14.1: Embryonic Development
- Page ID
- 5135
\( \newcommand{\vecs}[1]{\overset { \scriptstyle \rightharpoonup} {\mathbf{#1}} } \)
\( \newcommand{\vecd}[1]{\overset{-\!-\!\rightharpoonup}{\vphantom{a}\smash {#1}}} \)
\( \newcommand{\dsum}{\displaystyle\sum\limits} \)
\( \newcommand{\dint}{\displaystyle\int\limits} \)
\( \newcommand{\dlim}{\displaystyle\lim\limits} \)
\( \newcommand{\id}{\mathrm{id}}\) \( \newcommand{\Span}{\mathrm{span}}\)
( \newcommand{\kernel}{\mathrm{null}\,}\) \( \newcommand{\range}{\mathrm{range}\,}\)
\( \newcommand{\RealPart}{\mathrm{Re}}\) \( \newcommand{\ImaginaryPart}{\mathrm{Im}}\)
\( \newcommand{\Argument}{\mathrm{Arg}}\) \( \newcommand{\norm}[1]{\| #1 \|}\)
\( \newcommand{\inner}[2]{\langle #1, #2 \rangle}\)
\( \newcommand{\Span}{\mathrm{span}}\)
\( \newcommand{\id}{\mathrm{id}}\)
\( \newcommand{\Span}{\mathrm{span}}\)
\( \newcommand{\kernel}{\mathrm{null}\,}\)
\( \newcommand{\range}{\mathrm{range}\,}\)
\( \newcommand{\RealPart}{\mathrm{Re}}\)
\( \newcommand{\ImaginaryPart}{\mathrm{Im}}\)
\( \newcommand{\Argument}{\mathrm{Arg}}\)
\( \newcommand{\norm}[1]{\| #1 \|}\)
\( \newcommand{\inner}[2]{\langle #1, #2 \rangle}\)
\( \newcommand{\Span}{\mathrm{span}}\) \( \newcommand{\AA}{\unicode[.8,0]{x212B}}\)
\( \newcommand{\vectorA}[1]{\vec{#1}} % arrow\)
\( \newcommand{\vectorAt}[1]{\vec{\text{#1}}} % arrow\)
\( \newcommand{\vectorB}[1]{\overset { \scriptstyle \rightharpoonup} {\mathbf{#1}} } \)
\( \newcommand{\vectorC}[1]{\textbf{#1}} \)
\( \newcommand{\vectorD}[1]{\overrightarrow{#1}} \)
\( \newcommand{\vectorDt}[1]{\overrightarrow{\text{#1}}} \)
\( \newcommand{\vectE}[1]{\overset{-\!-\!\rightharpoonup}{\vphantom{a}\smash{\mathbf {#1}}}} \)
\( \newcommand{\vecs}[1]{\overset { \scriptstyle \rightharpoonup} {\mathbf{#1}} } \)
\(\newcommand{\longvect}{\overrightarrow}\)
\( \newcommand{\vecd}[1]{\overset{-\!-\!\rightharpoonup}{\vphantom{a}\smash {#1}}} \)
\(\newcommand{\avec}{\mathbf a}\) \(\newcommand{\bvec}{\mathbf b}\) \(\newcommand{\cvec}{\mathbf c}\) \(\newcommand{\dvec}{\mathbf d}\) \(\newcommand{\dtil}{\widetilde{\mathbf d}}\) \(\newcommand{\evec}{\mathbf e}\) \(\newcommand{\fvec}{\mathbf f}\) \(\newcommand{\nvec}{\mathbf n}\) \(\newcommand{\pvec}{\mathbf p}\) \(\newcommand{\qvec}{\mathbf q}\) \(\newcommand{\svec}{\mathbf s}\) \(\newcommand{\tvec}{\mathbf t}\) \(\newcommand{\uvec}{\mathbf u}\) \(\newcommand{\vvec}{\mathbf v}\) \(\newcommand{\wvec}{\mathbf w}\) \(\newcommand{\xvec}{\mathbf x}\) \(\newcommand{\yvec}{\mathbf y}\) \(\newcommand{\zvec}{\mathbf z}\) \(\newcommand{\rvec}{\mathbf r}\) \(\newcommand{\mvec}{\mathbf m}\) \(\newcommand{\zerovec}{\mathbf 