3.5: Antibodies in Quantitation and In Vivo Detection
- Page ID
- 64709
\( \newcommand{\vecs}[1]{\overset { \scriptstyle \rightharpoonup} {\mathbf{#1}} } \)
\( \newcommand{\vecd}[1]{\overset{-\!-\!\rightharpoonup}{\vphantom{a}\smash {#1}}} \)
\( \newcommand{\dsum}{\displaystyle\sum\limits} \)
\( \newcommand{\dint}{\displaystyle\int\limits} \)
\( \newcommand{\dlim}{\displaystyle\lim\limits} \)
\( \newcommand{\id}{\mathrm{id}}\) \( \newcommand{\Span}{\mathrm{span}}\)
( \newcommand{\kernel}{\mathrm{null}\,}\) \( \newcommand{\range}{\mathrm{range}\,}\)
\( \newcommand{\RealPart}{\mathrm{Re}}\) \( \newcommand{\ImaginaryPart}{\mathrm{Im}}\)
\( \newcommand{\Argument}{\mathrm{Arg}}\) \( \newcommand{\norm}[1]{\| #1 \|}\)
\( \newcommand{\inner}[2]{\langle #1, #2 \rangle}\)
\( \newcommand{\Span}{\mathrm{span}}\)
\( \newcommand{\id}{\mathrm{id}}\)
\( \newcommand{\Span}{\mathrm{span}}\)
\( \newcommand{\kernel}{\mathrm{null}\,}\)
\( \newcommand{\range}{\mathrm{range}\,}\)
\( \newcommand{\RealPart}{\mathrm{Re}}\)
\( \newcommand{\ImaginaryPart}{\mathrm{Im}}\)
\( \newcommand{\Argument}{\mathrm{Arg}}\)
\( \newcommand{\norm}[1]{\| #1 \|}\)
\( \newcommand{\inner}[2]{\langle #1, #2 \rangle}\)
\( \newcommand{\Span}{\mathrm{span}}\) \( \newcommand{\AA}{\unicode[.8,0]{x212B}}\)
\( \newcommand{\vectorA}[1]{\vec{#1}} % arrow\)
\( \newcommand{\vectorAt}[1]{\vec{\text{#1}}} % arrow\)
\( \newcommand{\vectorB}[1]{\overset { \scriptstyle \rightharpoonup} {\mathbf{#1}} } \)
\( \newcommand{\vectorC}[1]{\textbf{#1}} \)
\( \newcommand{\vectorD}[1]{\overrightarrow{#1}} \)
\( \newcommand{\vectorDt}[1]{\overrightarrow{\text{#1}}} \)
\( \newcommand{\vectE}[1]{\overset{-\!-\!\rightharpoonup}{\vphantom{a}\smash{\mathbf {#1}}}} \)
\( \newcommand{\vecs}[1]{\overset { \scriptstyle \rightharpoonup} {\mathbf{#1}} } \)
\(\newcommand{\longvect}{\overrightarrow}\)
\( \newcommand{\vecd}[1]{\overset{-\!-\!\rightharpoonup}{\vphantom{a}\smash {#1}}} \)
\(\newcommand{\avec}{\mathbf a}\) \(\newcommand{\bvec}{\mathbf b}\) \(\newcommand{\cvec}{\mathbf c}\) \(\newcommand{\dvec}{\mathbf d}\) \(\newcommand{\dtil}{\widetilde{\mathbf d}}\) \(\newcommand{\evec}{\mathbf e}\) \(\newcommand{\fvec}{\mathbf f}\) \(\newcommand{\nvec}{\mathbf n}\) \(\newcommand{\pvec}{\mathbf p}\) \(\newcommand{\qvec}{\mathbf q}\) \(\newcommand{\svec}{\mathbf s}\) \(\newcommand{\tvec}{\mathbf t}\) \(\newcommand{\uvec}{\mathbf u}\) \(\newcommand{\vvec}{\mathbf v}\) \(\newcommand{\wvec}{\mathbf w}\) \(\newcommand{\xvec}{\mathbf x}\) \(\newcommand{\yvec}{\mathbf y}\) \(\newcommand{\zvec}{\mathbf z}\) \(\newcommand{\rvec}{\mathbf r}\) \(\newcommand{\mvec}{\mathbf m}\) \(\newcommand{\zerovec}{\mathbf 0}\) \(\newcommand{\onevec}{\mathbf 1}\) \(\newcommand{\real}{\mathbb R}\) \(\newcommand{\twovec}[2]{\left[\begin{array}{r}#1 \\ #2 \end{array}\right]}\) \(\newcommand{\ctwovec}[2]{\left[\begin{array}{c}#1 \\ #2 \end{array}\right]}\) \(\newcommand{\threevec}[3]{\left[\begin{array}{r}#1 \\ #2 \\ #3 \end{array}\right]}\) \(\newcommand{\cthreevec}[3]{\left[\begin{array}{c}#1 \\ #2 \\ #3 \end{array}\right]}\) \(\newcommand{\fourvec}[4]{\left[\begin{array}{r}#1 \\ #2 \\ #3 \\ #4 \end{array}\right]}\) \(\newcommand{\cfourvec}[4]{\left[\begin{array}{c}#1 \\ #2 \\ #3 \\ #4 \end{array}\right]}\) \(\newcommand{\fivevec}[5]{\left[\begin{array}{r}#1 \\ #2 \\ #3 \\ #4 \\ #5 \\ \end{array}\right]}\) \(\newcommand{\cfivevec}[5]{\left[\begin{array}{c}#1 \\ #2 \\ #3 \\ #4 \\ #5 \\ \end{array}\right]}\) \(\newcommand{\mattwo}[4]{\left[\begin{array}{rr}#1 \amp #2 \\ #3 \amp #4 \\ \end{array}\right]}\) \(\newcommand{\laspan}[1]{\text{Span}\{#1\}}\) \(\newcommand{\bcal}{\cal B}\) \(\newcommand{\ccal}{\cal C}\) \(\newcommand{\scal}{\cal S}\) \(\newcommand{\wcal}{\cal W}\) \(\newcommand{\ecal}{\cal E}\) \(\newcommand{\coords}[2]{\left\{#1\right\}_{#2}}\) \(\newcommand{\gray}[1]{\color{gray}{#1}}\) \(\newcommand{\lgray}[1]{\color{lightgray}{#1}}\) \(\newcommand{\rank}{\operatorname{rank}}\) \(\newcommand{\row}{\text{Row}}\) \(\newcommand{\col}{\text{Col}}\) \(\renewcommand{\row}{\text{Row}}\) \(\newcommand{\nul}{\text{Nul}}\) \(\newcommand{\var}{\text{Var}}\) \(\newcommand{\corr}{\text{corr}}\) \(\newcommand{\len}[1]{\left|#1\right|}\) \(\newcommand{\bbar}{\overline{\bvec}}\) \(\newcommand{\bhat}{\widehat{\bvec}}\) \(\newcommand{\bperp}{\bvec^\perp}\) \(\newcommand{\xhat}{\widehat{\xvec}}\) \(\newcommand{\vhat}{\widehat{\vvec}}\) \(\newcommand{\uhat}{\widehat{\uvec}}\) \(\newcommand{\what}{\widehat{\wvec}}\) \(\newcommand{\Sighat}{\widehat{\Sigma}}\) \(\newcommand{\lt}{<}\) \(\newcommand{\gt}{>}\) \(\newcommand{\amp}{&}\) \(\definecolor{fillinmathshade}{gray}{0.9}\)Written by ?? Needs work
-
Differentiate Between Immunoassay Formats:
- Compare and contrast the principles and practical applications of ELISA, immunoassay microarrays, and immunohistochemistry.
- Identify the advantages and limitations of each method in terms of sensitivity, throughput, specificity, and spatial resolution.
-
Understand ELISA Methodologies:
- Describe the different ELISA formats (e.g., direct, indirect, sandwich, and competitive) and explain how antibody-antigen interactions are used to detect and quantify analytes.
- Explain how the use of labeled secondary antibodies can enhance assay sensitivity and specificity.
- Interpret how ELISA data can be used to determine the concentration of a target analyte.
-
Explore Multiplexing with Immunoassay Microarrays:
- Explain how miniaturization and robotic deposition techniques enable simultaneous detection of multiple analytes.
- Describe the design and function of peptide and protein microarrays in mapping epitopes and studying protein interactions.
- Evaluate the benefits of high-throughput immunoassays for large-scale proteomic studies, such as those conducted in the Human Protein Atlas.
-
Master Immunohistochemistry (IHC) Techniques:
- Outline the workflow of IHC, including tissue fixation, embedding, sectioning, antigen retrieval, and antibody staining.
