18.9: The Origin of Life
- Page ID
- 5932
\( \newcommand{\vecs}[1]{\overset { \scriptstyle \rightharpoonup} {\mathbf{#1}} } \)
\( \newcommand{\vecd}[1]{\overset{-\!-\!\rightharpoonup}{\vphantom{a}\smash {#1}}} \)
\( \newcommand{\dsum}{\displaystyle\sum\limits} \)
\( \newcommand{\dint}{\displaystyle\int\limits} \)
\( \newcommand{\dlim}{\displaystyle\lim\limits} \)
\( \newcommand{\id}{\mathrm{id}}\) \( \newcommand{\Span}{\mathrm{span}}\)
( \newcommand{\kernel}{\mathrm{null}\,}\) \( \newcommand{\range}{\mathrm{range}\,}\)
\( \newcommand{\RealPart}{\mathrm{Re}}\) \( \newcommand{\ImaginaryPart}{\mathrm{Im}}\)
\( \newcommand{\Argument}{\mathrm{Arg}}\) \( \newcommand{\norm}[1]{\| #1 \|}\)
\( \newcommand{\inner}[2]{\langle #1, #2 \rangle}\)
\( \newcommand{\Span}{\mathrm{span}}\)
\( \newcommand{\id}{\mathrm{id}}\)
\( \newcommand{\Span}{\mathrm{span}}\)
\( \newcommand{\kernel}{\mathrm{null}\,}\)
\( \newcommand{\range}{\mathrm{range}\,}\)
\( \newcommand{\RealPart}{\mathrm{Re}}\)
\( \newcommand{\ImaginaryPart}{\mathrm{Im}}\)
\( \newcommand{\Argument}{\mathrm{Arg}}\)
\( \newcommand{\norm}[1]{\| #1 \|}\)
\( \newcommand{\inner}[2]{\langle #1, #2 \rangle}\)
\( \newcommand{\Span}{\mathrm{span}}\) \( \newcommand{\AA}{\unicode[.8,0]{x212B}}\)
\( \newcommand{\vectorA}[1]{\vec{#1}} % arrow\)
\( \newcommand{\vectorAt}[1]{\vec{\text{#1}}} % arrow\)
\( \newcommand{\vectorB}[1]{\overset { \scriptstyle \rightharpoonup} {\mathbf{#1}} } \)
\( \newcommand{\vectorC}[1]{\textbf{#1}} \)
\( \newcommand{\vectorD}[1]{\overrightarrow{#1}} \)
\( \newcommand{\vectorDt}[1]{\overrightarrow{\text{#1}}} \)
\( \newcommand{\vectE}[1]{\overset{-\!-\!\rightharpoonup}{\vphantom{a}\smash{\mathbf {#1}}}} \)
\( \newcommand{\vecs}[1]{\overset { \scriptstyle \rightharpoonup} {\mathbf{#1}} } \)
\(\newcommand{\longvect}{\overrightarrow}\)
\( \newcommand{\vecd}[1]{\overset{-\!-\!\rightharpoonup}{\vphantom{a}\smash {#1}}} \)
\(\newcommand{\avec}{\mathbf a}\) \(\newcommand{\bvec}{\mathbf b}\) \(\newcommand{\cvec}{\mathbf c}\) \(\newcommand{\dvec}{\mathbf d}\) \(\newcommand{\dtil}{\widetilde{\mathbf d}}\) \(\newcommand{\evec}{\mathbf e}\) \(\newcommand{\fvec}{\mathbf f}\) \(\newcommand{\nvec}{\mathbf n}\) \(\newcommand{\pvec}{\mathbf p}\) \(\newcommand{\qvec}{\mathbf q}\) \(\newcommand{\svec}{\mathbf s}\) \(\newcommand{\tvec}{\mathbf t}\) \(\newcommand{\uvec}{\mathbf u}\) \(\newcommand{\vvec}{\mathbf v}\) \(\newcommand{\wvec}{\mathbf w}\) \(\newcommand{\xvec}{\mathbf x}\) \(\newcommand{\yvec}{\mathbf y}\) \(\newcommand{\zvec}{\mathbf z}\) \(\newcommand{\rvec}{\mathbf r}\) \(\newcommand{\mvec}{\mathbf m}\) \(\newcommand{\zerovec}{\mathbf 0}\) \(\newcommand{\onevec}{\mathbf 1}\) \(\newcommand{\real}{\mathbb R}\) \(\newcommand{\twovec}[2]{\left[\begin{array}{r}#1 \\ #2 \end{array}\right]}\) \(\newcommand{\ctwovec}[2]{\left[\begin{array}{c}#1 \\ #2 \end{array}\right]}\) \(\newcommand{\threevec}[3]{\left[\begin{array}{r}#1 \\ #2 \\ #3 \end{array}\right]}\) \(\newcommand{\cthreevec}[3]{\left[\begin{array}{c}#1 \\ #2 \\ #3 \end{array}\right]}\) \(\newcommand{\fourvec}[4]{\left[\begin{array}{r}#1 \\ #2 \\ #3 \\ #4 \end{array}\right]}\) \(\newcommand{\cfourvec}[4]{\left[\begin{array}{c}#1 \\ #2 \\ #3 \\ #4 \end{array}\right]}\) \(\newcommand{\fivevec}[5]{\left[\begin{array}{r}#1 \\ #2 \\ #3 \\ #4 \\ #5 \\ \end{array}\right]}\) \(\newcommand{\cfivevec}[5]{\left[\begin{array}{c}#1 \\ #2 \\ #3 \\ #4 \\ #5 \\ \end{array}\right]}\) \(\newcommand{\mattwo}[4]{\left[\begin{array}{rr}#1 \amp #2 \\ #3 \amp #4 \\ \end{array}\right]}\) \(\newcommand{\laspan}[1]{\text{Span}\{#1\}}\) \(\newcommand{\bcal}{\cal B}\) \(\newcommand{\ccal}{\cal C}\) \(\newcommand{\scal}{\cal S}\) \(\newcommand{\wcal}{\cal W}\) \(\newcommand{\ecal}{\cal E}\) \(\newcommand{\coords}[2]{\left\{#1\right\}_{#2}}\) \(\newcommand{\gray}[1]{\color{gray}{#1}}\) \(\newcommand{\lgray}[1]{\color{lightgray}{#1}}\) \(\newcommand{\rank}{\operatorname{rank}}\) \(\newcommand{\row}{\text{Row}}\) \(\newcommand{\col}{\text{Col}}\) \(\renewcommand{\row}{\text{Row}}\) \(\newcommand{\nul}{\text{Nul}}\) \(\newcommand{\var}{\text{Var}}\) \(\newcommand{\corr}{\text{corr}}\) \(\newcommand{\len}[1]{\left|#1\right|}\) \(\newcommand{\bbar}{\overline{\bvec}}\) \(\newcommand{\bhat}{\widehat{\bvec}}\) \(\newcommand{\bperp}{\bvec^\perp}\) \(\newcommand{\xhat}{\widehat{\xvec}}\) \(\newcommand{\vhat}{\widehat{\vvec}}\) \(\newcommand{\uhat}{\widehat{\uvec}}\) \(\newcommand{\what}{\widehat{\wvec}}\) \(\newcommand{\Sighat}{\widehat{\Sigma}}\) \(\newcommand{\lt}{<}\) \(\newcommand{\gt}{>}\) \(\newcommand{\amp}{&}\) \(\definecolor{fillinmathshade}{gray}{0.9}\)To account for the origin of life on our earth requires solving several problems:
- How the organic molecules that define life, e.g. amino acids, nucleotides, were created.
- How these were assembled into macromolecules, e.g. proteins and nucleic acids, - a process requiring catalysts.
- How these were able to reproduce themselves.
- How these were assembled into a system delimited from its surroundings (i.e., a cell).
A number of theories address each of these problems. As for the first problem, four scenarios have been proposed. Organic molecules:
- were synthesized from inorganic compounds in the atmosphere,
- rained down on earth from outer space,
- were synthesized at hydrothermal vents on the ocean floor,
- were synthesized when comets or asteroids struck the early earth.
Scenario 1: Miller's Experiment
Stanley Miller, a graduate student in biochemistry, built the apparatus shown in Figure \(\PageIndex{1}\). He filled it with water (H2O), methane (CH4), ammonia (NH3) and hydrogen (H2), but no oxygen. He hypothesized that this mixture resembled the atmosphere of the early earth. The mixture was kept circulating by continuously boiling and then condensing the water. The gases passed through a chamber containing two electrodes with a spark passing between them.
