a pentapeptide on another strand. (i.e., there is no penta-Gly spacer). The connector peptide is Ala-D-isoGlu-m-A2pm-D-Ala-D-Ala
m-A2pm replaces Lys 3 of the peptide in most Gram-negative species and Gram-positive bacteria of the genus Bacillus and mycobacteria. The stereochemistries at each chiral center are different (R and S), but because the molecule has a plane of symmetry, it is an example of a meso-compound, a diastereoisomer of a molecule that does not have a different enantiomeric version. The structure is shown in Figure \(\PageIndex{17}\).
A small part of the structure of a Gram-negative bacterial cell wall is shown in Figure \(\PageIndex{18}\).
Figure \(\PageIndex{19}\) shows an image of a computed model (not a crystal or NMR structure) of the Gram-negative peptidoglycan of E. Coli. Jame Gumbart kindly provided the PDB coordinates. The repeating (GlcNAc-MurNAc)n are shown in sticks, alanines in red spheres, m-A2-pm (meso-diaminopimelic acid) in gray spheres, and DiG in orange spheres. The glycine pentapeptide is not shown since it is not found in Gram-negative bacteria.

Figure \(\PageIndex{19}\): Part of a Gram-negative peptidoglycan of E. Coli. PDB coordinates kindly provided by Jame Gumbart.
Follow these instructions to get an interactive iCn3D model of the structure.
- open iCn3D
- Download this file to your computer's download folder. IMPORTANT: If the file opens as an image in a new browser window, right-click the image and save the file to download it!
- File, Open File, iCn3D png (appendable), and choose the downloaded file (it takes a while).
In addition, Gram-negative bacteria don't have teichoic acid polymers. Rather, they have a second, outer lipid bilayer. The cell wall peptidoglycan (PG) is sandwiched between the inner and outer bilayers. The space between the lipid bilayers is called the periplasm. The outer leaflet of the outer membrane is coated with a lipopolysaccharide (LPS) (a glycolipid) of varying composition. The LPS determines the antigenicity of the bacteria. The different LPS are called the O-antigens. Figure \(\PageIndex{20}\) shows the structure of the Gram-negative bacterial membrane organization. (In the figure, PS is LPS, PG is peptidoglycan). The LPS in the outer leaflet is amphiphilic, with nonpolar acyl chains forming a more classic bilayer, and the inner leaflet phospholipid and polar/charged sugars form the LPS fringe. The extra membrane of Gram-negative bacteria makes them the major source of antibiotic resistance.

A detailed view of the structure of the lipopolysaccharide (LPS) from Salmonella typhimurium is shown in Figure \(\PageIndex{21}\) below.
Figure \(\PageIndex{21}\): Lipopolysaccharide (LPS) from Salmonella Typhimurium
Recent Updates: 11/8/24
LPS is synthesized in the inner leaflet of the inner membrane and must translocate all the way to the outer leaflet of the outer membrane. Movement requires an ATP-binding cassette transporter LptB2FG. Figure \(\PageIndex{22}\) shows the machinery used to move LPS to the outer membrane. We'll discuss membrane proteins and transport thoroughly in Chapter 11.
Figure \(\PageIndex{22}\): LPS transport from the IM to the OM by the trans-envelope complex LptABCDEFG. Dong, H., Zhang, Z., Tang, X. et al. Structural and functional insights into the lipopolysaccharide ABC transporter LptB2FG. Nat Commun 8, 222 (2017). https://doi-org.ezproxy.csbsju.edu/1...67-017-00273-5. Creative Commons Attribution 4.0 International License. http://creativecommons.org/licenses/by/4.0/.
Legend: LPS is extracted from the periplasmic side of the IM by the ABC transporter LptB2FG, and is delivered to an IM protein LptC, which forms a complex with LptB2FG. LptC comprises a single membrane-spanning domain and a large periplasmic domain, forming a periplasmic bridge with LptA and the N-terminal domain of LptD. LPS is then inserted into the OM by the LptD/E complex. LPS contains O-antigen, core oligosaccharide, and lipid A components, of which the O-antigen has 4-40 O-antigen repeat units. Ra-LPS, rough LPS, Kdo, 3-deoxy-D-manno-oct-2-ulosonic acid, Hep, L-glycero-D-manno-heptose, Glc, D-glucose, Gal, D-galactose.