- Discuss the role of primary and secondary antibodies in IHC and how reporter systems (enzymatic or fluorescent) generate visual signals.
- Analyze how IHC provides spatial information about protein expression and localization in complex tissues and its application in clinical diagnostics.
-
Evaluate Factors Affecting Antibody Binding:
- Assess the importance of antibody specificity, affinity, and optimization (titration, selection of monoclonal vs. polyclonal antibodies) in ensuring reliable results.
- Examine how sample preparation (e.g., tissue type, fixation method, antigen retrieval) can impact the sensitivity and background of immunoassays.
-
Integrate Immunoassay Data in Research and Diagnostics:
- Explain how immunoassays are used in mapping the human proteome, biomarker discovery, and patient stratification in cancer and other diseases.
- Discuss how quantitative and qualitative immunoassay data can guide both basic research and clinical decision-making.
These learning goals are intended to enable students to not only understand the technical principles behind immunoassays but also to critically assess their applications and limitations in modern biochemistry and clinical practice.
We will describe three different uses of antibodies for detecting and quantifying analytes.
Enzyme-Linked Immunosorbent Assay (ELISA)
(work derived from the Human Atlas Project)
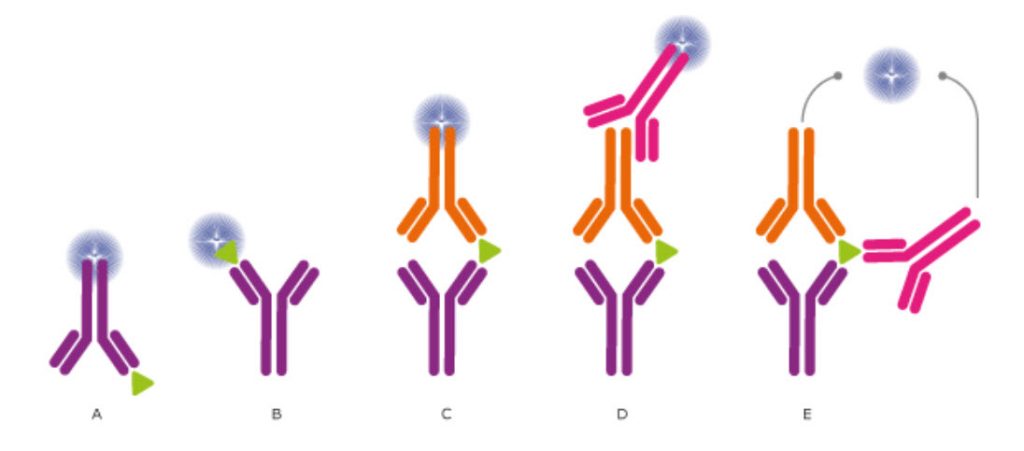
Since the first use of antibodies to detect antigens, many technologies have been developed that exploit antibodies' ability to bind other molecules. During the 1950s, Yalow and Berson developed a method using radioactivity to determine the amount of an analyte in a solution. This 'radioimmunoassay' (RIA), for which Yalow received the Nobel prize in 1977, was a very sensitive method for detecting hormones. However, using radioactivity for antigen detection is not safe and suitable for general use. Hence, an alternative procedure was developed by linking enzymes to antibodies rather than a radioactive molecule and by attaching molecules to surfaces. This is the basis of the widely used "enzyme-linked immunosorbent assay" (ELISA). Many variants of experimental procedures have been developed, and it is common to build assays using multiple antibodies to detect a target of interest (see Figure 3.5.1). To further enhance the capabilities of the immunoassay format, microarray-based applications have been developed that enable the measurement of multiple analytes in a single reaction chamber (see below). Different detection methods are described in Figure \(\PageIndex{1}\).
Figure \(\PageIndex{1}\): Different detection methods for ELISA and Other Immunoassays. (CC BY-SA 3.0; The Human Atlas Project)
In ELISA assays, the antibodies may (A) detect an immobilized antigen, (B) capture a labeled antigen, (C) capture an unlabeled antigen and use a second labeled antibody to detect the captured antigen, (D) use a third antibody for detection, or even use two antibodies for detection (E). Direct labeling of the antibody or antigen as in (A), (B), and (C) is the simplest and fastest method for detection. Using a secondary antibody for detection, as shown in (D) and (E), will further increase the sensitivity and selectivity of the analysis. The technique (D) also allows greater flexibility, whereas method (E) further increases the specificity, as three antibodies must bind the antigen to produce a reporter molecule. Out of the presented assays, the most commonly used concepts are shown in (C) and (D). We will describe how ELISA data are used to determine analyte concentrations in Chapter 5.