0}\) \(\newcommand{\onevec}{\mathbf 1}\) \(\newcommand{\real}{\mathbb R}\) \(\newcommand{\twovec}[2]{\left[\begin{array}{r}#1 \\ #2 \end{array}\right]}\) \(\newcommand{\ctwovec}[2]{\left[\begin{array}{c}#1 \\ #2 \end{array}\right]}\) \(\newcommand{\threevec}[3]{\left[\begin{array}{r}#1 \\ #2 \\ #3 \end{array}\right]}\) \(\newcommand{\cthreevec}[3]{\left[\begin{array}{c}#1 \\ #2 \\ #3 \end{array}\right]}\) \(\newcommand{\fourvec}[4]{\left[\begin{array}{r}#1 \\ #2 \\ #3 \\ #4 \end{array}\right]}\) \(\newcommand{\cfourvec}[4]{\left[\begin{array}{c}#1 \\ #2 \\ #3 \\ #4 \end{array}\right]}\) \(\newcommand{\fivevec}[5]{\left[\begin{array}{r}#1 \\ #2 \\ #3 \\ #4 \\ #5 \\ \end{array}\right]}\) \(\newcommand{\cfivevec}[5]{\left[\begin{array}{c}#1 \\ #2 \\ #3 \\ #4 \\ #5 \\ \end{array}\right]}\) \(\newcommand{\mattwo}[4]{\left[\begin{array}{rr}#1 \amp #2 \\ #3 \amp #4 \\ \end{array}\right]}\) \(\newcommand{\laspan}[1]{\text{Span}\{#1\}}\) \(\newcommand{\bcal}{\cal B}\) \(\newcommand{\ccal}{\cal C}\) \(\newcommand{\scal}{\cal S}\) \(\newcommand{\wcal}{\cal W}\) \(\newcommand{\ecal}{\cal E}\) \(\newcommand{\coords}[2]{\left\{#1\right\}_{#2}}\) \(\newcommand{\gray}[1]{\color{gray}{#1}}\) \(\newcommand{\lgray}[1]{\color{lightgray}{#1}}\) \(\newcommand{\rank}{\operatorname{rank}}\) \(\newcommand{\row}{\text{Row}}\) \(\newcommand{\col}{\text{Col}}\) \(\renewcommand{\row}{\text{Row}}\) \(\newcommand{\nul}{\text{Nul}}\) \(\newcommand{\var}{\text{Var}}\) \(\newcommand{\corr}{\text{corr}}\) \(\newcommand{\len}[1]{\left|#1\right|}\) \(\newcommand{\bbar}{\overline{\bvec}}\) \(\newcommand{\bhat}{\widehat{\bvec}}\) \(\newcommand{\bperp}{\bvec^\perp}\) \(\newcommand{\xhat}{\widehat{\xvec}}\) \(\newcommand{\vhat}{\widehat{\vvec}}\) \(\newcommand{\uhat}{\widehat{\uvec}}\) \(\newcommand{\what}{\widehat{\wvec}}\) \(\newcommand{\Sighat}{\widehat{\Sigma}}\) \(\newcommand{\lt}{<}\) \(\newcommand{\gt}{>}\) \(\newcommand{\amp}{&}\) \(\definecolor{fillinmathshade}{gray}{0.9}\)In animals, one can usually distinguish 4 stages of embryonic development.
- Cleavage
- Patterning
- Differentiation
- Growth
Cleavage
Mitosis and cytokinesis of the zygote, an unusually large cell, produces an increasing number of smaller cells, each with an exact copy of the genome present in the zygote. However, the genes of the zygote are not expressed at first. The early activities of cleavage are controlled by the mother's genome; that is, by mRNAs and proteins she deposited in the unfertilized egg. In humans, the switch-over occurs after 4–8 cells have been produced; in frogs not until thousands of cells have been produced. Cleavage ends with the formation of a blastula.