Figure \(\PageIndex{23}\) shows an interactive iCn3D model of the E. Coli lipopolysaccharide ABC transporter LptB2FG (6MHU). The left panel shows the membrane protein that binds and transports the LPS (spacefill) from the inner to the outer membrane. The right panel shows the bound LPS. Just part of the inner core is shown since other parts were likely too flexible to be observed. The outer O antigen is not shown since a mutation in the protein prevented it. Six acyl chains are evident. The phosphorylated GlcNAc, KDO, and HEP "layers" are labeled in the right panel.
|
Click the image for a popup or use this external link: https://structure.ncbi.nlm.nih.gov/i...CoPtdH2RVY82P6. Click Style, Background, Transparent in the iCn3D window for a better view. |
Click the image for a popup (instructions below) |
Figure \(\PageIndex{23}\): E. Coli lipopolysaccharide ABC transporter LptB2FG (6MHU) (left panel) and the bound LPS (right panel). (Copyright; author via source).
To see an interactive iCn3D model of the E. Coli LPS from the right panel, follow these steps:
- open iCn3D
- Download this file to your computer's download folder. IMPORTANT: If the file opens as an image in a new browser window, right-click the image and save the file to download it!
- File, Open File, iCn3D png (appendable), and choose the downloaded file.
c. Archaeal Cell Membranes and Walls
We have already discussed that the lipids in Archaeal cell membranes contain L (instead of D) glycerol derivatives and that ether links (more stable in reactive environments) replace ester links with isoprenoid (sometimes branched) chains, replacing fatty acid chains. The cell wall is also quite different, and some don't have one. The type of cell wall depends on the environmental need for stability. They don't contain peptidoglycans. Figure \(\PageIndex{24}\) shows four different types.
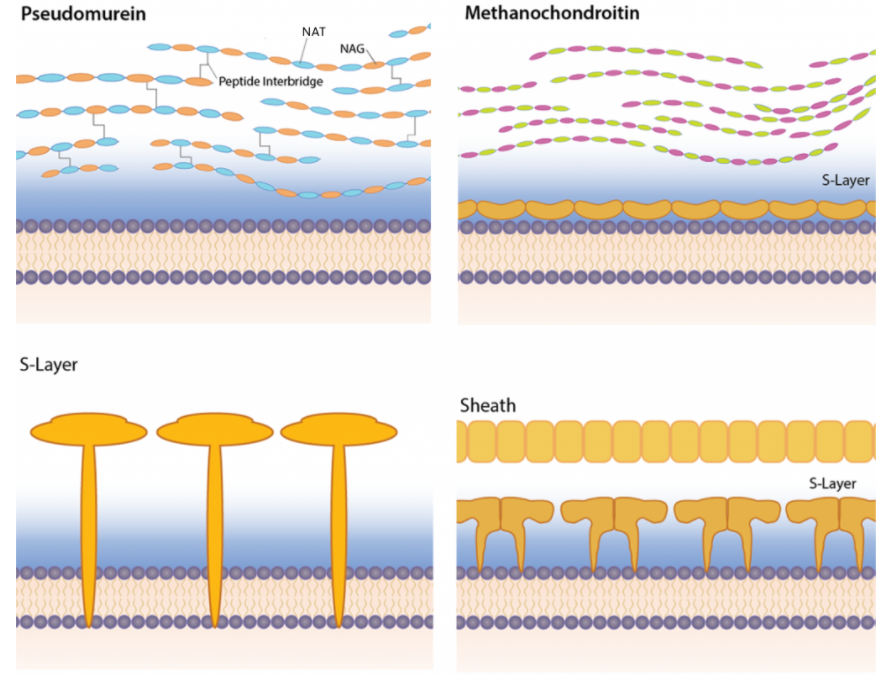
Some differences include the presence of
- pseudomurein - This is the closest to the peptidoglycans presented above. Instead of repeating disaccharide units of (NAM-NAG)n, they have a repeating disaccharide unit of N-acetylalosaminuronic acid (NAT)-NAG. The structure of NAT is shown in Figure \(\PageIndex{25}\).
Figure \(\PageIndex{25}\): N-acetylalosaminuronic acid (NAT)compared to N-acetylmuramic acid (NAM)
- methanochondroitin - This is similar to the glycosaminoglycan chondroitin sulfate
- S-Layer
- Sheath/S-Layer
d. Plant Cell Wall
If you thought bacterial cell walls were complicated, wait until you see plant cell walls! There are about 35 different types of plant cells, and each may have a different cell wall depending on the local needs of a given cell. Cells synthesize thin cell walls that remain thin as the cell grows.
