3.6: Golgi Apparatus
- Page ID
- 3970
\( \newcommand{\vecs}[1]{\overset { \scriptstyle \rightharpoonup} {\mathbf{#1}} } \)
\( \newcommand{\vecd}[1]{\overset{-\!-\!\rightharpoonup}{\vphantom{a}\smash {#1}}} \)
\( \newcommand{\dsum}{\displaystyle\sum\limits} \)
\( \newcommand{\dint}{\displaystyle\int\limits} \)
\( \newcommand{\dlim}{\displaystyle\lim\limits} \)
\( \newcommand{\id}{\mathrm{id}}\) \( \newcommand{\Span}{\mathrm{span}}\)
( \newcommand{\kernel}{\mathrm{null}\,}\) \( \newcommand{\range}{\mathrm{range}\,}\)
\( \newcommand{\RealPart}{\mathrm{Re}}\) \( \newcommand{\ImaginaryPart}{\mathrm{Im}}\)
\( \newcommand{\Argument}{\mathrm{Arg}}\) \( \newcommand{\norm}[1]{\| #1 \|}\)
\( \newcommand{\inner}[2]{\langle #1, #2 \rangle}\)
\( \newcommand{\Span}{\mathrm{span}}\)
\( \newcommand{\id}{\mathrm{id}}\)
\( \newcommand{\Span}{\mathrm{span}}\)
\( \newcommand{\kernel}{\mathrm{null}\,}\)
\( \newcommand{\range}{\mathrm{range}\,}\)
\( \newcommand{\RealPart}{\mathrm{Re}}\)
\( \newcommand{\ImaginaryPart}{\mathrm{Im}}\)
\( \newcommand{\Argument}{\mathrm{Arg}}\)
\( \newcommand{\norm}[1]{\| #1 \|}\)
\( \newcommand{\inner}[2]{\langle #1, #2 \rangle}\)
\( \newcommand{\Span}{\mathrm{span}}\) \( \newcommand{\AA}{\unicode[.8,0]{x212B}}\)
\( \newcommand{\vectorA}[1]{\vec{#1}} % arrow\)
\( \newcommand{\vectorAt}[1]{\vec{\text{#1}}} % arrow\)
\( \newcommand{\vectorB}[1]{\overset { \scriptstyle \rightharpoonup} {\mathbf{#1}} } \)
\( \newcommand{\vectorC}[1]{\textbf{#1}} \)
\( \newcommand{\vectorD}[1]{\overrightarrow{#1}} \)
\( \newcommand{\vectorDt}[1]{\overrightarrow{\text{#1}}} \)
\( \newcommand{\vectE}[1]{\overset{-\!-\!\rightharpoonup}{\vphantom{a}\smash{\mathbf {#1}}}} \)
\( \newcommand{\vecs}[1]{\overset { \scriptstyle \rightharpoonup} {\mathbf{#1}} } \)
\(\newcommand{\longvect}{\overrightarrow}\)
\( \newcommand{\vecd}[1]{\overset{-\!-\!\rightharpoonup}{\vphantom{a}\smash {#1}}} \)
\(\newcommand{\avec}{\mathbf a}\) \(\newcommand{\bvec}{\mathbf b}\) \(\newcommand{\cvec}{\mathbf c}\) \(\newcommand{\dvec}{\mathbf d}\) \(\newcommand{\dtil}{\widetilde{\mathbf d}}\) \(\newcommand{\evec}{\mathbf e}\) \(\newcommand{\fvec}{\mathbf f}\) \(\newcommand{\nvec}{\mathbf n}\) \(\newcommand{\pvec}{\mathbf p}\) \(\newcommand{\qvec}{\mathbf q}\) \(\newcommand{\svec}{\mathbf s}\) \(\newcommand{\tvec}{\mathbf t}\) \(\newcommand{\uvec}{\mathbf u}\) \(\newcommand{\vvec}{\mathbf v}\) \(\newcommand{\wvec}{\mathbf w}\) \(\newcommand{\xvec}{\mathbf x}\) \(\newcommand{\yvec}{\mathbf y}\) \(\newcommand{\zvec}{\mathbf z}\) \(\newcommand{\rvec}{\mathbf r}\) \(\newcommand{\mvec}{\mathbf m}\) \(\newcommand{\zerovec}{\mathbf 0}\) \(\newcommand{\onevec}{\mathbf 1}\) \(\newcommand{\real}{\mathbb R}\) \(\newcommand{\twovec}[2]{\left[\begin{array}{r}#1 \\ #2 \end{array}\right]}\) \(\newcommand{\ctwovec}[2]{\left[\begin{array}{c}#1 \\ #2 \end{array}\right]}\) \(\newcommand{\threevec}[3]{\left[\begin{array}{r}#1 \\ #2 \\ #3 \end{array}\right]}\) \(\newcommand{\cthreevec}[3]{\left[\begin{array}{c}#1 \\ #2 \\ #3 \end{array}\right]}\) \(\newcommand{\fourvec}[4]{\left[\begin{array}{r}#1 \\ #2 \\ #3 \\ #4 \end{array}\right]}\) \(\newcommand{\cfourvec}[4]{\left[\begin{array}{c}#1 \\ #2 \\ #3 \\ #4 \end{array}\right]}\) \(\newcommand{\fivevec}[5]{\left[\begin{array}{r}#1 \\ #2 \\ #3 \\ #4 \\ #5 \\ \end{array}\right]}\) \(\newcommand{\cfivevec}[5]{\left[\begin{array}{c}#1 \\ #2 \\ #3 \\ #4 \\ #5 \\ \end{array}\right]}\) \(\newcommand{\mattwo}[4]{\left[\begin{array}{rr}#1 \amp #2 \\ #3 \amp #4 \\ \end{array}\right]}\) \(\newcommand{\laspan}[1]{\text{Span}\{#1\}}\) \(\newcommand{\bcal}{\cal B}\) \(\newcommand{\ccal}{\cal C}\) \(\newcommand{\scal}{\cal S}\) \(\newcommand{\wcal}{\cal W}\) \(\newcommand{\ecal}{\cal E}\) \(\newcommand{\coords}[2]{\left\{#1\right\}_{#2}}\) \(\newcommand{\gray}[1]{\color{gray}{#1}}\) \(\newcommand{\lgray}[1]{\color{lightgray}{#1}}\) \(\newcommand{\rank}{\operatorname{rank}}\) \(\newcommand{\row}{\text{Row}}\) \(\newcommand{\col}{\text{Col}}\) \(\renewcommand{\row}{\text{Row}}\) \(\newcommand{\nul}{\text{Nul}}\) \(\newcommand{\var}{\text{Var}}\) \(\newcommand{\corr}{\text{corr}}\) \(\newcommand{\len}[1]{\left|#1\right|}\) \(\newcommand{\bbar}{\overline{\bvec}}\) \(\newcommand{\bhat}{\widehat{\bvec}}\) \(\newcommand{\bperp}{\bvec^\perp}\) \(\newcommand{\xhat}{\widehat{\xvec}}\) \(\newcommand{\vhat}{\widehat{\vvec}}\) \(\newcommand{\uhat}{\widehat{\uvec}}\) \(\newcommand{\what}{\widehat{\wvec}}\) \(\newcommand{\Sighat}{\widehat{\Sigma}}\) \(\newcommand{\lt}{<}\) \(\newcommand{\gt}{>}\) \(\newcommand{\amp}{&}\) \(\definecolor{fillinmathshade}{gray}{0.9}\)The Golgi apparatus is a cell structure mainly devoted to processing the proteins synthesized in the endoplasmic reticulum (ER). Some of these will eventually end up as integral membrane proteins embedded in the plasma membrane. Other proteins moving through the Golgi will end up in lysosomes or be secreted by exocytosis (e.g., digestive enzymes). The major processing activity is glycosylation: the adding of sugar molecules to form glycoproteins. In some cells, e.g., mucus-secreting cells in epithelia, the amount of carbohydrate so far exceeds that of the protein that the product is called a mucopolysaccharide (also known as a proteoglycan). In plant cells, the Golgi secretes the cell plate and cell wall.
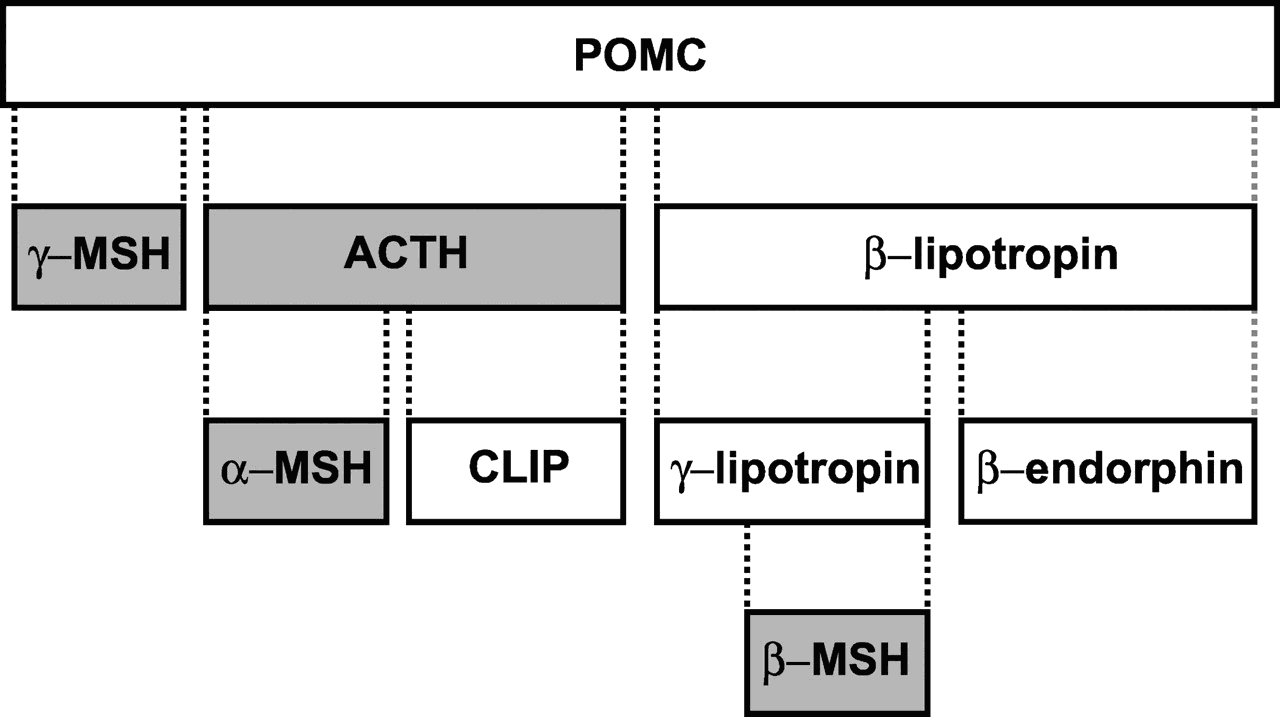
Small peptides, e.g., some hormones and neurotransmitters, are typically too small to be synthesized directly by ribosomes. Instead, the ribosomes on the ER synthesize a large precursor protein that is later cut up into small peptide fragments as it traverses the Golgi. For example, proopiomelanocortin (POMC) is a polypeptide of 241 amino acids from which is cut ACTH, alpha and beta MSH, beta- endorphin, and others. POMC is cleaved to give rise to multiple peptide hormones.
