W2017_Lecture_13_reading
- Page ID
- 8137
\( \newcommand{\vecs}[1]{\overset { \scriptstyle \rightharpoonup} {\mathbf{#1}} } \)
\( \newcommand{\vecd}[1]{\overset{-\!-\!\rightharpoonup}{\vphantom{a}\smash {#1}}} \)
\( \newcommand{\dsum}{\displaystyle\sum\limits} \)
\( \newcommand{\dint}{\displaystyle\int\limits} \)
\( \newcommand{\dlim}{\displaystyle\lim\limits} \)
\( \newcommand{\id}{\mathrm{id}}\) \( \newcommand{\Span}{\mathrm{span}}\)
( \newcommand{\kernel}{\mathrm{null}\,}\) \( \newcommand{\range}{\mathrm{range}\,}\)
\( \newcommand{\RealPart}{\mathrm{Re}}\) \( \newcommand{\ImaginaryPart}{\mathrm{Im}}\)
\( \newcommand{\Argument}{\mathrm{Arg}}\) \( \newcommand{\norm}[1]{\| #1 \|}\)
\( \newcommand{\inner}[2]{\langle #1, #2 \rangle}\)
\( \newcommand{\Span}{\mathrm{span}}\)
\( \newcommand{\id}{\mathrm{id}}\)
\( \newcommand{\Span}{\mathrm{span}}\)
\( \newcommand{\kernel}{\mathrm{null}\,}\)
\( \newcommand{\range}{\mathrm{range}\,}\)
\( \newcommand{\RealPart}{\mathrm{Re}}\)
\( \newcommand{\ImaginaryPart}{\mathrm{Im}}\)
\( \newcommand{\Argument}{\mathrm{Arg}}\)
\( \newcommand{\norm}[1]{\| #1 \|}\)
\( \newcommand{\inner}[2]{\langle #1, #2 \rangle}\)
\( \newcommand{\Span}{\mathrm{span}}\) \( \newcommand{\AA}{\unicode[.8,0]{x212B}}\)
\( \newcommand{\vectorA}[1]{\vec{#1}} % arrow\)
\( \newcommand{\vectorAt}[1]{\vec{\text{#1}}} % arrow\)
\( \newcommand{\vectorB}[1]{\overset { \scriptstyle \rightharpoonup} {\mathbf{#1}} } \)
\( \newcommand{\vectorC}[1]{\textbf{#1}} \)
\( \newcommand{\vectorD}[1]{\overrightarrow{#1}} \)
\( \newcommand{\vectorDt}[1]{\overrightarrow{\text{#1}}} \)
\( \newcommand{\vectE}[1]{\overset{-\!-\!\rightharpoonup}{\vphantom{a}\smash{\mathbf {#1}}}} \)
\( \newcommand{\vecs}[1]{\overset { \scriptstyle \rightharpoonup} {\mathbf{#1}} } \)
\(\newcommand{\longvect}{\overrightarrow}\)
\( \newcommand{\vecd}[1]{\overset{-\!-\!\rightharpoonup}{\vphantom{a}\smash {#1}}} \)
\(\newcommand{\avec}{\mathbf a}\) \(\newcommand{\bvec}{\mathbf b}\) \(\newcommand{\cvec}{\mathbf c}\) \(\newcommand{\dvec}{\mathbf d}\) \(\newcommand{\dtil}{\widetilde{\mathbf d}}\) \(\newcommand{\evec}{\mathbf e}\) \(\newcommand{\fvec}{\mathbf f}\) \(\newcommand{\nvec}{\mathbf n}\) \(\newcommand{\pvec}{\mathbf p}\) \(\newcommand{\qvec}{\mathbf q}\) \(\newcommand{\svec}{\mathbf s}\) \(\newcommand{\tvec}{\mathbf t}\) \(\newcommand{\uvec}{\mathbf u}\) \(\newcommand{\vvec}{\mathbf v}\) \(\newcommand{\wvec}{\mathbf w}\) \(\newcommand{\xvec}{\mathbf x}\) \(\newcommand{\yvec}{\mathbf y}\) \(\newcommand{\zvec}{\mathbf z}\) \(\newcommand{\rvec}{\mathbf r}\) \(\newcommand{\mvec}{\mathbf m}\) \(\newcommand{\zerovec}{\mathbf 0}\) \(\newcommand{\onevec}{\mathbf 1}\) \(\newcommand{\real}{\mathbb R}\) \(\newcommand{\twovec}[2]{\left[\begin{array}{r}#1 \\ #2 \end{array}\right]}\) \(\newcommand{\ctwovec}[2]{\left[\begin{array}{c}#1 \\ #2 \end{array}\right]}\) \(\newcommand{\threevec}[3]{\left[\begin{array}{r}#1 \\ #2 \\ #3 \end{array}\right]}\) \(\newcommand{\cthreevec}[3]{\left[\begin{array}{c}#1 \\ #2 \\ #3 \end{array}\right]}\) \(\newcommand{\fourvec}[4]{\left[\begin{array}{r}#1 \\ #2 \\ #3 \\ #4 \end{array}\right]}\) \(\newcommand{\cfourvec}[4]{\left[\begin{array}{c}#1 \\ #2 \\ #3 \\ #4 \end{array}\right]}\) \(\newcommand{\fivevec}[5]{\left[\begin{array}{r}#1 \\ #2 \\ #3 \\ #4 \\ #5 \\ \end{array}\right]}\) \(\newcommand{\cfivevec}[5]{\left[\begin{array}{c}#1 \\ #2 \\ #3 \\ #4 \\ #5 \\ \end{array}\right]}\) \(\newcommand{\mattwo}[4]{\left[\begin{array}{rr}#1 \amp #2 \\ #3 \amp #4 \\ \end{array}\right]}\) \(\newcommand{\laspan}[1]{\text{Span}\{#1\}}\) \(\newcommand{\bcal}{\cal B}\) \(\newcommand{\ccal}{\cal C}\) \(\newcommand{\scal}{\cal S}\) \(\newcommand{\wcal}{\cal W}\) \(\newcommand{\ecal}{\cal E}\) \(\newcommand{\coords}[2]{\left\{#1\right\}_{#2}}\) \(\newcommand{\gray}[1]{\color{gray}{#1}}\) \(\newcommand{\lgray}[1]{\color{lightgray}{#1}}\) \(\newcommand{\rank}{\operatorname{rank}}\) \(\newcommand{\row}{\text{Row}}\) \(\newcommand{\col}{\text{Col}}\) \(\renewcommand{\row}{\text{Row}}\) \(\newcommand{\nul}{\text{Nul}}\) \(\newcommand{\var}{\text{Var}}\) \(\newcommand{\corr}{\text{corr}}\) \(\newcommand{\len}[1]{\left|#1\right|}\) \(\newcommand{\bbar}{\overline{\bvec}}\) \(\newcommand{\bhat}{\widehat{\bvec}}\) \(\newcommand{\bperp}{\bvec^\perp}\) \(\newcommand{\xhat}{\widehat{\xvec}}\) \(\newcommand{\vhat}{\widehat{\vvec}}\) \(\newcommand{\uhat}{\widehat{\uvec}}\) \(\newcommand{\what}{\widehat{\wvec}}\) \(\newcommand{\Sighat}{\widehat{\Sigma}}\) \(\newcommand{\lt}{<}\) \(\newcommand{\gt}{>}\) \(\newcommand{\amp}{&}\) \(\definecolor{fillinmathshade}{gray}{0.9}\)Fermentation and regeneration of NAD+
Section Summary
This section discusses the process of fermentation. Due to the heavy emphasis in this course on central carbon metabolism the discussion of fermentation understandably focuses on the fermentation of pyruvate. Nevertheless, some of the core the principles that we cover in this section apply equally well to the fermentation of many other small molecules.
The Purpose of Fermentation
The oxidation of a variety of small organic compounds is a process that is utilized by many organisms to garner energy for cellular maintenance and growth. The oxidation of glucose via glycolysis is one such pathway. Several key steps in the oxidation of glucose to pyruvate involve the reduction of the electron/energy shuttle NAD+ to NADH. At the end of section 5.3 you were posed with the challenge of trying to figure out what options the cell might reasonably have to re-oxidize the NADH to NAD+ in order to avoid consuming the available pools of NAD+ and thus stopping glycolysis. Put differently, during glycolysis cells can generate large amounts of NADH and slowly exhaust their supplies of NAD+. If glycolysis is to continue, the cell must find a way to regenerate NAD+, either by synthesis or by some form of recycling.