Patterning
During this phase, the cells produced by cleavage organize themselves in layers and masses, a process called gastrulation. The pattern of the future animal appears:
- front to rear (the anterior-posterior axis)
- back side and belly side (its dorsal-ventral axis)
- left and right sides.
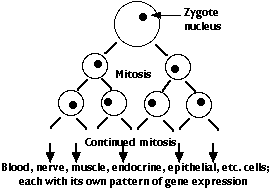
The genome of the zygote contains all the genes needed to make the hundreds of different types of cells that will make up the complete animal. There are two major categories of these genes:
- "housekeeping" genes = those that encode the RNAs and proteins needed by all kinds of cells. Examples:
- genes for tRNAs, rRNAs
- genes encoding the enzymes of glycolysis.
- tissue-specific genes = those that encode mRNAs and hence proteins that are used by one or a few specific kinds of cell. Examples:
- genes for hemoglobin expressed in the precursors of red blood cells
- the gene for insulin expressed in the beta cells of the islets of Langerhans
However, every cell descended from the zygote has been produced by mitosis and thus contains the complete genome of the organism (with a very few exceptions).
Two pieces of evidence:
- Dolly - Dolly is the sheep that was formed by inserting a nucleus from a single cell of an adult sheep into an enucleated sheep egg. She proves that the cell from the adult had lost none of the genes needed to build all the tissues of a sheep.
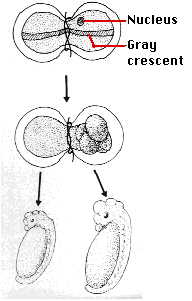
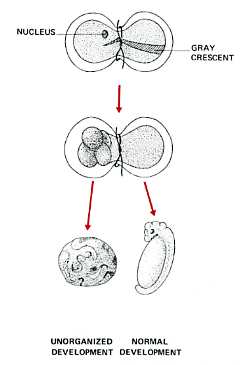
- Spemann's egg-tying experiments - Many years earlier, the German embryologist Hans Spemann demonstrated the same truth. He used strands of baby hair to tie loops around fertilized salamander eggs. Although the egg half with the nucleus began cleaving normally, the other side did not begin cleavage until a nucleus finally slipped through the knot. So long as the egg was tied so that both halves contained some of the gray crescent, the second half began normal cleavage and ultimately produced a second tadpole (right). Even after 5 mitotic division of the zygote nucleus (the 32-cell stage), the entire genome was still available in each descendant nucleus.
- A fertilized egg is much larger than the normal cells of an animal's body. Some (e.g., a hen's egg) are truly huge. The frog egg has a volume 1.6 millions times larger than a typical frog cell. The photo is of a 16-cell frog embryo. This mass of cells is no larger than the original egg. The eggs of mammals are smaller, but even they are larger than their descendant cells will be.
- The cytoplasm of the fertilized egg is not homogeneous. It contains gradients of mRNAs and proteins. These are the products of the mother's genes and were deposited in the egg by her.
- Cleavage of the fertilized egg partitions it into thousands of cells of normal size. Each contains a nucleus descended from the zygote nucleus.
- But each nucleus finds itself partitioned off in cytoplasm containing a particular mix of mRNAs and proteins.
- When the frog blastula has produced some 4,000 cells, transcription and translation of its nuclear genes begins (and the mother's mRNA molecules, that up to now have been the source of all protein synthesis, are destroyed).
- The genes that are expressed by the nucleus in a given cell are regulated by the molecules, mostly protein transcription factors and microRNAs (miRNAs), found in the cytoplasm surrounding that nucleus.
- Once a cell-specific pattern of gene expression is launched, that cell may release molecules that regulate the genes of nearby cells.
- In this way, the foundation is laid for the building of an organism with hundreds of types of differentiated cells — each in its correct location and performing its correct functions.