- α-MSH produced by neurons in the arcuate nucleus has important roles in the regulation of appetite and sexual behavior, while α-MSH secreted from the intermediate lobe of the pituitary regulates the production of melanin.
- ACTH is a peptide hormone that regulates the secretion of glucocorticoids from the adrenal cortex.
- β-Endorphin and [Met]enkephalin are endogenous opioid peptides with widespread actions in the brain.
The Golgi consists of a stack of membrane-bounded cisternae located between the endoplasmic reticulum and the cell surface (Figure \(\PageIndex{2}\)). Many different enzymes (proteins) are present in the Golgi to perform its various synthetic activities. So there must be mechanisms to sort out the processed proteins and send them on to their destinations while reclaiming processing proteins (e.g., glycosylases) for reuse.
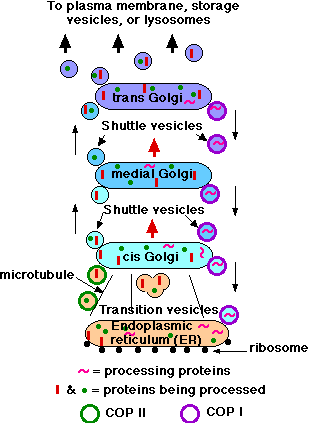
The Outbound Path (Membrane Fission)
Two mechanisms appear to participate in the migration of proteins from the endoplasmic reticulum through the Golgi apparatus.
- Mechanism 1: Transition vesicles pinch off from the surface of the endoplasmic reticulum carrying integral membrane proteins, soluble proteins awaiting processing, and processing enzymes. Pinching off requires that the vesicle be coated with COPII (Coat Protein II). The transition vesicles move toward the cis Golgi on microtubules. As they do so, their COPII coat is removed and they may fuse together forming larger vesicles; these fuse with the cis Golgi. Sugars are added to proteins in small packets so many glycoproteins have to undergo a large number of sequential steps of glycosylation, each requiring its own enzymes. These steps take place as shuttle vesicles carry the proteins from cis to medial to the trans Golgi compartments. At the outer face of the trans Golgi, vesicles pinch off and carry their completed products to their various destinations.
- Mechanism 2: In addition to the pinching off and fusing of shuttle vesicles, the cisternae of the Golgi actually migrate themselves, that is, the cis Golgi gradually migrates up the stack becoming a medial and finally a trans Golgi (Figure \(\PageIndex{3}\)).
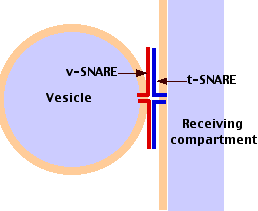
The Inbound Path (Membrane Fusion)
The movement of cisternal contents through the stack means that essential processing enzymes are also moving away from their proper site of action. Using a variety of signals, the Golgi separates the products from the processing enzymes that made them and returns the enzymes back to the endoplasmic reticulum. This transport is also done by pinching off vesicles, but the inbound vesicles are coated with COPI (coat protein I). A vesicle recognize its correct target by involving pairs of complementary integral membrane proteins:
- v-SNAREs = "vesicle SNAREs" — on the vesicle surface
- t-SNAREs = "target SNAREs" — on the surface of the target membrane
v-SNAREs and t-SNAREs bind specifically to each other thanks to the complementary structure of their surface domains. Binding is followed by fusion of the two membranes (Figure \(\PageIndex{4}\)).
The Golgi breaks up and disappears at the onset of mitosis. By telophase of mitosis, the Golgi reappears. How it is recreated is still uncertain.