A new era in immunoassays started with the development of microarrays. Microarrays most commonly use small-volume droplets that have dried on a small surface area. The reaction dimensions are miniaturized so that many assays can be performed in multiple samples in parallel. Glass slides can be used, and robotic pipettors can deposit very small drops of liquid (1 nL = 10-9 L) on the glass surface in an ordered fashion, with the spots having sizes of around 0.15 mm. Another common multiplexing technique uses smaller, color-coded particles (0.005 mm in diameter). These particles can be coated with antibodies to fish out the analyte from the solution.
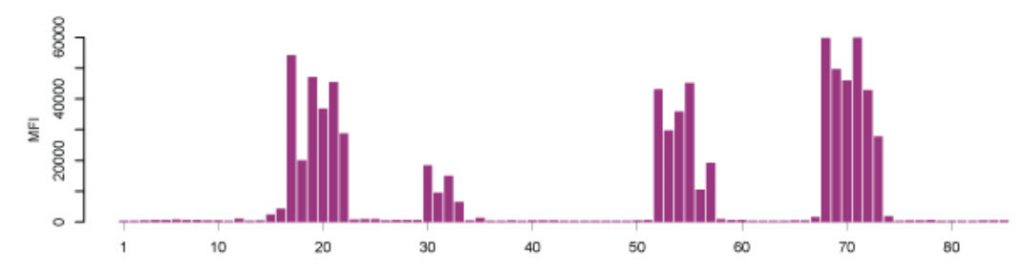
Microarray assays are used for parallel analysis of DNA and RNA molecules. In addition, multiplexed techniques detect multiple proteins simultaneously and study post-translational modifications, such as phosphorylation. Another example is the analysis of antibodies circulating in patients' blood. Protein microarrays can reveal ligand interactions with the entire protein or with larger protein fragments. In contrast, peptide microarrays detect small peptides (epitopes) of the proteins that bind antibodies. A typical epitope mapping result is shown in Figure \(\PageIndex{2}\) (Edfors et al., 2014).
Figure \(\PageIndex{2}\): Epitope Mapping of Polyclonal Antibodies. Polyclonal antibodies bind to a peptide array, where the result displays four distinct linear epitopes and the consecutive overlapping peptides that are bound. X-axis: peptides, Y-axis: mean fluorescence intensity (MFI). (Edfors et al., 2014) Image from The Human Atlas Project
Synthesizing millions of overlapping peptides with only a single amino acid shift on such arrays enables mapping antibody-binding regions at high resolution. This gives a detailed analysis of the linear (continuous) epitopes recognized by an antibody.
Immunohistochemistry - Detecting Proteins in Vivo
(Work derived from the Human Atlas Project)
Immunohistochemistry (IHC) is a powerful microscopy-based technique for visualizing cellular components in tissue samples, such as proteins or other macromolecules. Its strength lies in its intuitive visual output, which reveals the target protein's presence and localization across different cell types, biological states, and subcellular compartments within complex tissues.
The IHC technique was invented during the 1940s (Coons, Creech, & Jones, 1941) and is routinely used as an important tool in health care and pathology, e.g., for diagnostic purposes or to stratify patients for optimized treatment regimes. IHC is also widely used in research, where molecules of interest are analyzed to study their roles in healthy and diseased cells and tissues at the molecular, cellular, or tissue levels. There are various methods to visualize targets in tissues using IHC or IHC-based methods, and numerous protocols exist for different applications and assays. Even though IHC is generally robust and well-established, new assays often require careful optimization depending on the tissue or the properties of the target protein, binder molecule, and/or reporter system.