Figure \(\PageIndex{26}\) shows the primary cell wall of plants. The primary cell wall contains cellulose microfibrils (no surprise) and two other polymers, pectin and hemicellulose. The middle lamella, consisting of pectins, is somewhat analogous to the extracellular matrix discussed above.
After cell growth, the cell often synthesizes a secondary cell wall thicker than the first for extra rigidity. The enzymatic machinery for its synthesis is in the cytoplasm and the cell membrane. It is deposited between the cell membrane and the primary cell wall, as shown in the animated image in Figure \(\PageIndex{27}\).
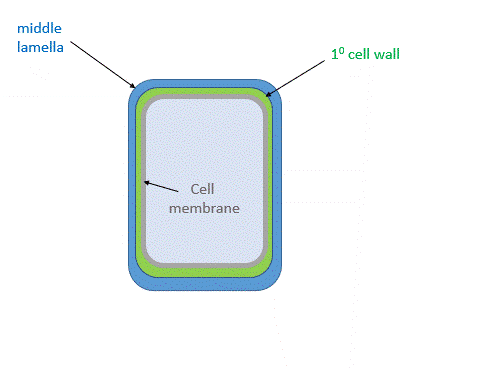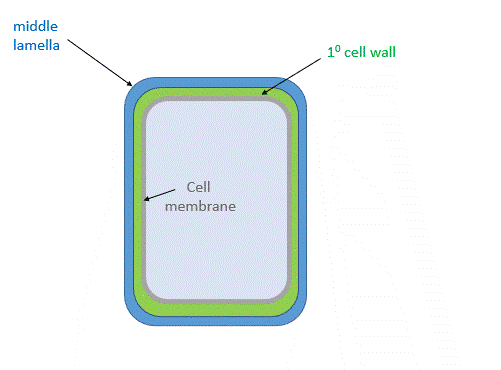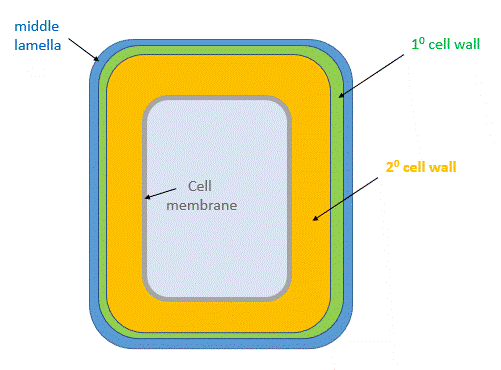
Figure \(\PageIndex{28}\) shows a structural representation of both the primary and secondary cell walls.
The middle lamella, which contains pectins, lignins and some proteins, helps "glue together" the primary cell walls of surrounding plants.
Primary Cell Wall:
The main component of the primary plant wall is the homopolymer cellulose (40% -60% mass) in which the glucose monomers are linked β(1→4)-linked into strands that collect into microfibrils through hydrogen bond interactions. Two other groups of polymers, hemicellulose and pectin, make up the plant cell wall.
Hemicellulose can make up to 20-40% of the mass. These polymers have β(1,4) backbones of glucose, mannose, or xylose (called xyloglucans, xylans, mannans, galactomannans, glucomannans, and galactoglucomannans along with some β(1,3 and 1,4)-glucans. The most abundant hemicellulose in higher plants are the xyloglucans with a cellulose backbone linked at O6 to α-D-xylose. Pectin consists of linked galacturonic acids forming homogalacturonans, rhamnogalacturonans, and rhamnogalacturonans II (RGII) [12] [13]. Homogalacturonans (α1→4) linked D-GalA, making up more than 50% of the pectin
Figure \(\PageIndex{29}\) shows some variants of the cell wall components of a plant.
Secondary Cell Wall
The secondary cell wall's structure depends on the cell's function and environment. It contains cellulose fibers, hemicellulose, and, in addition, a new polymer, lignin. It is abundant in xylem vessels and fiber cells of woody plants. It gives the plant extra stability and new functions, including the transport of fluids within the plant through channels.
Lignins, which can make up to 25% of the biomass weight, are made from phenylalanine derivatives but more directly from cinnamic acid. This derivative is made from phenylalanine, which is hydroxylated and converted through other steps to hydroxycinnamyl alcohols called monolignols, as shown in Figure \(\PageIndex{30}\). Three common monomeric (M) derivatives, p-coumaryl, coniferyl, and sinapyl alcohols, can polymerize into lignins, with the units in the polymer (P) named hydroxyphenyl, guaiacyl, and syringyl, respectively.
