16.2A: Xylem
- Page ID
- 5782
Most plants secure the water and minerals they need from their roots. The path taken is:
\[\text{soil} \rightarrow \text{roots} \rightarrow \text{stems} \rightarrow \text{leaves}\]
The minerals (e.g., K+, Ca2+) travel dissolved in the water (often accompanied by various organic molecules supplied by root cells), but less than 1% of the water reaching the leaves is used in photosynthesis and plant growth. Most of it is lost in transpiration, which serve two useful functions- it provides the force for lifting the water up the stems and it cools the leaves. Water and minerals enter the root by separate paths which eventually converge in the stele.
The Pathway of Water and Minerals
Soil water enters the root through its epidermis. It appears that water then travels in both the cytoplasm of root cells - called the symplast (i.e., it crosses the plasma membrane and then passes from cell to cell through plasmodesmata) and in the nonliving parts of the root - called the apoplast (i.e., in the spaces between the cells and in the cells walls themselves. This water has not crossed a plasma membrane. However, the inner boundary of the cortex, the endodermis, is impervious to water because of a band of lignified matrix called the casparian strip. Therefore, to enter the stele, apoplastic water must enter the symplasm of the endodermal cells. From here it can pass by plasmodesmata into the cells of the stele. Once inside the stele, water is again free to move between cells as well as through them. In young roots, water enters directly into the xylem vessels and/or tracheids. These are nonliving conduits so are part of the apoplast.
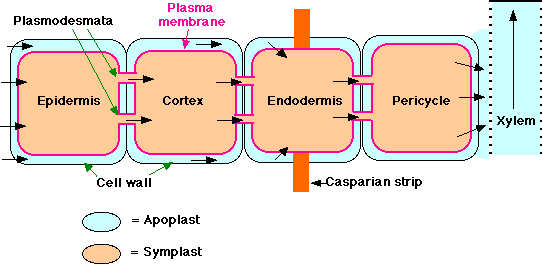
Once in the xylem, water with the minerals that have been deposited in it (as well as occasional organic molecules supplied by the root tissue) move up in the vessels and tracheids. At any level, the water can leave the xylem and pass laterally to supply the needs of other tissues. At the leaves, the xylem passes into the petiole and then into the veins of the leaf. Water leaves the finest veins and enters the cells of the spongy and palisade layers. Here some of the water may be used in metabolism, but most is lost in transpiration.
Minerals enter the root by active transport into the symplast of epidermal cells and move toward and into the stele through the plasmodesmata connecting the cells. They enter the water in the xylem from the cells of the pericycle (as well as of parenchyma cells surrounding the xylem) through specialized transmembrane channels.
What Forces Water Through the Xylem?
Observations
- The mechanism is based on purely physical forces because the xylem vessels and tracheids are lifeless.
- Roots are not needed. This was demonstrated over a century ago by a German botanist who sawed down a 70-ft (21 meters) oak tree and placed the base of the trunk in a barrel of picric acid solution. The solution was drawn up the trunk, killing nearby tissues as it went.
- However, leaves are needed. When the acid reached the leaves and killed them, the upward movement of water ceased.
- Removing a band of bark from around the trunk - a process called girdling - does not interrupt the upward flow of water. Girdling removes only the phloem, not the xylem, and so the foliage does not wilt. (In due course, however, the roots - and thus the entire plant - will die because the roots cannot receive any of the food manufactured by the leaves.)
Transpiration-Pull
In 1895, the Irish plant physiologists H. H. Dixon and J. Joly proposed that water is pulled up the plant by tension (negative pressure) from above. As we have seen, water is continually being lost from leaves by transpiration. Dixon and Joly believed that the loss of water in the leaves exerts a pull on the water in the xylem ducts and draws more water into the leaf. But even the best vacuum pump can pull water up to a height of only 34 ft (10.4 m) or so. This is because a column of water that high exerts a pressure of ~15 lb/in2 (103 kilopascals, kPa) just counterbalanced by the pressure of the atmosphere. How can water be drawn to the top of a sequoia (the tallest is 370 feet [113 meters] high)? Taking all factors into account, a pull of at least 270 lb/in2 (~1.9 x 103 kPa) is probably needed.
The answer to the dilemma lies the cohesion of water molecules; that is the property of water molecules to cling to each through the hydrogen bonds they form. When ultrapure water is confined to tubes of very small bore, the force of cohesion between water molecules imparts great strength to the column of water. It has been reported that tensions as great as 3000 lb/in2 (21 x 103 kPa) are needed to break the column, about the value needed to break steel wires of the same diameter. In a sense, the cohesion of water molecules gives them the physical properties of solid wires. Because of the critical role of cohesion, the transpiration-pull theory is also called the cohesion theory.
support for Cohesion theory
- If sap in the xylem is under tension, we would expect the column to snap apart if air is introduced into the xylem vessel by puncturing it. This is the case.
