7.2: The Cell Cycle
- Page ID
- 4846
\( \newcommand{\vecs}[1]{\overset { \scriptstyle \rightharpoonup} {\mathbf{#1}} } \)
\( \newcommand{\vecd}[1]{\overset{-\!-\!\rightharpoonup}{\vphantom{a}\smash {#1}}} \)
\( \newcommand{\dsum}{\displaystyle\sum\limits} \)
\( \newcommand{\dint}{\displaystyle\int\limits} \)
\( \newcommand{\dlim}{\displaystyle\lim\limits} \)
\( \newcommand{\id}{\mathrm{id}}\) \( \newcommand{\Span}{\mathrm{span}}\)
( \newcommand{\kernel}{\mathrm{null}\,}\) \( \newcommand{\range}{\mathrm{range}\,}\)
\( \newcommand{\RealPart}{\mathrm{Re}}\) \( \newcommand{\ImaginaryPart}{\mathrm{Im}}\)
\( \newcommand{\Argument}{\mathrm{Arg}}\) \( \newcommand{\norm}[1]{\| #1 \|}\)
\( \newcommand{\inner}[2]{\langle #1, #2 \rangle}\)
\( \newcommand{\Span}{\mathrm{span}}\)
\( \newcommand{\id}{\mathrm{id}}\)
\( \newcommand{\Span}{\mathrm{span}}\)
\( \newcommand{\kernel}{\mathrm{null}\,}\)
\( \newcommand{\range}{\mathrm{range}\,}\)
\( \newcommand{\RealPart}{\mathrm{Re}}\)
\( \newcommand{\ImaginaryPart}{\mathrm{Im}}\)
\( \newcommand{\Argument}{\mathrm{Arg}}\)
\( \newcommand{\norm}[1]{\| #1 \|}\)
\( \newcommand{\inner}[2]{\langle #1, #2 \rangle}\)
\( \newcommand{\Span}{\mathrm{span}}\) \( \newcommand{\AA}{\unicode[.8,0]{x212B}}\)
\( \newcommand{\vectorA}[1]{\vec{#1}} % arrow\)
\( \newcommand{\vectorAt}[1]{\vec{\text{#1}}} % arrow\)
\( \newcommand{\vectorB}[1]{\overset { \scriptstyle \rightharpoonup} {\mathbf{#1}} } \)
\( \newcommand{\vectorC}[1]{\textbf{#1}} \)
\( \newcommand{\vectorD}[1]{\overrightarrow{#1}} \)
\( \newcommand{\vectorDt}[1]{\overrightarrow{\text{#1}}} \)
\( \newcommand{\vectE}[1]{\overset{-\!-\!\rightharpoonup}{\vphantom{a}\smash{\mathbf {#1}}}} \)
\( \newcommand{\vecs}[1]{\overset { \scriptstyle \rightharpoonup} {\mathbf{#1}} } \)
\(\newcommand{\longvect}{\overrightarrow}\)
\( \newcommand{\vecd}[1]{\overset{-\!-\!\rightharpoonup}{\vphantom{a}\smash {#1}}} \)
\(\newcommand{\avec}{\mathbf a}\) \(\newcommand{\bvec}{\mathbf b}\) \(\newcommand{\cvec}{\mathbf c}\) \(\newcommand{\dvec}{\mathbf d}\) \(\newcommand{\dtil}{\widetilde{\mathbf d}}\) \(\newcommand{\evec}{\mathbf e}\) \(\newcommand{\fvec}{\mathbf f}\) \(\newcommand{\nvec}{\mathbf n}\) \(\newcommand{\pvec}{\mathbf p}\) \(\newcommand{\qvec}{\mathbf q}\) \(\newcommand{\svec}{\mathbf s}\) \(\newcommand{\tvec}{\mathbf t}\) \(\newcommand{\uvec}{\mathbf u}\) \(\newcommand{\vvec}{\mathbf v}\) \(\newcommand{\wvec}{\mathbf w}\) \(\newcommand{\xvec}{\mathbf x}\) \(\newcommand{\yvec}{\mathbf y}\) \(\newcommand{\zvec}{\mathbf z}\) \(\newcommand{\rvec}{\mathbf r}\) \(\newcommand{\mvec}{\mathbf m}\) \(\newcommand{\zerovec}{\mathbf 0}\) \(\newcommand{\onevec}{\mathbf 