Lignols are activated phenolic compounds that form phenoxide free radicals (catalyzed by peroxidase enzymes), which can attack other lignols to form covalent dimers. Reaction mechanisms for dimerizing the MS sinapyl alcohol free radical are shown as an example in Figure \(\PageIndex{31}\).
Figure \(\PageIndex{31}\): Dimers of lignols
Now imagine this polymerization continuing by forming additional phenolic free radicals and coupling at many sites to form a huge covalent lignin polymer. Figure \(\PageIndex{32}\) shows one example of a larger lignin.

Finally, Figure \(\PageIndex{33}\) shows an image of a poplar tree cell wall, made using surface Raman scattering, showing lignin, cellulose, and lipids in secondary xylem cell walls.
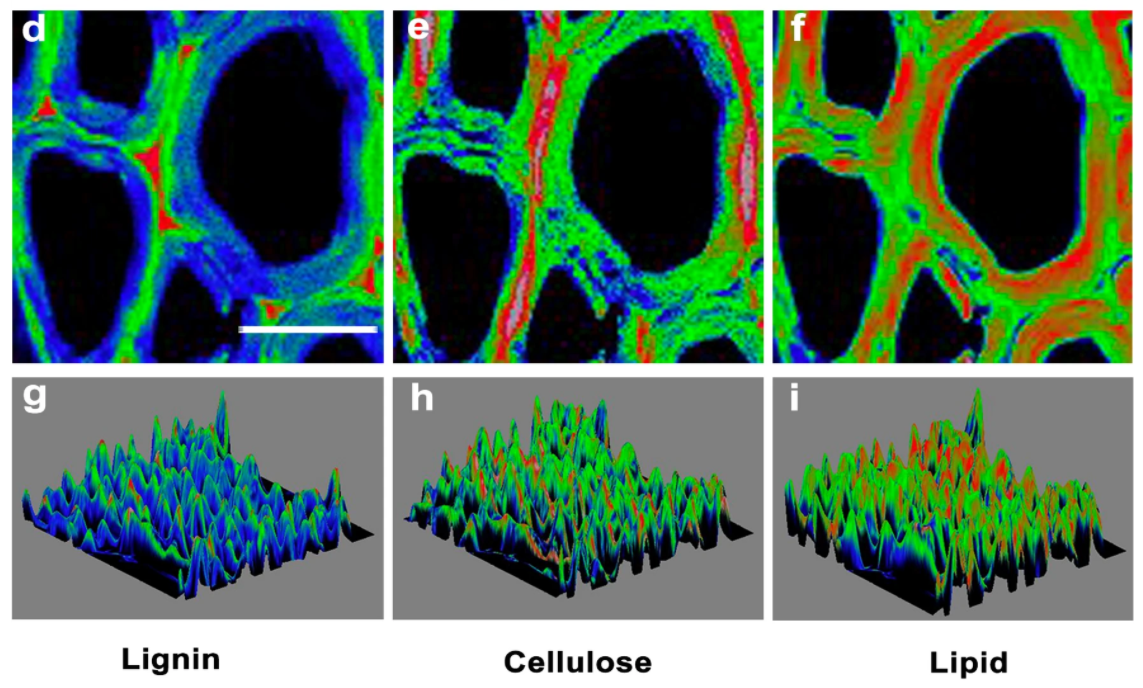
The Extracellular Matrix (ECM) and Basement Membranes
We won't formally discuss cell membranes until Chapter 11, but since anyone reading this book has previously seen biological membranes (including the Gram-negative and positive bilayers discussed above), let's explore a term that most chemistry students, but perhaps not biology students, will find very confusing. That topic is the basement membrane. The basement membrane is encountered so often that we will explore its overall structure here, even though it is not a lipid bilayer. It fits well here since it is a complex structure consisting of proteins and proteoglycans. It's very amorphous, making its structure difficult for those hoping for crystal structures or complex bilayers. It is somewhat similar to the cell wall in functionality. We will offer a cursory explanation. Please visit Introduction to Extracellular Matrix and Cell Adhesion in BioLibre texts for a great overall introduction. Some of the images (when noted) below come from that Cell Biology book chapter.
The extracellular matrix (ECM) is a general term for the large protein and polysaccharide network formed on secretion by some cells in a multicellular organism. They act as connective material to hold cells in a defined space. Cell density can vary greatly between different tissues of an animal, from tightly-packed muscle cells with many direct cell-to-cell contacts to liver tissue, in which some of the cells are only loosely organized, suspended in a web of extracellular matrix, shown in Figure \(\PageIndex{34}\).