In the absence of any other process - that is, if we consider glycolysis alone - it is not immediately obvious what the cell might do. One choice is to try putting the electrons that were once stripped off of the glucose derivatives right back onto the downstream product, pyruvate or one of its derivatives. We can generalize the process by describing it as the returning of electrons to the molecule that they were once removed from, usually to restore pools of a oxidizing agent. This, in short, is fermentation. As we will discuss in a different section, the process of respiration can also regenerate the pools of NAD+ from NADH. Cells lacking respiratory chains or in conditions where using the respiratory chain is unfavorable may choose fermentation as an alternative mechanism for garnering energy from small molecules.
An Example: Lactic Acid Fermentation
An everyday example of a fermentation reaction is the reduction of pyruvate to lactate by the lactic acid fermentation reaction. This reaction should be familiar to you, it occurs in our muscles when we exert ourselves during exercise. When we exert ourselves our muscles require large amounts of ATP to perform the work we are demanding of them. As the ATP is consumed, the muscle cells are unable to keep up with the demand for respiration, O2 becomes limiting and NADH accumulates. Cells need to get rid of the excess and regenerate NAD+, so pyruvate serves as an electron acceptor, generating lactate and oxidizing NADH to NAD+. Many bacteria use this pathway as a way to complete the NADH/NAD+ cycle. You may be familiar with this process from in products like sauerkraut and yogurt. The chemical reaction of lactic acid fermentation is the following:
Pyruvate + NADH ↔ lactic acid + NAD+

Lactic acid fermentation converts pyruvate (a slightly oxidized carbon compound) to lactic acid. In the process, NADH is oxidized to form NAD+.
Attribution: Marc T. Facciotti (original work)
Energy Story for Fermentation of Pyruvate to Lactate
An example (if a bit lengthy) energy story for lactic acid fermentation:
The reactants are pyruvate, NADH and a proton. The products are lactate and NAD+. The process of fermentation results in the reduction of pyruvate to form lactic acid and the oxidation of NADH to form NAD+. Electrons from NADH and a proton are used to reduce pyruvate into lactate. If we examine a table of standard reduction potential we see under standard conditions that a transfer of electrons from NADH to pyruvate to form lactate is exergonic and thus thermodynamically spontaneous. The reduction and oxidation steps of the reaction are coupled and catalyzed by the enzyme lactate dehydrogenase.
A second example: Alcohol Fermentation
Another familiar fermentation process is alcohol fermentation, which produces ethanol, an alcohol. The alcohol fermentation reaction is the following:
Ethanol fermentation is a two step process. Pyruvate (pyruvic acid) is first converted into carbon dioxide and acetaldehyde. The second step, converts acetaldehyde to ethanol and oxidizes NADH to NAD+.
Attribution: Marc T. Facciotti (original work)
In the first reaction, a carboxyl group is removed from pyruvic acid, releasing carbon dioxide as a gas (some of you may be familiar with this as a key component of various beverages). The second reaction removes electrons from NADH, forming NAD+ and producing ethanol (another familiar compound - usually in the same beverage) from the acetaldehyde, which accepts the electrons.
Suggested discussion
Write a complete energy story for alcohol fermentation. Propose possible benefits of this type of fermentation for the single celled yeast organism.
Potentially Helpful Videos
Here is a chemwiki link on fermentation reactions.