1}\) \(\newcommand{\real}{\mathbb R}\) \(\newcommand{\twovec}[2]{\left[\begin{array}{r}#1 \\ #2 \end{array}\right]}\) \(\newcommand{\ctwovec}[2]{\left[\begin{array}{c}#1 \\ #2 \end{array}\right]}\) \(\newcommand{\threevec}[3]{\left[\begin{array}{r}#1 \\ #2 \\ #3 \end{array}\right]}\) \(\newcommand{\cthreevec}[3]{\left[\begin{array}{c}#1 \\ #2 \\ #3 \end{array}\right]}\) \(\newcommand{\fourvec}[4]{\left[\begin{array}{r}#1 \\ #2 \\ #3 \\ #4 \end{array}\right]}\) \(\newcommand{\cfourvec}[4]{\left[\begin{array}{c}#1 \\ #2 \\ #3 \\ #4 \end{array}\right]}\) \(\newcommand{\fivevec}[5]{\left[\begin{array}{r}#1 \\ #2 \\ #3 \\ #4 \\ #5 \\ \end{array}\right]}\) \(\newcommand{\cfivevec}[5]{\left[\begin{array}{c}#1 \\ #2 \\ #3 \\ #4 \\ #5 \\ \end{array}\right]}\) \(\newcommand{\mattwo}[4]{\left[\begin{array}{rr}#1 \amp #2 \\ #3 \amp #4 \\ \end{array}\right]}\) \(\newcommand{\laspan}[1]{\text{Span}\{#1\}}\) \(\newcommand{\bcal}{\cal B}\) \(\newcommand{\ccal}{\cal C}\) \(\newcommand{\scal}{\cal S}\) \(\newcommand{\wcal}{\cal W}\) \(\newcommand{\ecal}{\cal E}\) \(\newcommand{\coords}[2]{\left\{#1\right\}_{#2}}\) \(\newcommand{\gray}[1]{\color{gray}{#1}}\) \(\newcommand{\lgray}[1]{\color{lightgray}{#1}}\) \(\newcommand{\rank}{\operatorname{rank}}\) \(\newcommand{\row}{\text{Row}}\) \(\newcommand{\col}{\text{Col}}\) \(\renewcommand{\row}{\text{Row}}\) \(\newcommand{\nul}{\text{Nul}}\) \(\newcommand{\var}{\text{Var}}\) \(\newcommand{\corr}{\text{corr}}\) \(\newcommand{\len}[1]{\left|#1\right|}\) \(\newcommand{\bbar}{\overline{\bvec}}\) \(\newcommand{\bhat}{\widehat{\bvec}}\) \(\newcommand{\bperp}{\bvec^\perp}\) \(\newcommand{\xhat}{\widehat{\xvec}}\) \(\newcommand{\vhat}{\widehat{\vvec}}\) \(\newcommand{\uhat}{\widehat{\uvec}}\) \(\newcommand{\what}{\widehat{\wvec}}\) \(\newcommand{\Sighat}{\widehat{\Sigma}}\) \(\newcommand{\lt}{<}\) \(\newcommand{\gt}{>}\) \(\newcommand{\amp}{&}\) \(\definecolor{fillinmathshade}{gray}{0.9}\)A eukaryotic cell cannot divide into two, the two into four, etc. unless two processes alternate:
- doubling of its genome (DNA) in S phase (synthesis phase) of the cell cycle;
- halving of that genome during mitosis (M phase).
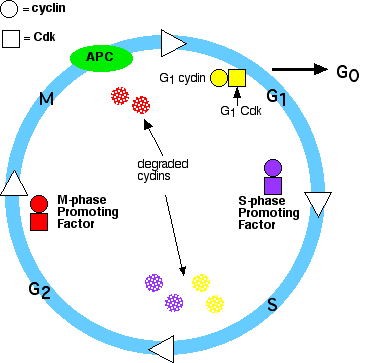
Control of the Cell Cycle
The passage of a cell through the cell cycle is controlled by proteins in the cytoplasm. Among the main players in animal cells are:
- Their levels in the cell rise and fall with the stages of the cell cycle.
- Their levels in the cell remain fairly stable, but each must bind the appropriate cyclin (whose levels fluctuate) in order to be activated. They add phosphate groups to a variety of protein substrates that control processes in the cell cycle.