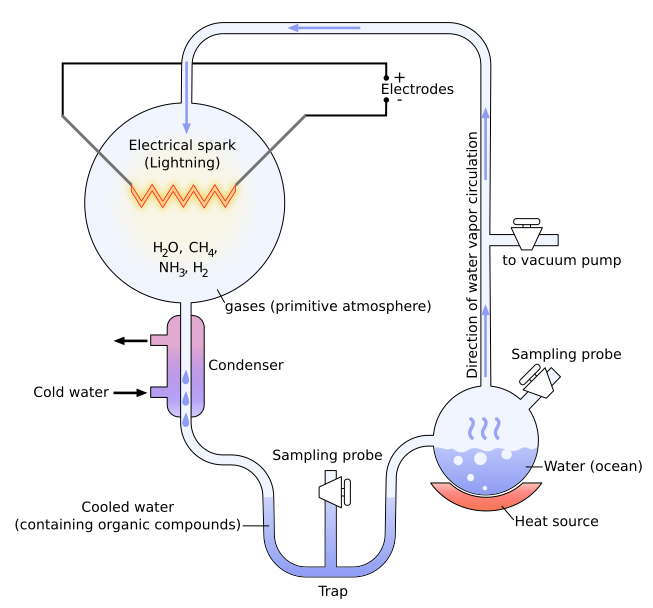
At the end of a week, Miller used paper chromatography to show that the flask now contained several amino acids as well as some other organic molecules. However, it is now thought that the atmosphere of the early earth was not rich in methane and ammonia - essential ingredients in Miller's experiments. In the years since Miller's work, many variants of his procedure have been tried. Virtually all the small molecules that are associated with life have been formed:
- 17 of the 20 amino acids used in protein synthesis, and all the purines and pyrimidines used in nucleic acid synthesis.
- But abiotic synthesis of ribose - and thus of nucleotides - has been much more difficult. However, success in synthesizing pyrimidine ribonucleotides under conditions that might have existed in the early earth was reported in the 14 May 2009 issue of Nature.
- And in 2015, chemists in Cambridge England led by John Sutherland reported that they had been able to synthesize precursors of 12 of the 20 amino acids and two (of the four) ribonucleotides used by life as well as glycerol-1-phosphate, a precursor of lipids. They created all of these molecules using only hydrogen cyanide (HCN) and hydrogen sulfide (H2S) irradiated with ultraviolet light in the presence of mineral catalysts.
Scenario 2: Molecules from Outer Space
Astronomers, using infrared spectroscopy, have identified a variety of organic molecules in interstellar space, including methane (CH4), methanol (CH3OH), formaldehyde (HCHO), cyanoacetylene (HC3N) (which in spark-discharge experiments is a precursor to the pyrimidine cytosine), polycyclic aromatic hydrocarbonsas well as such inorganic building blocks as carbon dioxide (CO2), carbon monoxide (CO), ammonia (NH3), hydrogen sulfide (H2S), and hydrogen cyanide (HCN).
There have been several reports of producing amino acids and other organic molecules in laboratories by taking a mixture of molecules known to be present in interstellar space such as ammonia (NH3), carbon monoxide (CO), methanol (CH3OH) and water (H2O), hydrogen cyanide (HCN) and exposing it to a temperature close to that of space (near absolute zero) and intense ultraviolet (UV) radiation. Whether or not the molecules that formed terrestrial life arrived here from space, there is little doubt that organic matter continuously rains down on the earth (estimated at 30 tons per day).
Alternatively, organic molecules can be transport to Earth via meteorites as demonstrated with the Murchison Meteorite that that fell near Murchison, Australia on 28 September 1969. This meteorite turned out to contain a variety of organic molecules including: purines and pyrimidines, polyols - compounds with hydroxyl groups on a backbone of 3 to 6 carbons such as glycerol and glyceric acid (sugars are polyols) and the amino acids listed in Table \(\PageIndex{1}\). The amino acids and their relative proportions were quite similar to the products formed in Miller's experiments.

Murchison meteorite at the The National Museum of Natural History (Washington). (CC SA-BY 3.0; :Basilicofresco).
| Glycine | Glutamic acid |
| Alanine | Isovaline |
| Valine | Norvaline |
| Proline | N-methylalanine |
| Aspartic acid | N-ethylglycine |
The question is if these molecules identified in the Murchison meteorite were simply terrestrial contaminants that got into the meteorite after it fell to earth? Probably not:
- Some of the samples were collected on the same day it fell and subsequently handled with great care to avoid contamination.
- The polyols contained the isotopes carbon-13 and hydrogen-2 (deuterium) in greater amounts than found here on earth.