Fermentation pathways are numerous
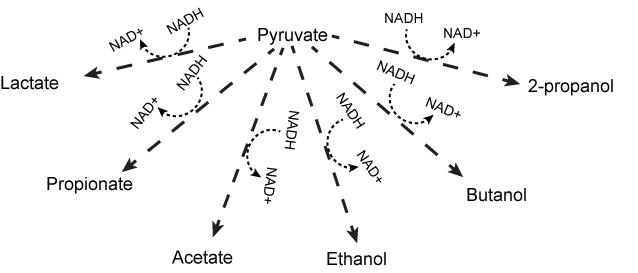
While the lactic acid fermentation and alcohol fermentation pathways described above are examples, there are many more reactions (too numerous to go over) that Nature has evolved to complete the NADH/NAD+ cycle. It is important that you understand the general concepts behind these reactions. In general, cells try to maintain a balance or constant ratio between NADH and NAD+; when this ratio becomes unbalanced, the cell compensates by modulating other reactions to compensate. The only requirement for a fermentation reaction is that it uses a small organic compound as an electron acceptor for NADH and regenerates NAD+. Other familiar fermentation reactions include, ethanol fermentation (as in beer and bread) and propionic fermentation (it's what makes the holes in swiss cheese) and malolactic fermentation (it's what gives chardonnay is more mellow flavor, more conversion of malate to lactate the softer the wine). In the figure below you can see a large variety of fermentation reactions that various bacteria use to reoxidize NADH to NAD+. All of these reactions start with pyruvate or a derivative of pyruvate matabolism, such as oxaloacetate, or formate. Pyruvate is produced from the oxidation of sugars (glucose or ribose) or other small reduced organic molecules. It should also be noted that other compounds can be used as fermentation substrates besides pyruvate and its derivatives. These include: methane fermentation, sulfide fermentation, or the fermentation of nitrogenous compounds such as amino acids. You are not expected to memorize all of these pathways. You are, however, expected to recognize a pathway that returns electrons to products of the compounds that were originally oxidized to recycle the NAD+/NADH pool and to associate that process with fermentation.
Various fermentation pathways using pyruvate as the initial substrate. In the figure, pyruvate is reduced to a variety of products via different and sometimes multistep (dashed arrows represent possible multiple step processes) reactions. All details are deliberately not shown. The key point is to appreciate that fermentation is a broad term not solely associated with the conversion of pyruvate to lactic acid or ethanol.
Source: Original work Marc T. Facciotti
A note on the link between substrate level phosphorylation and fermentation
Fermentation occurs in the absence of molecular oxygen (O2). It is an anaerobic process. Notice there is no O2 in any of the fermentation reactions shown above. Many of these reactions are quite ancient, hypothesized to be some of the first energy generating metabolic reactions to evolve. This makes sense if we consider the following:
- The early atmosphere was highly reduced, with little molecular oxygen readily available.
- Small, highly reduced organic molecules were relatively available, arising from a variety of chemical reactions.
- These types of reactions, pathways and enzymes are found in many different types of organisms, including bacteria, archaea and eukaryotes, suggesting these are very ancient reactions.
- The process evolved long before O2 was found in the environment.
- The substrates, highly reduced small organic molecules, like glucose, were readily available.
- The end products of many fermentation reactions are small organic acids, produced by the oxidation of the initial substrate.
- The process is coupled to substrate level phosphorylation reactions. That is, small reduced organic molecules are oxidized and ATP is generated by first a red/ox reaction followed by the substrate level phosphorylation.
- This suggests that substrate level phosphorylation and fermentation reactions co-evolved.
Suggested discussion
If the hypothesis is correct, that substrate level phosphorylation and fermentation reactions co-evolved and were the first forms of energy metabolism that cells used to generate ATP, then what would be the consequences of such reactions over time? What if these were the only forms of energy metabolism available over hundreds of thousands of years? What if cells were isolated in a small closed environment? What if the small reduced substrates were not being produced at the same rate of consumption during this time?
Consequences of fermentation
Imagine the world where fermentation is the primary mode for extracting energy from small molecules. As populations thrive, they reproduce and consume the abundance of small reduced organic molecules in the environment, producing acids. One consequence is the acidification (decrease of pH) of the environment, including the internal cellular environment. This is not so good, since changes in pH can have a profound influence on the function and interactions among various biomolecules. Therefore mechanisms needed to evolved that could remove the various acids. Fortunately, in an environment rich in reduced compounds, substrate level phosphorylation and fermentation can produce large quantities of ATP.
It is hypothesized that this scenario was the beginning of the evolution of the F0F1 ATPase, a molecular machine that hydrolyzes ATP and translocates protons across the membrane (we'll see this again in the next section). With the F0F1 ATPase, the ATP produced from fermentation could now allow for the cell to maintain pH homeostasis by coupling the free energy of hydrolysis of ATP to the transport of protons out of the cell. The down side is that now cells are pumping all of these protons into the environment, which will now start to acidify.