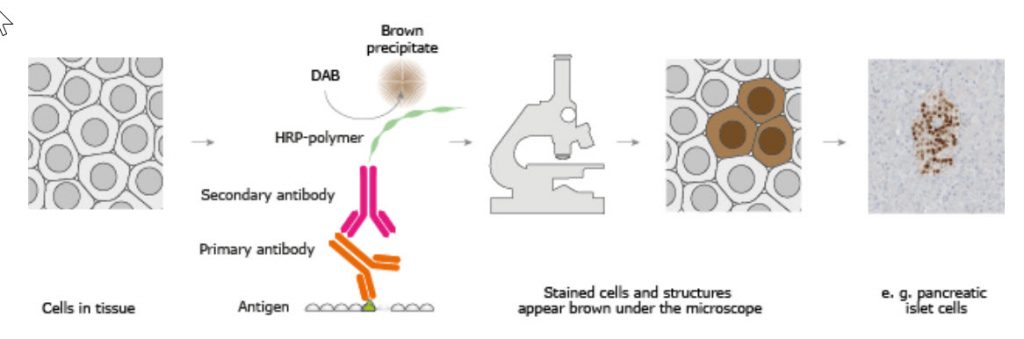
The classical IHC assay is illustrated in Figure \(\PageIndex{3}\) and involves detecting epitopes expressed by a single protein target in a tissue sample using a "primary antibody" that binds those epitopes with high specificity. After the epitope-antibody binding event, a "secondary antibody" that binds the primary antibody with high specificity is added. The secondary antibody is coupled to a reporter molecule. After the antibody-antibody binding event, a chemical substrate is added, which reacts with the reporter molecule to produce a colored precipitate at the site of the whole epitope-antibody complex.
Figure \(\PageIndex{3}\): The Basic Principle of Immunohistochemistry. Image from The Human Atlas Project
In the schematic illustration, a formalin-fixed paraffin-embedded tissue section is stained using a primary antibody directed toward a specific protein target. A solution containing the primary antibody is added to the tissue section, and the antibodies are allowed to bind to their targets. After this step, unbound and surplus antibodies are washed away, and the secondary antibody is added. The secondary antibody, which carries a linker molecule conjugated to horseradish peroxidase (HRP), is allowed to bind to the primary antibody, followed by another washing step. After this, 3,3' Diaminobenzidine (DAB) is added. The HRP enzyme converts the DAB substrate into a brown precipitate deposited in the tissue at the reaction site, providing a visual representation of where the primary antibody first binds its target.
Tissue preparation
The tissue plays a central role in the experiment and must be processed to preserve epitopes and proper morphology. The most common processing for IHC is the preparation of formalin-fixed, paraffin-embedded (FFPE) tissue blocks. Formalin fixation aims to cross-link proteins within the tissue chemically. This terminates all cellular processes, freezes the cellular components in place and conformation at the time of fixation, and prevents degradation. After adequate fixation, the tissue is further processed and ultimately embedded in paraffin blocks, which are then sectioned into thin slices (usually 4-10µm) using a microtome. The sections are transferred to glass slides and allowed to adhere before further processing.
Other fixation methods besides formalin are sometimes used. These include other types of aldehydes or using different alcohol solutions. The best choice of fixative depends heavily on the assay. A common alternative to FFPE is to prepare frozen tissue samples. In this case, the tissue is embedded in a cryoprotective medium, frozen, and fixed post-sectioning. Frozen tissues are sectioned in cryostats and have the advantage of short processing times and better preservation of sensitive epitopes, but can often be inferior to FFPE tissues in terms of preserving histological morphology.
Antigen (epitope) retrieval
A concern associated with cross-linking fixatives like formalin, or with the length of time spent in the fixative medium, is the masking of epitopes, which can prevent the primary antibody from binding to its target. Especially with FFPE samples, there is often a need to revert some of the chemical crosslinking and "retrieve" the epitopes before proceeding to the actual IHC. Several antigen retrieval protocols are available, and the main strategies include heating the tissue slide, using digestive enzymes, using detergents, or combining these approaches. The most common method for antigen retrieval in FFPE samples is to pressure-boil the tissue slides in an acidic citrate buffer for around 15-20 minutes.
Antibody binding
The quality and specificity of the binding molecule are crucial for any IHC-based technique, and the choice of binder can directly affect the outcome, reliability, and possibly the assay's interpretation. Antibodies are by far the most common type of binding molecule used for IHC. Although most antibodies can detect the intended molecule of interest, they may vary widely in their specificity for that target. Antibodies with high specificity are therefore more reliable when interpreting "on-target" binding since they produce little or no "off-target" binding or "background". Less specific antibodies can exhibit greater off-target binding, and the resulting background may interfere with the correct interpretation of on-target signals. There are two main types of antibodies: polyclonal antibodies, which are heterogeneous mixtures that bind different epitopes on the target, and monoclonal antibodies, which bind the same epitope. Polyclonal antibodies are often highly potent because they can detect and bind multiple epitopes on the same target. However, the epitopes they bind are often poorly defined, and with multiple, varying epitope-specificities comes an increased likelihood of off-target binding events and background noise. However, the potency of polyclonal antibodies can be advantageous since the concentration of binding events around the on-target molecule usually outweighs potential background noise. A drawback is that polyclonal antibodies are usually limited resources since they are derived from animal sera. Monoclonal antibodies, by contrast, exhibit greater consistency because they can be produced in hybridoma cell lines. Monoclonal antibodies are also often well-defined in epitope binding. However, they can still yield results that are hard to interpret when specificity is low or the target epitope is in low abundance.