Xenopus
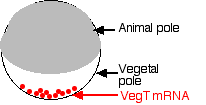
- During egg formation, molecules of mRNA encoding the protein VegT are deposited at the vegetal pole of the cell.
- Cells that form there during cleavage translate the mRNA into the VegT protein.
- VegT is a transcription factor that turns on genes that produce members of the transforming growth factor-beta (TGF-β) family (e.g., activin).
- These proteins are needed for cells to start down the path to becoming mesoderm.
- Some of those cells will, in turn, become the Spemann organizer.
- Later, the Spemann organizer will secrete molecules that induce the ectodermal cells above them to develop into the tissues of the brain and spinal cord.
Demonstration
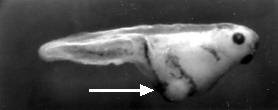

Inject the anterior of the fertilized egg with nanos mRNA. The result: another double-posterior larva.
Make female fruit flies that are transgenic for a recombinant gene containing:
- the gene for nanos
- coupled to the 3´ anterior-directing signal of the bicoid gene.
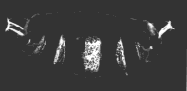
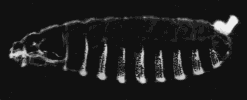
A normal larva is shown on the right. The bright object at the right end of the normal larva and at both ends of the double posterior larva is the tip of the tail. These micrographs are courtesy of Elizabeth Gavis and Ruth Lehmann, in whose lab the third demonstration was performed.
The Mud Snail
The mud snail, Ilyanassa obsoleta, is a small gastropod that lives in mud flats along the Atlantic coast.
Like other protostomes, cleavage of the zygote produces daughter cells that are already committed to their fate. In other words, even as early as the two-cell stage, the cells are no longer totipotent. Unlike humans and other deuterostomes, then, identical twins cannot form.
In the 12 December 2002 issue of Nature, J. David Lambert and Lisa Nagy reported another mechanism by which two daughter cells become committed to different fates even though they have inherited the same genome.
They traced the distribution in the cells of early embryos of the messenger RNAs (mRNAs) encoding 3 proteins that are known to be important in the development of other animals such as Xenopus and Drosophila.
- IoEve, which is Ilyanassa obsoleta's version of even-skipped (eve) in Drosophila;
- IoDpp, which is the snail's version of
- decapentaplegic (dpp) in Drosophila and the genes encoding
- bone morphogenic proteins (BMP2 and BMP4) in vertebrates
- IoTld, which encodes the snail's version of a protein called tolloid in Drosophila.
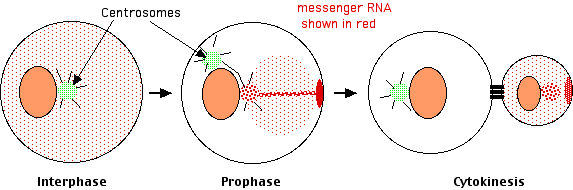
Lambert and Nagy found that
- in interphase the messenger RNAs were distributed diffusely throughout the cytosol, but
- as the cell got ready for cleavage, the mRNAs collected at only one of the now pair of centrosomes. They were collected there by traveling along the microtubules that radiate out from the centrosome.
- As cleavage continued, the mRNAs moved from the centrosome to a spot on the inner surface of the plasma membrane. They got there by traveling along actin filaments.
- At cytokinesis, this patch of accumulated mRNAs was incorporated exclusively into the smaller daughter cell.
Centrosome sorting (of proteins in this case) also plays a role in determining whether embryonic cells of Caenorhabditis elegans remain in the germline or become the somatic cells of the worm.
What comes next?
Development in Xenopus and Drosophila passes through three rather different (although often overlapping) phases:
- Establishing the main axes (anterior-posterior; dorsal-ventral; left-right). This is done by gradients of mRNAs and proteins encoded by the mother's genes and placed in the egg by her. It has been discussed here.
- Establishing the main body parts such as the notochord and central nervous system in vertebratesand the segments in DrosophilaThese are run by genes of the zygote itself.
- Filling in the details; that is, building the various organs of the animal.