Suggested discussion
If the hypothesis is correct, that the F0F1 ATPase also co-evolved with substrate level phosphorylation and fermentation reactions, then what would happen over time to the environment? While small reduced organic compounds may have been initially abundant, if fermentation "took off" at some point the reduced compounds would run out and ATP might then become scarce as well. That's a problem. Thinking with the design challenge rubric define the problem(s) facing the cell in this hypothesized environment. What are other potential mechanism or ways Nature could overcome the problem(s)?
Oxidation of Pyruvate and the TCA Cycle
Overview of Pyruvate Metabolism and the TCA Cycle
Under appropriate conditions, pyruvate can be further oxidized. One of the most studied oxidation reactions involving pyruvate is a two part reaction involving NAD+ and molecule called co-enzyme A, often abbreviated simply as "CoA". This reaction oxidizes pyruvate, leads to a loss of one carbon via decarboxylation, and creates a new molecule called acetyl-CoA. The resulting acetyl-CoA can enter several pathways for the biosynthesis of larger molecules or it can be routed to another pathway of central metabolism called the Citric Acid Cycle, sometimes also called the Krebs Cycle, or Tricarboxylic Acid (TCA) Cycle. Here the remaining two carbons in the acetyl group can either be further oxidized or serve again as precursors for the construction of various other molecules. We discuss these scenarios below.
The different fates of pyruvate and other end products of glycolysis
The glycolysis module left off with the end-products of glycolysis: 2 pyruvate molecules, 2 ATPs and 2 NADH molecules. This module and the module on fermentation will explore what the cell may now do with the pyruvate, ATP and NADH that were generated.
The fates of ATP and NADH
In general, ATP can be used for or coupled to a variety of cellular functions including biosynthesis, transport, replication etc. We will see many such examples throughout the course.
What to do with the NADH however, depends on the conditions under which the cell is growing. In some cases, the cell will opt to rapidly recycle NADH back into to NAD+. This occurs through a process called fermentation in which the electrons initially taken from the glucose derivatives are returned to more downstream products via another redox transfer (described in more detail in the module on fermentation). Alternatively, NADH can be recycled back into NAD+ by donating electrons to something known as an electron transport chain (this is covered in the module on respiration and electron transport).
The fate of cellular pyruvate
- Pyruvate can be used as a terminal electron acceptor (either directly or indirectly) in fermentation reactions, and is discussed in the fermentation module.
- Pyruvate could be secreted from the cell as a waste product.
- Pyruvate could be further oxidized to extract more free energy from this fuel.
- Pyruvate can serve as a valuable intermediate compound linking some of the core carbon processing metabolic pathways
The further oxidation of pyruvate
In respiring bacteria and archaea, the pyruvate is further oxidized in the cytoplasm. In aerobically respiring eukaryotic cells, the pyruvate molecules produced at the end of glycolysis are transported into mitochondria, which are sites of cellular respiration and house oxygen consuming electron transport chains (ETC in module on respiration and electron transport). Organisms from all three domains of life share similar mechanisms to further oxidize the pyruvate to CO2. First pyruvate is decarboxylated and covalently linked to co-enzyme A via a thioester linkage to form the molecule known as acetyl-CoA. While acetyl-CoA can feed into multiple other biochemical pathways we now consider its role in feeding the circular pathway known as the Tricarboxylic Acid Cycle, also referred to as the TCA cycle, the Citric Acid Cycle or the Krebs Cycle. This process is detailed below.
Conversion of Pyruvate into Acetyl-CoA
In a multistep reaction catalyzed by the enzyme pyruvate dehydrogenase, pyruvate is oxidized by NAD+, decarboxylated, and covalently linked to a molecule of co-enzyme A via a thioester bond. Remember: there are two pyruvate molecules produced at the end of glycolysis for every molecule of glucose metabolized; thus, two of the six carbons will have been removed at the end of both steps. The release of the carbon dioxide is important here, this reaction often results in a loss of mass from the cell as the CO2 will diffuse or be transported out of the cell and become a waste product. In addition, one molecule of NAD+ is reduced to NADH during this process per molecule of pyruvate oxidized.
Suggested discussion
We have already discussed the formation of a thioester bond in another unit and lecture. Where was this specifically? What was the energetic significance of this bond? What are the similarities and differences between this example (formation of thioester with CoA) and the previous example of this chemistry?