Careful optimization and titration of antibody concentration for each assay are needed, as the results depend not only on the antibody's specificity and affinity for the target but also on the concentration and availability of on-target and potential off-target epitopes in the sample. Adding too many antibodies to the sample increases the likelihood of low-affinity off-target binding events once the on-target epitope(s) are saturated with binders. By lowering the antibody concentration, off-target binding events become rarer as they usually have lower affinity than on-target binding events. The risk when attempting to reduce background with a low-affinity antibody is that on-target signals are weakened, leading to a false negative result.
Other types of binder molecules sometimes used in IHC-based techniques include affibodies, peptides, antibody fragments, or other small molecules.
Detection systems
The whole purpose of performing IHC is to obtain a visual representation of where the target is located within the experimental tissue and to gain information about the target's expression pattern across heterogeneous cell populations and/or subcellular sites. This is exemplified in Figure \(\PageIndex{4}\), which illustrates how different antibodies visualize different cellular or tissue compartments within a complex tissue. Visualizing the target-antibody interaction requires a detection system that produces an observable stain or signal. The most common method for introducing a detection system into the experiment is to use a secondary antibody that carries a prebound reporter molecule, e.g., an enzyme or a fluorophore. Secondary antibodies are usually specifically targeted to antibodies from different species. For example, if the primary antibody is raised in a rabbit, the secondary antibody must be raised in another animal and explicitly targeted towards rabbit antibodies.
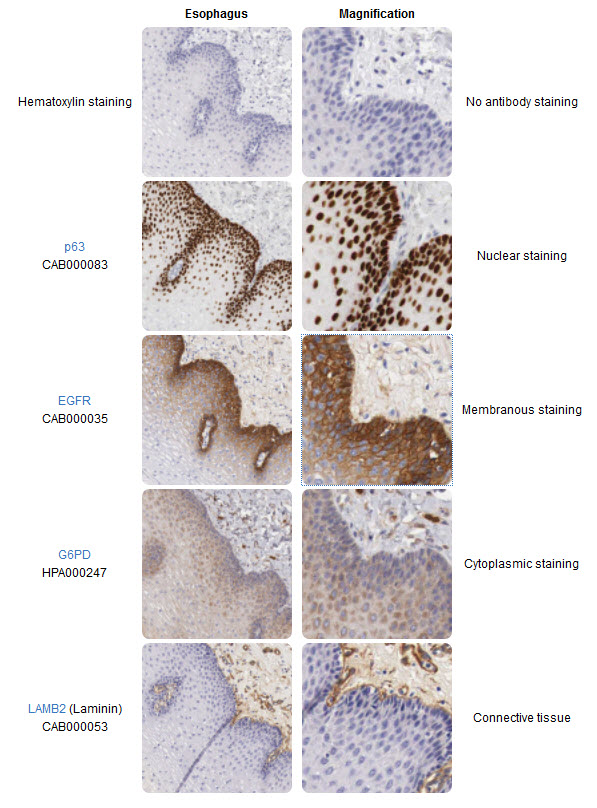
The right column magnifies the corresponding images in the left column. In the IHC image, consecutive sections of a human esophagus stained with four antibodies allow direct comparison of protein expression patterns within the tissue and subcellular compartments. The top images are only counterstained for comparison with hematoxylin. The p63 antibody stains cell nuclei in a population of cells residing in the esophageal epithelium's basal part. The EGFR (Epidermal growth factor receptor) antibody appears to stain the same cell population as p63, but it stains cellular membranes rather than nuclei. The G6PD (Glucose-6-phosphate dehydrogenase) antibody stains the cytoplasm of a broader range of esophageal epithelial cells and connective tissue cells. The Laminin (LAMB2) antibody stains only cells and structures in the connective tissue underlying the esophagus.
