2.3: Fats
- Page ID
- 3781
Fat molecules are made up of four parts: a molecule of glycerol (on the right) and three molecules of fatty acids. Each fatty acid consists of a hydrocarbon chain with a carboxyl group at one end. The glycerol molecule has three hydroxyl groups, each able to interact with the carboxyl group of a fatty acid. Removal of a water molecule at each of the three positions forms a triglyceride. The three fatty acids in a single fat molecule may be all alike (as shown here for tristearin) or they may be different. They may contain as few as 4 carbon atoms or as many as 24.
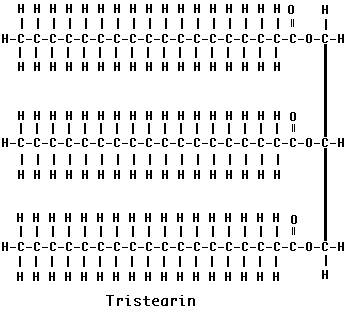
Because fatty acids are synthesized from fragments containing two carbon atoms, the number of carbon atoms in the chain is almost always an even number. In animal fats, 16-carbon (palmitic acid) and 18-carbon (stearic acid - shown here) fatty acids are the most common.
Unsaturated Fats
Some fatty acids have one or more double bonds between their carbon atoms. They are called unsaturated because they could hold more hydrogen atoms than they do. Monounsaturated fats have a single double bond in their fatty acids and polyunsaturated fats, such as trilinolein shown here, have two or more.
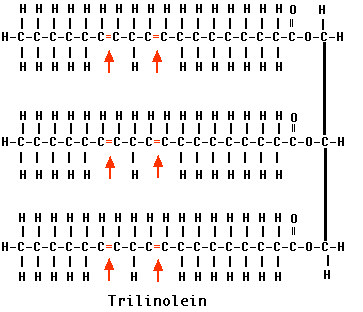
Double bonds are rigid and those in natural fats introduce a kink in the molecule. This prevents the fatty acids from packing close together and as a result, unsaturated fats have a lower melting point than do saturated fats. Because most of them are liquid at room temperature, we call them oils. Corn oil, canola oil, cottonseed oil, peanut oil, and olive oil are common examples. As this list suggests, plant fats tend to be unsaturated (therefore "oils"). Fats from such animals as cattle tend to be saturated.
Trans Fatty Acids
The most abundant (and least expensive) source of fat is from plant oils but many cooking applications, particularly baked products, need solid fats. The food industry uses hydrogenated oils for things like shortening and margarine. In hydrogenation, plant oils are exposed to hydrogen at a high temperature and in the presence of a catalyst.
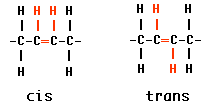
Two things result:(1) some double bonds are converted into single bonds and (2) other double bonds are converted from cis to trans configuration. Both these effects straighten out the molecules so they can lie closer together and become solid rather than liquid.
Omega fatty acids
One system for naming unsaturated fatty acids is to indicate the position of the first double bond counting from the opposite end from the carboxyl group. That terminal carbon atom (shown here in blue) is called the omega carbon atom. Thus a monounsaturated fatty acid with its single double bond after carbon #3 (counting from and including the omega carbon) is called an omega-3 fatty acid. But so is a polyunsaturated fatty acid, such as linolenic acid (shown here), if its first double bond is in that position.
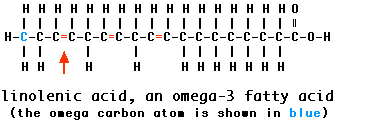
Some studies have suggested that omega-3 fatty acids help protect against cardiovascular disease. For this reason, a Dietary Reference Intake (DRI) of 1.1 grams/day for women (1.6 for men) was established in September 2002.