Suggested discussion
Describe the flow and transfer of energy in this reaction using good vocabulary - (e.g. reduced, oxidized, redox, endergonic, exergonic, thioester, etc. etc.). You can peer edit - someone can start a description, another person can make it better, another person can improve it more etc. . .
In the presence of a suitable terminal electron acceptor, acetyl CoA delivers (exchanges a bond) its acetyl group to a four-carbon molecule, oxaloacetate, to form citrate (designated the first compound in the cycle). This cycle is called by different names: the citric acid cycle (for the first intermediate formed—citric acid, or citrate), the TCA cycle (since citric acid or citrate and isocitrate are tricarboxylic acids), and the Krebs cycle, after Hans Krebs, who first identified the steps in the pathway in the 1930s in pigeon flight muscles.
The Tricarboxcylic Acid (TCA) Cycle
In bacteria and archaea reactions in the TCA cycle typically happen in the cytosol. In eukaryotes, the TCA cycle takes place in the matrix of mitochondria. Almost all (but not all) of the enzymes of the TCA cycle are water soluble (not in the membrane), with the single exception of the enzyme succinate dehydrogenase, which is embedded in the inner membrane of the mitochondrion (in eukaryotes). Unlike glycolysis, the TCA cycle is a closed loop: the last part of the pathway regenerates the compound used in the first step. The eight steps of the cycle are a series of redox, dehydration, hydration, and decarboxylation reactions that produce two carbon dioxide molecules, one ATP, and reduced forms of NADH and FADH2.
In the TCA cycle, the acetyl group from acetyl CoA is attached to a four-carbon oxaloacetate molecule to form a six-carbon citrate molecule. Through a series of steps, citrate is oxidized, releasing two carbon dioxide molecules for each acetyl group fed into the cycle. In the process, three NAD+ molecules are reduced to NADH, one FAD molecule is reduced to FADH2, and one ATP or GTP (depending on the cell type) is produced (by substrate-level phosphorylation). Because the final product of the TCA cycle is also the first reactant, the cycle runs continuously in the presence of sufficient reactants. (credit: modification of work by “Yikrazuul”/Wikimedia Commons)
Steps in the TCA Cycle
Step 1:
The first step of the cycle is a condensation reaction involving the two-carbon acetyl group of acetyl-CoA with one four-carbon molecule of oxaloacetate. The products of this reaction are the six-carbon molecule citrate and free co-enzyme A. This step is considered irreversible because it is so highly exergonic. Moreover, the rate of this reaction is controlled through negative feedback by ATP. If ATP levels increase, the rate of this reaction decreases. If ATP is in short supply, the rate increases. If not already, the reason will become evident shortly.
Step 2:
In step two, citrate loses one water molecule and gains another as citrate is converted into its isomer, isocitrate.
Step 3:
In step three, isocitrate is oxidized by NAD+ and decarboxylated. Keep track of the carbons! This carbon now more than likely leaves the cell as waste and is no longer available for building new biomolecules. The oxidation of isocitrate therefore produces a five-carbon molecule, α-ketoglutarate, a molecule of CO2 and reduced NADH. This step is also regulated by negative feedback from ATP and NADH, and a positive effect from ADP.
Step 4:
Step 4 is catalyzed by the enzyme succinate dehydrogenase. Here, α-ketoglutarate is further oxidized by NAD+. This oxidation again leads to a decarboxylation and thus the loss of another carbon as waste. So far two carbons have come into the cycle from acetyl-CoA and two have left as CO2. At this stage, There is no net gain of carbons assimilated from the glucose molecules that are oxidized to this stage of metabolism. Unlike the previous step however succinate dehydrogenase - like pyruvate dehydrogenase before it - couples the free energy of the exergonic redox and decarboxylation reaction to drive the formation of a thioester bond between the substrate co-enzyme A and succinate (what is left after the decarboxylation). Succinate dehydrogenase is regulated by feedback inhibition of ATP, succinyl-CoA, and NADH.
Suggested discussion
We have seen several steps in this and other pathways that are regulated by allosteric feedback mechanisms. Is there something(s) in common about these reactions? Why might these be good steps to regulate?
Suggested discussion
The thioester bond has reappeared! Use the terms we've been learning (e.g. reduction, oxidation, coupling, exergonic, endergonic etc.) to describe the formation of this bond and below its hydrolysis.