Image from The Human Atlas Project
For FFPE tissue samples, the most common detection method is an enzymatic reaction that generates a colored precipitate at the antibody-binding site. Secondary antibodies with an attached enzyme, e.g., horseradish peroxidase (HRP) or alkaline phosphatase (AP), are capable of converting chromogens like 3,3' Diaminobenzidine (DAB) or 5-bromo-4-chloro-3-indolyl phosphate/ p-nitroblue tetrazolium chloride (BCIP/NBT) into brown or bluish precipitates that are deposited in the tissue at the site of the reaction. Chromogenic stains are observable using light microscopy and are usually very stable over long periods, which is beneficial if the experiment needs to be archived or reviewed later.
For frozen tissue sections, it is more common to use fluorophore-conjugated secondary antibodies that emit a specific color (usually green, red, or blue) upon excitation at the appropriate wavelengths of light. Moreover, fluorophores are usually unstable over long periods. However, the benefit of using fluorophores is that they provide an easy method for performing double-labeling experiments, in which several antibodies targeting multiple targets are assayed in the same sample. The secondary antibodies must be targeted towards different primary antibodies and are coupled to different fluorophores. The different secondary antibodies are then observed separately by exciting them sequentially with different wavelengths of light. These different excitation results are saved as separate images (or color channels) and may later be overlaid to infer protein co-localizations.
Using reporter-carrying secondary antibodies for detection is an amplification step since several secondary antibodies can bind to a single primary antibody. Still, additional amplification steps may be desired to increase the signal and sensitivity of the experiment. In such cases, the secondary antibody may instead carry "linker molecules," such as biotin polymers, which can recruit more reporter molecules in subsequent steps. This strategy for signal amplification is applicable to both enzymatic and fluorescent detection methods.
Counterstaining
Immunohistochemical staining with chromogens offers the benefit of a counterstain that enhances contrast and facilitates the observation of histological features. The most common counterstain for FFPE samples is hematoxylin, which stains cellular cytoplasm with a pale bluish color and cell nuclei in a darker bluish hue. Fluorescent stainings are usually not counterstained with hematoxylin since the detection method is not based on light microscopy. Instead, the most common way to counterstain fluorescence is to label cell nuclei with fluorescent dyes that bind nucleic acids. After the immunohistochemical reaction, the remaining steps are to use a coverslip to seal and protect the sample, and for long-term storage. The most common way is to "glue" the coverslip to the sample using commercially available purpose-made resins. Figure \(\PageIndex{5}\) below shows endothelial cells stained with various fluorescent dyes.


Specific examples
IHC is widely used in both research and clinical practice. The Human Protein Atlas (HPA) project is a prime example of how high-throughput IHC is used to map the human proteome at a large scale across many tissues, cancers, and cell types. In the HPA project, a streamlined in-house large-scale antibody production chain facilitates the generation of specific antibodies, which are used to systematically stain tissue microarrays containing hundreds of tissue cores within a single experiment after passing basic characterization and validation regimes. The IHC system employed by HPA relies heavily on standardized protocols and automated processes. Still, the optimal titration for each antibody is evaluated manually before the antibody is approved for staining across the full set of tissues. Each stained tissue core is annotated with respect to immunohistochemical staining in tissues and cell types, and is thereafter published as a high-resolution image on the web portal for free viewing by anyone.
In clinical practice, IHC is primarily used in pathology to aid physicians in evaluating tissue specimens for healthy and/or diseased states, establishing diagnoses, and defining molecular subtypes of various cancers. A specific example of when IHC is used diagnostically is when pathologists are presented with a metastatic tumor sample, and the primary tumor origin is unknown. In these cases, pathologists use a panel of antibodies targeting tissue-specific proteins, such as prostate-specific antigen for prostate cancer, estrogen receptor for gynecological cancers, or cytokeratin 20 for gastrointestinal cancers (Gremel et al., 2014). Once a broad classification is made, additional tissue-specific antibodies are used to further pinpoint the primary tumor's origin. This information helps choose the best or most appropriate strategy for drug therapy/or locating the primary tumor for radiation therapy and/or surgery.
Summary
This chapter explores the diverse applications of antibodies in detecting and quantifying biological analytes, emphasizing three main methodologies: Enzyme-Linked Immunosorbent Assay (ELISA), immunoassay microarrays, and immunohistochemistry (IHC). It begins by outlining the historical development of immunoassays—from early radioimmunoassays to the safer, enzyme-linked formats—and explains how antibodies’ high specificity and binding affinity are harnessed for analytical purposes.