- The anaphase-promoting complex (APC). (The APC is also called the cyclosome, and the complex is often designated as the APC/C.) The APC/C
- triggers the events leading to destruction of cohesin (as described below) thus allowing the sister chromatids to separate
- degrades the mitotic (B) cyclins
Steps in the cycle
- A rising level of G1-cyclins bind to their Cdks and signal the cell to prepare the chromosomes for replication.
- A rising level of S-phase promoting factor (SPF) — which includes A cyclins bound to Cdk2 — enters the nucleus and prepares the cell to duplicate its DNA (and its centrosomes).
- As DNA replication continues, cyclin E is destroyed, and the level of mitotic cyclins begins to rise (in G2).
- Translocation of M-phase promoting factor (the complex of mitotic [B] cyclins with the M-phase Cdk [Cdk1]) into the nucleus initiates
- assembly of the mitotic spindle
- breakdown of the nuclear envelope
- cessation of all gene transcription
- condensation of the chromosomes
- These events take the cell to metaphase of mitosis.
- At this point, the M-phase promoting factor activates the anaphase-promoting complex (APC/C) which
- allows the sister chromatids at the metaphase plate to separate and move to the poles (= anaphase), completing mitosis.
Separation of the sister chromatids depends on the breakdown of the cohesin that has been holding them together. It works like this.
- Cohesin breakdown is caused by a protease called separase (also known as separin).
- Separase is kept inactive until late metaphase by an inhibitory chaperone called securin.
- Anaphase begins when the anaphase promoting complex (APC/C) destroys securin (by tagging it with ubiquitin for deposit in a proteasome) thus ending its inhibition of separase and allowing
- separase to break down cohesin
- destroys B cyclins. This is also done by attaching them to ubiquitin which targets them for destruction by proteasomes.
- turns on synthesis of G1 cyclins (D) for the next turn of the cycle.
- degrades geminin, a protein that has kept the freshly-synthesized DNA in S phase from being re-replicated before mitosis.
This is only one of the mechanisms by which the cell ensures that every portion of its genome is copied once — and only once — during S phase
Some cells deliberately cut the cell cycle short allowing repeated S phases without completing mitosis and/or cytokinesis. This is called endoreplication.
Meiosis and the Cell Cycle
The special behavior of the chromosomes in meiosis I requires some special controls. Nonetheless, passage through the cell cycle in meiosis I (as well as meiosis II, which is essentially a mitotic division) uses many of the same players, e.g., MPF and APC. (In fact, MPF is also called maturation-promoting factor for its role in meiosis I and II of developing oocytes.
Quality Control of the Cell Cycle
The cell has several systems for interrupting the cell cycle if something goes wrong.
DNA damage checkpoints. These sense DNA damage both before the cell enters S phase (a G1 checkpoint) as well as after S phase (a G2 checkpoint). Damage to DNA before the cell enters S phase inhibits the action of Cdk2 thus stopping the progression of the cell cycle until the damage can be repaired. If the damage is so severe that it cannot be repaired, the cell self-destructs by apoptosis. Damage to DNA after S phase (the G2 checkpoint), inhibits the action of Cdk1 thus preventing the cell from proceeding from G2 to mitosis. A check on the successful replication of DNA during S phase. If replication stops at any point on the DNA, progress through the cell cycle is halted until the problem is solved.
Spindle checkpoints. Some of these that have been discovered to detect any failure of spindle fibers to attach to kinetochores and arrest the cell in metaphase until all the kinetochores are attached correctly. They detect improper alignment of the spindle itself and block cytokinesis. Furthermore, they trigger apoptosis if the damage is irreparable. All the checkpoints examined require the services of a complex of proteins. Mutations in the genes encoding some of these have been associated with cancer; that is, they are oncogenes. This should not be surprising since checkpoint failures allow the cell to continue dividing despite damage to its integrity.
Examples of checkpoints
p53
The p53 protein senses DNA damage and can halt progression of the cell cycle in G1 (by blocking the activity of Cdk2). Both copies of the p53 gene must be mutated for this to fail so mutations in p53 are recessive, and p53 qualifies as a tumor suppressor gene.
The p53 protein is also a key player in apoptosis, forcing "bad" cells to commit suicide. So if the cell has only mutant versions of the protein, it can live on — perhaps developing into a cancer. More than half of all human cancers do, in fact, harbor p53 mutations and have no functioning p53 protein.


