15.4O: Dendritic Cells
- Page ID
- 5451
Dendritic cells (DCs) get their name from their surface projections (that resemble the dendrites of neurons). They are found in most tissues of the body and are particularly abundant in those that are interfaces between the external and internal environments (e.g., skin, lungs, and the lining of the gastrointestinal tract) where they are ideally placed to encounter extrinsic antigens, including those expressed by invading pathogens.
Although there are several distinct subtypes of DCs, they all share these features:
- They are actively motile.
- They continuously sample their surroundings — ingesting antigens by endocytosis (using phagocytosis, receptor-mediated endocytosis, and pinocytosis).
- Many of these antigens are "self" antigens, e.g., dead cells, proteins in the extracellular fluid.
- But the antigens can also be foreign antigens, for example, bacteria that are resident in the body (e.g., in the colon) or that invade the body.
In either case, the ingested antigens are degraded in lysosomes into peptide fragments that are then displayed at the cell-surface nestled in class II MHC molecules.
Having ingested antigen in the tissue, they migrate to lymph nodes and spleen where they can meet up with T cells bearing the appropriate T-cell receptor for antigen (TCR).
What happens next depends on the nature of the antigen.
- Self antigens are presented to T cells without any costimulatory molecules. This interaction causes the T cells to divide for a brief time, but then they commit suicide by apoptosis and so cannot attack tissues of the body. The animal becomes tolerant to that antigen.
- Foreign antigens produce a different outcome. The dendritic cells becomes "activated' and begin to display not only
- the MHC-peptide complex for the TCR of the T cells but also
- costimulatory molecules, e.g. B7 which binds to CD28 on the T cell
The importance of dendritic cells in developing immunity to pathogens is dramatically shown in those rare infants who lack a functioning gene needed for the formation of dendritic cells. They are so severely immunodeficient that they are at risk of life-threatening infections.
What accounts for the activation of dendritic cells by foreign antigens but not by self antigens? Pathogens, especially bacteria, have molecular structures that
- are not shared with their host
- are shared by many related pathogens
- are relatively invariant; that is, do not evolve rapidly (in contrast, for example, to such pathogen molecules as the hemagglutinin and neuraminidase of influenza viruses).
These are called Pathogen-Associated Molecular Patterns (PAMPs)
Examples:
- the flagellin of bacterial flagella
- the peptidoglycan of Gram-positive bacteria
- the lipopolysaccharide (LPS, also called endotoxin) of Gram-negative bacteria
- double-stranded RNA. (Some viruses of both plants and animals have a genome of dsRNA. And many other viruses of both plants and animals have an RNA genome that in the host cell is briefly converted into dsRNA).
- unmethylated DNA (eukaryotes have many times more cytosines, in the dinucleotide CpG, with methyl groups attached).
Dendritic cells have a set of transmembrane receptors that recognize different types of PAMPs. These are called Toll-like receptors (TLRs) because of their homology to receptors first discovered and named in Drosophila.
TLRs identify the nature of the pathogen and turn on an effector response appropriate for dealing with it. These signaling cascades lead to the expression of various cytokine genes.
- Interleukin 12 (IL-12) drives the nearby T cells to become Th1 cells, which will provide help for cell-mediated immunity including attack against intracellular pathogens.
- IL-23, which promotes differentiation of the T cells into Th17 helper cells, which can deal with extracellular bacteria.
- IL-4, which promotes differentiation of the T cells into Th2 cells which provide help for antibody production by B cells.
Under other circumstances, activated dendritic cells may secrete TGF-β and IL-10, leading to the formation of regulatory T cells (Treg) that dampen immune responses.
Dendritic Cell Subsets
While all DCs share certain features, they actually represent a variety of cell types with different differentiation histories, phenotypic traits and, as outlined above, different effector functions.
Examples:
Myeloid Dendritic Cells
As their name implies, these cells ("mDCs") are derived from the same myeloid progenitors in the bone marrow that give rise to granulocytes and monocytes. They present antigen to T cells and activate the T cells by secreting large amounts of IL-12.
Plasmacytoid Dendritic Cells
These cells ("pDCs") get their name from their extensive endoplasmic reticulum which resembles that of plasma cells. However, unlike plasma cells that are machines for pumping out antibodies, pDCs secrete huge amounts of interferon-alpha especially in response to viral infections.