- The samples lacked certain amino acids that are found in all earthly proteins.
- Only L amino acids occur in earthly proteins, but the amino acids in the meteorite contain both D and L forms (although L forms were slightly more prevalent).
Scenario 3: Deep-Sea Hydrothermal Vents
Some deep-sea hydrothermal vents discharge copious amounts of hydrogen, hydrogen sulfide, and carbon dioxide at temperatures around 100°C. (These are not "black smokers".) These gases bubble up through chambers rich in iron sulfides (FeS, FeS2). These can catalyze the formation of simple organic molecules like acetate. (And life today depends on enzymes that have Fe and S atoms in their active sites.)
Scenario 4: Laboratory Synthesis of Nucleobases Under Conditions Mimicking the Impact of Asteroids or Comets on the Early Earth
Researchers in the Czech Republic reported in 2014 that they had succeeded in the abiotic synthesis of adenine (A), guanine (G), cytosine (C), and uracil (U) — the four bases found in RNA (an RNA beginning?) and three of the four found in DNA. They achieved this by bombarding a mixture of formamide and clay with powerful laser pulses that mimicked the temperature and pressure expected when a large meteorite strikes the earth. Formamide is a simple substance, CH3NO, thought to have been abundant on the early earth and containing the four elements fundamental to all life.
Assembling Polymers
Another problem is how polymers - the basis of life itself - could be assembled.
- In solution, hydrolysis of a growing polymer would soon limit the size it could reach.
- Abiotic synthesis produces a mixture of L and D enantiomers. Each inhibits the polymerization of the other. (So, for example, the presence of D amino acids inhibits the polymerization of L amino acids (the ones that make up proteins here on earth).
This has led to a theory that early polymers were assembled on solid, mineral surfaces that protected them from degradation, and in the laboratory polypeptides and polynucleotides (RNA molecules) containing about ~50 units have been synthesized on mineral (e.g., clay) surfaces.
An RNA Beginning?
All metabolism depends on enzymes and, until recently, every enzyme has turned out to be a protein. But proteins are synthesized from information encoded in DNA and translated into mRNA. So here is a chicken-and-egg dilemma. The synthesis of DNA and RNA requires proteins. So proteins cannot be made without nucleic acids and nucleic acids cannot be made without proteins. The discovery that certain RNA molecules have enzymatic activity provides a possible solution. These RNA molecules — called ribozymes — incorporate both the features required of life: storage of information and the ability to act as catalysts.
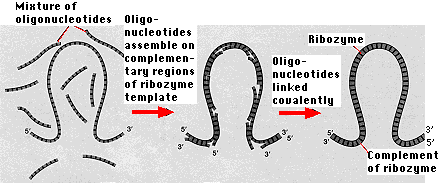
While no ribozyme in nature has yet been found that can replicate itself, ribozymes have been synthesized in the laboratory that can catalyze the assembly of short oligonucleotides into exact complements of themselves. The ribozyme serves as both the template on which short lengths of RNA ("oligonucleotides" are assembled following the rules of base pairing and the catalyst for covalently linking these oligonucleotides.
In principal, the minimal functions of life might have begun with RNA and only later did proteins take over the catalytic machinery of metabolism and DNA take over as the repository of the genetic code. Several other bits of evidence support this notion of an original "RNA world":
- Many of the cofactors that play so many roles in life are based on ribose; for example:
- ATP
- NAD
- FAD
- coenzyme A
- cyclic AMP
- GTP
- In the cell, all deoxyribonucleotides are synthesized from ribonucleotide precursors.
- Many bacteria control the transcription and/or translation of certain genes with RNA molecules, not protein molecules.
Reproduction?
Perhaps the earliest form of reproduction was a simple fission of the growing aggregate into two parts - each with identical metabolic and genetic systems intact.
To function, the machinery of life must be separated from its surroundings - some form of extracellular fluid (ECF). This function is provided by the plasma membrane. Today's plasma membranes are made of a double layer of phospholipids. They are only permeable to small, uncharged molecules like H2O, CO2, and O2. Specialized transmembrane transporters are needed for ions, hydrophilic, and charged organic molecules (e.g., amino acids and nucleotides) to pass into and out of the cell.
However, the same Szostak lab that produced the finding described above reported in the 3 July 2008 issue of Nature that fatty acids, fatty alcohols, and monoglycerides - all molecules that can be synthesized under prebiotic conditions - can also form lipid bilayers and these can spontaneously assemble into enclosed vesicles.