The ECM is a generic term encompassing mixtures of polysaccharides and proteins, including collagens, bronectins, laminins, and proteoglycans, all secreted by the cell. The proportions of these components can vary greatly depending on tissue type. Two quite different examples of ECM are the basement membrane underlying the epidermis of the skin, a thin, almost two-dimensional layer that helps to organize the skin cells into a nearly impenetrable barrier to most simple biological insults, and the massive three-dimensional matrix surrounding each chondrocyte in cartilaginous tissue. The ability of the cartilage in your knee to withstand the repeated shock of your footsteps is due to the ECM proteins in which the cells are embedded, not to the actual cells that are, instead, few and sparsely distributed. Although both types of ECM share some components in common, they are distinguishable not just in function or appearance but in the proportions and identity of the constituent molecules
Figure \(\PageIndex{35}\) shows a general structure of the basement membrane. Think of it as an amorphous polymer mixture (somewhat similar to a polyacrylamide gel).

Summary
This chapter explores the multifaceted world of protein glycosylation, highlighting the complex nature and functional significance of carbohydrates attached to proteins. It emphasizes how post-translational modifications by glycans greatly expand the informational content and functional diversity of the proteome, far beyond what is encoded in the genome.
Key Themes:
-
Glycosylation and Its Biological Importance:
The chapter begins by explaining that many proteins destined for secretion or membrane insertion undergo glycosylation. This modification, typically attached to asparagine (N-linked) or serine/threonine (O-linked) side chains, plays critical roles in:- Mediating recognition and binding interactions.
- Preventing aggregation during protein folding.
- Protecting proteins from proteolysis.
- Increasing the half-life of proteins in circulation.
-
N-linked Glycoproteins:
A central focus is placed on N-linked glycosylation, where a core oligosaccharide (Man)3(GlcNAc)2(\text{Man})_3(\text{GlcNAc})_2 is covalently attached to asparagine in a specific consensus sequence. Variants of N-linked glycans include:- High Mannose: Retaining terminal mannose residues.
- Complex: Featuring GlcNAc branches and terminal sialic acid residues.
- Hybrid: Displaying characteristics of both high mannose and complex types. The chapter uses SNFG (Symbolic Nomenclature For Glycans) to depict these structures and discusses their impact on protein function and immune recognition—as seen, for example, on the heavily glycosylated surface of the SARS-CoV-2 spike protein.
-
O-linked Glycoproteins and Blood Group Antigens:
O-linked glycosylation, which attaches sugars to serine or threonine residues (often starting with a Gal(β1,3)GalNAc moiety), is examined next. This section explains how such modifications give rise to blood group antigens and affect cell–cell recognition and immune responses. -
Proteoglycans and the Extracellular Matrix:
The chapter outlines how extensive glycosylation can result in proteoglycans—proteins heavily modified with glycosaminoglycan (GAG) chains. Proteoglycans are major components of the extracellular matrix (ECM) and basement membranes, where they contribute to structural integrity, cell adhesion, and signal transduction. -
Carbohydrate Structures in Cell Walls and Membranes:
The discussion is broadened to cover the roles of carbohydrates in cell wall structures across different organisms:- Bacterial Cell Walls: Differences between Gram-positive and Gram-negative bacteria are highlighted, including the structure of peptidoglycan, the role of teichoic acids, and the unique organization of the outer membrane in Gram-negative species.
- Plant Cell Walls: The synthesis and structure of primary and secondary cell walls, with components such as cellulose, hemicellulose, pectin, and lignin, are described, underscoring their importance in plant rigidity and growth.
- Archaeal Cell Walls: Distinct features of archaeal membranes and cell walls are noted, emphasizing the unique lipid composition and structural adaptations in extreme environments.
-
Clinical Relevance and Glycan Diversity:
The chapter concludes by discussing how variations in glycan structures, such as the presence of Gal(α1,3)Gal, have profound implications for immune recognition and xenotransplantation. It also touches on the role of glycosylation in pathogen-host interactions and cell signaling.
Overall, this chapter provides a comprehensive overview of glycosylation as a key post-translational modification, illustrating how complex carbohydrate structures enrich protein function and influence a wide range of biological processes—from cellular communication to structural support.




.png?revision=1&size=bestfit&width=251&height=265)
.png?revision=1&size=bestfit&width=199&height=294)
Snip.png?revision=1&size=bestfit&width=147&height=290)