Plasmacytoid DCs have internal toll-like receptors:
- TLR-7 and TLR-8, which bind to the single-stranded RNA (ssRNA) genomes of such viruses as influenza, measles, and mumps.
- TLR-9, which binds to the unmethylated cytosines in the dinucleotide CpG in the DNA of the pathogen. (The cytosines in the host's CpG dinucleotides often have methyl groups attached.)
CD8+ vs. CD8− Dendritic Cells
These subsets are found in the mouse spleen.
- The CD8− subset presents antigen engulfed from the surroundings — using the class II pathway — to CD4+ helper T cells.
- The CD8+ subset can present extracellular antigens using the class I pathway as well. The peptide/MHC class I molecules are presented to CD8+ T cells which go on to become cytotoxic T lymphocytes (CTL). This phenomenon is called cross-presentation.
Dendritic cells can also present undegraded antigen to B cells; that is, antigen that has not been processed into peptide/MHC complexes.
Monocyte-derived Dendritic Cells (Mo-DCs)
Humans (and mice) have another population of dendritic cells that develop from blood-borne monocytes that have been exposed to Gram-negative bacteria (or their LPS). The LPS is detected by their TLR4 molecules. Mo-DCs can present antigen to both CD4+ T cells and CD8+ T cells (cross-presentation).
Ralph Steinman
Ralph Steinman, the pioneer in the study of dendritic cells, has provided striking visual evidence of the cellular interactions between antigen-presenting dendritic cells, T cells, and B cells. When spleen cells are cultured with antigen, tight clusters of cells form (see figure). The clustering occurs in two phases:
- an early phase (days 0–2) during which only the dendritic cells and T cells need to be present to form clusters
- a later phase (days 2–5) when antigen-primed B cells enter the cluster and differentiate into antibody-secreting cells

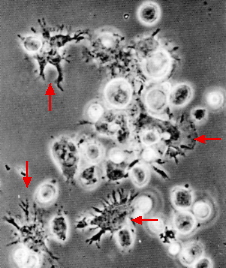
Pictures Courtesy of Ralph Steinman from K. Inaba et al., J. Exp. Med. 160:858, 1984
Homing
Some dendritic cells not only activate T cells to respond to a particular antigen but tell them where to go to deal with that antigen.
Two examples:
Antigens in the skin
- Dendritic cells engulf antigens in (or even on!*) the skin and, while doing so, convert calciferol (vitamin D3) present in the skin into calcitriol (1,25[OH]2 Vitamin D3).
- When they activate the appropriate T cells in a nearby lymph node, the calcitriol induces those T cells to express a surface receptor designated CCR10 (a member of the CC chemokine receptor family).
- CCR10 binds the chemokine CCL27 — which is present in the skin.
- So when these T cells reajavascript:void('Remove Anchor')ch the skin, they stop their travels and go to work there.
Antigens in the GI tract
- Dendritic cells in the lining of the intestine are always busy engulfing the many antigens present there.
- While doing so, they convert the abundant amount of retinol (vitamin A) there into retinoic acid.
- When they activate the appropriate T cells in a nearby lymph node, the retinoic acid induces those T cells to express another CC chemokine receptor designated CCR9.
- CCR9 binds the chemokine CCL25 present in the intestine.
- So when these T cells reach the intestine, they stop their travels and go to work. (CCL25 also attracts IgA-secreting B cells.)
Switching Homing Directions
Most vaccines are given by injection into muscle or skin. This works very well for inducing systemic immunity; that is, IgG antibodies in the blood able to attack pathogens (e.g., tetanus) that are present in the blood.
Injected vaccines do not work as well for illnesses caused by intestinal pathogens such as
- typhoid fever (caused by Salmonella typhi)
- cholera (caused by Vibrio cholerae)
However, a group of German immunologists reported in July 2011 that:
- whereas dendritic cells receiving antigens from injections under the skin influence T cells to migrate back to the skin (as we saw above),
- if these subcutaneous injections are accompanied by injections of retinoic acid, the T cells migrate to the intestine instead.
- CCR9+ plasma cells secreting antigen-specific IgA antibodies also appeared in the intestine.
Using this technique, these workers were able to protect their mice from mouse typhoid (Salmonella typhimurium) and cholera toxin.