Unlike phospholipid vesicles, these
- admit from the external medium charged molecules like nucleotides
- admit from the external medium hydrophilic molecules like ribose
- grow by self-assembly
- are impermeable to, and thus retain, polymers like oligonucleotides.
These workers loaded their synthetic vesicles with a short single strand of deoxycytidine (dC) structured to provide a template for its replication. When the vesicles were placed in a medium containing (chemically modified) dG, these nucleotides entered the vesicles and assembled into a strand of Gs complementary to the template strand of Cs. Here, then, is a simple system that is a plausible model for the creation of the first cells from the primeval "soup" of organic molecules.
From Unicellular to Multicellular Organisms
This transition is probably the easiest to understand.
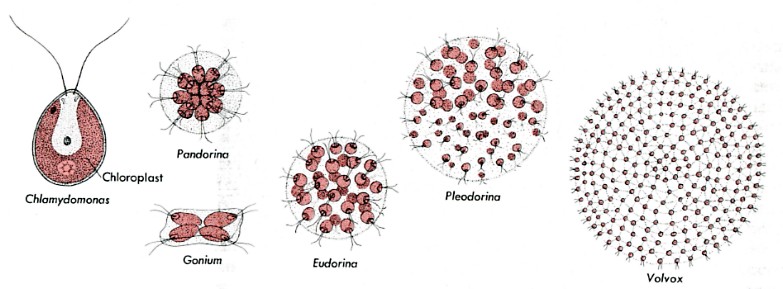
Several colonial flagellated green algae provide a clue. These species are called colonial because they are made up simply of clusters of independent cells. If a single cell of Gonium, Pandorina, or Eudorina is isolated from the rest of the colony, it will swim away looking quite like a Chlamydomonas cell. Then, as it undergoes mitosis, it will form a new colony with the characteristic number of cells in that colony.
(The figures are not drawn to scale. Their sizes range from Chlamydomonas which is about 10 µm in diameter - little larger than a human red blood cell - to Volvox whose sphere is some 350 µm in diameter - visible to the naked eye.)
The situation in Pleodorina and Volvox is different. In these organisms, some of the cells of the colony (most in Volvox) are not able to live independently. If a nonreproductive cell is isolated from a Volvox colony, it will fail to reproduce itself by mitosis and eventually will die. What has happened? In some way, as yet unclear, Volvox has crossed the line separating simple colonial organisms from truly multicellular ones. Unlike Gonium, Volvox cannot be considered simply a colony of individual cells. It is a single organism whose cells have lost their ability to live independently. If a sufficient number of them become damaged, the entire sphere of cells will die.
What has Volvox gained? In giving up their independence, the cells of Volvox have become specialists. No longer does every cell carry out all of life's functions (as in colonial forms); instead certain cells specialize to carry out certain functions while leaving other functions to other specialists. In Volvox this process goes no further than having certain cells specialize for reproduction while others, unable to reproduce themselves, fulfill the needs for photosynthesis and locomotion.
In more complex multicellular organisms, the degree of specialization is carried much further. Each cell has one or two precise functions to carry out. It depends on other cells to carry out all the other functions needed to maintain the life of the organism and thus its own.
The specialization and division of labor among cells is the outcome of their history of differentiation. One of the great problems in biology is how differentiation arises among cells, all of which having arisen by mitosis, share the same genes.
The genomes of both Chlamydomonas and Volvox have been sequenced. Although one is unicellular, the other multicellular, they have not only about the same number of protein-encoding genes (14,516 in Chlamydomonas, 14,520 in Volvox) but most of these are homologous. Volvox has only 58 genes that have no relatives in Chlamydomonas and even fewer unique mRNAs.
At one time, many of us would have expected that a multicellular organism like Volvox with its differentiated cells and complex life cycle would have had many more genes than a single-celled organism like Chlamydomonas. But that turns out not to be the case.
How to explain this apparent paradox? My guess is that just as we have seen in the evolution of animals, we are seeing here that the evolution of organismic complexity is not so much a matter of the evolution of new genes but rather the evolution of changes in the control elements (promoters and enhancers) that dictate how and where the basic tool kit of eukaryotic genes will be expressed .
The evidence is compelling that all these organisms are close relatives; that is, belong to the same clade. They illustrate how colonial forms could arise from unicellular ones and multicellular forms from colonial ones.
The Last Universal Common Ancestor (LUCA)?