At the forefront is ELISA, where different assay formats (direct, indirect, sandwich, and competitive) are described, each utilizing either directly labeled antibodies or multiple antibody layers to amplify signal and enhance sensitivity. The chapter then transitions into the realm of multiplexed microarrays, which allow for simultaneous detection of numerous analytes in a miniaturized format. These platforms can perform high-throughput epitope mapping and proteomic analyses by spotting nanoliter droplets on glass slides or by using color-coded particles.
The discussion further extends to immunohistochemistry, a technique that localizes protein expression within the complex architecture of tissues. Detailed attention is given to tissue processing (e.g., formalin fixation, paraffin embedding, and antigen retrieval), the critical roles of primary and secondary antibodies, and the various detection systems—ranging from chromogenic reactions (using enzymes such as HRP to generate colored precipitates) to fluorescence-based methods for multiplex detection.
Throughout the chapter, examples drawn from large-scale projects such as the Human Protein Atlas and clinical diagnostic applications illustrate how these techniques contribute to our understanding of protein expression patterns, tissue-specific localization, and disease pathology. Overall, this chapter emphasizes the integration of antibody-based assays as indispensable tools for both basic research and clinical diagnostics in modern biochemistry.
References
Molnar, C. and Gair, J. (2013) Antibodies. Chapter in Concepts in Biology, Published by B.C. Open Textbook Project. Available at: https://opentextbc.ca/biology/chapter/23-3-antibodies/
The Human Atlas Project. (2019) Methods. Available at: https://www.proteinatlas.org/learn/method
Uhlén M et al, 2015. Tissue-based map of the human proteome. Science
PubMed: 25613900 DOI: 10.1126/science.1260419
Thul PJ et al, 2017. A subcellular map of the human proteome. Science.
PubMed: 28495876 DOI: 10.1126/science.aal3321
Uhlen M et al, 2017. A pathology atlas of the human cancer transcriptome. Science.
PubMed: 28818916 DOI: 10.1126/science.aan2507
Ahern, K. and Rajagopal, I. (2019) Biochemistry Free and Easy. Published by Libretexts. Available at: https://bio.libretexts.org/Bookshelves/Biochemistry/Book%3A_Biochemistry_Free_and_Easy_(Ahern_and_Rajagopal)/09%3A_Techniques/9.04%3A_Gel_Exclusion_Chromatography.
Magdeldin, S. (2012) Gel Electrophoresis - Principles and Basics. Published by InTech under Creative Commons Attribution 3.0. Available at: https://pdfs.semanticscholar.org/4b93/70ac3946cec6e12c369679c4178a5ef38e61.pdf
Structural Biochemistry/Proteins/X-ray Crystallography. (2018, November 19). Wikibooks, The Free Textbook Project. Retrieved 15:40, August 17, 2019 from en.wikibooks.org/w/index.php?title=Structural_Biochemistry/Proteins/X-ray_Crystallography&oldid=3488057.
UCD: Biophysics 200A (2019) "NMR Spectroscopy vs X-ray Crystallography", Chapter published in Current Techniques in Biophysics. Published by Libretexts and available at: https://phys.libretexts.org/Courses/University_of_California_Davis/UCD%3A_Biophysics_200A_-_Current_Techniques_in_Biophysics/NMR_Spectroscopy_vs._X-ray_Crystallography
Wikipedia contributors. (2019, June 27). Protein purification. In Wikipedia, The Free Encyclopedia. Retrieved 23:32, July 28, 2019, from en.Wikipedia.org/w/index.php?title=Protein_purification&oldid=903657925
Wikipedia contributors. (2019, February 15). Fast protein liquid chromatography. In Wikipedia, The Free Encyclopedia. Retrieved 17:14, August 15, 2019, from en.Wikipedia.org/w/index.php?title=Fast_protein_liquid_chromatography&oldid=883530035
Wikipedia contributors. (2019, July 9). Protein mass spectrometry. In Wikipedia, The Free Encyclopedia. Retrieved 15:27, August 16, 2019, from en.Wikipedia.org/w/index.php?title=Protein_mass_spectrometry&oldid=905547289
Wikipedia contributors. (2019, July 8). Peptide synthesis. In Wikipedia, The Free Encyclopedia. Retrieved 06:13, August 17, 2019, from en.Wikipedia.org/w/index.php?title=Peptide_synthesis&oldid=905401648