- If the water in all the xylem ducts is under tension, there should be a resulting inward pull (because of adhesion) on the walls of the ducts. This inward pull in the band of sapwood in an actively transpiring tree should, in turn, cause a decrease in the diameter of the trunk.
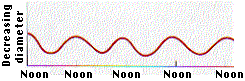
- The graph shows the results of obtained by D. T. MacDougall when he made continuous measurements of the diameter of a Monterey pine. The diameter fluctuated on a daily basis reaching its minimum when the rate of transpiration reached its maximum (around noon)
- The rattan vine may climb as high as 150 ft (45.7 m) on the trees of the tropical rain forest in northeastern Australia to get its foliage into the sun. When the base of a vine is severed while immersed in a basin of water, water continues to be taken up. A vine less than 1 inch (2.5 cm) in diameter will "drink" water indefinitely at a rate of up to 12 ml/minute.
If forced to take water from a sealed container, the vine does so without any decrease in rate, even though the resulting vacuum becomes so great that the remaining water begins to boil spontaneously. (The boiling temperature of water decreases as the air pressure over the water decreases, which is why it takes longer to boil an egg in Denver than in New Orleans.)
- Transpiration-pull enables some trees and shrubs to live in seawater. Seawater is markedly hypertonic to the cytoplasm in the roots of the red mangrove (Rhizophora mangle), and we might expect water to leave the cells resulting in a loss in turgor and wilting. In fact, the remarkably high tensions on the order of 500–800 lb/in2 (~3 to 5 thousand kPa) in the xylem can pull water into the plant against this osmotic gradient. So mangroves literally desalt seawater to meet their needs.
Problems with the theory
When water is placed under a high vacuum, any dissolved gases come out of solution as bubbles (as we saw above with the rattan vine) - this is called cavitation. Any impurities in the water enhance the process. So measurements showing the high tensile strength of water in capillaries require water of high purity - not the case for sap in the xylem. So might cavitation break the column of water in the xylem and thus interrupt its flow? Probably not so long as the tension does not greatly exceed 270 lb/in2 (~1.9 x 103 kPa).
By spinning branches in a centrifuge, it has been shown that water in the xylem avoids cavitation at negative pressures exceeding 225 lb/in2 (~1.6 x 103 kPa). And the fact that sequoias can successfully lift water 358 ft (109 m) - which would require a tension of 270 lb/in2 (~1.9 x 103 kPa) - indicates that cavitation is avoided even at that value. However, such heights may be approaching the limit for xylem transport. Measurements close to the top of the tallest living sequoia (370 ft [=113 m] high) show that the high tensions needed to get water up there have resulted in smaller stomatal openings, causing lower concentrations of CO2 in the needles, causing reduced photosynthesis, causing reduced growth (smaller cells and much smaller needles). (Reported by Koch, G. W. et al., in Nature, 22 April 2004.) So the limits on water transport limit the ultimate height which trees can reach. The tallest tree ever measured, a Douglas fir, was 413 ft. (125.9 meters) high.
Root Pressure
When a tomato plant is carefully severed close to the base of the stem, sap oozes from the stump. The fluid comes out under pressure which is called root pressure. Root pressure is created by the osmotic pressure of xylem sap which is, in turn, created by dissolved minerals and sugars that have been actively transported into the apoplast of the stele.
One important example is the sugar maple when, in very early spring, it hydrolyzes the starches stored in its roots into sugar. This causes water to pass by osmosis through the endodermis and into the xylem ducts. The continuous inflow forces the sap up the ducts.
Although root pressure plays a role in the transport of water in the xylem in some plants and in some seasons, it does not account for most water transport.
- Few plants develop root pressures greater than 30 lb/in2 (207 kPa), and some develop no root pressure at all.
- The volume of fluid transported by root pressure is not enough to account for the measured movement of water in the xylem of most trees and vines.
- Those plants with a reasonably good flow of sap are apt to have the lowest root pressures and vice versa.
- The highest root pressures occur in the spring when the sap is strongly hypertonic to soil water, but the rate of transpiration is low. In summer, when transpiration is high and water is moving rapidly through the xylem, often no root pressure can be detected.
So although root pressure may play a significant role in water transport in certain species (e.g., the coconut palm) or at certain times, most plants meet their needs by transpiration-pull.