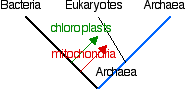 The 3 kingdoms of contemporary life — archaea, bacteria, and eukaryotes — all share many similarities of their metabolic and genetic systems . Presumably these were present in an organism that was ancestral to these groups: the "LUCA". Although there are not enough data at present to describe LUCA, comparative genomics and proteomics reveal a closer relationship between archaea and eukaryotes than either shares with the bacteria. Except, of course, for the mitochondria and chloroplasts that eukaryotes gained from bacterial endosymbionts. Whether the endosymbionts were acquired before or after a lineage of archaea had acquired a nucleus - and thus started the lineage of eukaryotes - is still uncertain.
The 3 kingdoms of contemporary life — archaea, bacteria, and eukaryotes — all share many similarities of their metabolic and genetic systems . Presumably these were present in an organism that was ancestral to these groups: the "LUCA". Although there are not enough data at present to describe LUCA, comparative genomics and proteomics reveal a closer relationship between archaea and eukaryotes than either shares with the bacteria. Except, of course, for the mitochondria and chloroplasts that eukaryotes gained from bacterial endosymbionts. Whether the endosymbionts were acquired before or after a lineage of archaea had acquired a nucleus - and thus started the lineage of eukaryotes - is still uncertain.
Creating Life?
When I headed off to college (in 1949), I wrote an essay speculating on the possibility that some day we would be able to create a living organism from nonliving ingredients. By the time I finished my formal studies in biology — having learned of the incredible complexity of even the simplest organism — I concluded that such a feat could never be accomplished.
Now I'm not so sure.
Several recent advances suggest that we may be getting close to creating life. (But note that these examples represent laboratory manipulations that do not necessarily reflect what may have happened when life first appeared.)
Examples:
- The ability to created membrane-enclosed vesicles that can take in small molecules and assemble them into polymers which remain within the "cell".
- The ability to assemble functional ribosomes — the structures that convert the information encoded in the genome into the proteins that run life — from their components.
- In 2008, scientists at the J. Craig Venter Institute (JCVI) reported (in Science 29 February 2008) that they had succeeded in synthesizing a complete bacterial chromosome — containing 582,970 base pairs — starting from single deoxynucleotides. The entire sequence of the genome of Mycoplasma genitalium was already known. Using this information, they synthesized some 10,000 short oligonucleotides (each about 50 bp long) representing the entire genitalium genome and then - step by step - assembled these into longer and longer fragments until finally they had made the entire circular DNA molecule that is the genome.
Could this be placed in the cytoplasm of a living cell and run it?
The same team showed in the previous year (see Science 3 August 2007) that they could insert an entire chromosome from one species of mycoplasma into the cytoplasm of a related species and, in due course, the recipient lost its own chromosome (perhaps destroyed by restriction enzymes encoded by the donor chromosome) and began expressing the phenotype of the donor. In short, they had changed one species into another. But the donor chromosome was made by the donor bacterium, not synthesized in the laboratory. However, there should be no serious obstacle to achieving the same genome transplantation with a chemically-synthesized chromosome.
They've done it! The same team reported on 20 May 2010 in the online Science Express that they had successfully transplanted a completely synthetic genome — based on that of Mycoplasma mycoides — into the related species Mycoplasma capricolum. The recipient strain grew well and soon acquired the phenotype of the M. mycoides donor.
- In the 4 April 2014 issue of Science (Annaluru, N. et al.), a large group of researchers - including many undergraduates at Johns Hopkins University - reported that they had successfully replaced the natural chromosome 3 in Saccharomyces cerevisiae (which has 16 chromosomes) with a totally-synthetic chromosome.
Their procedure:
- Chemically synthesize 69- to 79-nt oligonucleotides representing all the stretches of the known chromosome 9 sequence (which contains 316,617 base pairs) except for certain sequences such as transposons, many introns, and transfer RNA genes. In addition new, non-native, sequences such as loxP sites were included to aid future manipulations of the genome.
- Stitch these together into blocks of ~750 base pairs. This step was done in vitro by undergraduates enrolled in the "Build A Genome" class at Johns Hopkins.
- Introduce these into yeast cells which ligated them into stretches of DNA containing 2–4 thousand base pairs.
- Introduce these stepwise into yeast cells so that they replace the equivalent portions of the native chromosome.
- The result: a strain of yeast that grows just as well with its new artificial chromosome (now containing only 272,871 base pairs) as it did before.