Step 5:
In step five, There is a substrate level phosphorylation event, where inorganic phosphate (Pi) is added to GDP or ADP to form GTP (an ATP equivalent for our purposes)or ATP. The energy that drives this substrate level phosphorylation event comes from the hydrolysis of the CoA molecule from succinyl~CoA to form succinate. Why is either GTP or ATP produced? In animal cells there are two isoenzymes (different forms of an enzyme that carries out the same reaction), for this step, depending upon the type of animal tissue in which they are found. One form is found in tissues that use large amounts of ATP, such as heart and skeletal muscle. This form produces ATP. The second form of the enzyme is found in tissues that have a high number of anabolic pathways, such as liver. This form produces GTP. GTP is energetically equivalent to ATP; however, its use is more restricted. In particular, the process of protein synthesis primarily uses GTP. Most bacterial systems produce GTP in this reaction.
Step 6:
Step six is another redox reactions in which succinate is oxidized by FAD+ into fumarate. Two hydrogen atoms are transferred to FAD+, producing FADH2. The difference in reduction potential between the fumarate/succinate and NAD+/NADH half reactions is insufficient to make NAD+ a suitable reagent for oxidizing succinate with NAD+ under cellular conditions. However, the difference in reduction potential with the FAD+/FADH2 half reaction is adequate to oxidize succinate and reduce FAD+. Unlike NAD+, FAD+ remains attached to the enzyme and transfers electrons to the electron transport chain directly. This process is made possible by the localization of the enzyme catalyzing this step inside the inner membrane of the mitochondrion or plasma membrane (depending on whether the organism in question is eukaryotic or not).
Step 7:
Water is added to fumarate during step seven, and malate is produced. The last step in the citric acid cycle regenerates oxaloacetate by oxidizing malate with NAD+. Another molecule of NADH is produced in the process.
Summary
Note that this process completely oxidizes 1 molecule of pyruvate, a 3 carbon organic acid, to 3 molecules of CO2. During this process, 4 molecules of NADH, 1 molecule of FADH2, and 1 molecule of GTP (or ATP) are produced. For respiring organisms this is a significant source of energy, since each molecule of NADH and FAD2 can feed directly into the electron transport chain, and as we will soon see, the subsequent redox reactions will indirectly energetically drive the synthesis of additional ATP. This suggests that the TCA cycle is primarily an energy generating mechanism; evolved to extract or convert as much potential energy form the original energy source to a form cells can use, ATP (or the equivalent) or an energized membrane. However, - and let us not forget - the other important outcome of evolving this pathway is the ability to produce several precursor or substrate molecules necessary for various catabolic reactions (this pathway provides some of the early building blocks to make bigger molecules). As we will discuss below, there is a strong link between carbon metabolism and energy metabolism.
Energy Stories
Work on building some energy stories yourself
There are a few interesting reactions that involve large transfers of energy and rearrangements of matter. Pick a few. Rewrite a reaction in your notes, and practice constructing an energy story. You now have the tools to discuss the energy redistribution in the context of broad ideas and terms like exergonic and endergonic. You also have the ability to begin discussing mechanism (how these reactions happen) by invoking enzyme catalysts. See your instructor and/or TA and check with you classmates to self-test on how you're doing.
Connections to Carbon Flow
One hypothesis that we have started exploring in this reading and in class is the idea that "central metabolism" evolved as a means of generating carbon precursors for catabolic reactions. Our hypothesis also states that as cells evolved, these reactions became linked into pathways: glycolysis and the TCA cycle, as a means to maximize their effectiveness for the cell. A side benefit to this evolving metabolic pathway was the generation of NADH from the complete oxidation of glucose - we saw the beginning of this idea when we discussed fermentation. We have already discussed how glycolysis not only provides ATP from substrate level phosphorylation, but also yields a net of 2 NADH molecules and 6 essential precursores: glucose-6-P, fructose-6-P, trios-P, 3-phosphoglycerate, phosphoenolphyruvate, and of course pyruvate. While ATP can be used by the cell directly as an energy source, NADH posses a problem and must be recycled back into NAD+, to keep the cycle in balance. As we see in detail in the fermentation module, the most ancient way cells deal with this poblem is to use fermentation reactions to regenerate NAD+.
During the process of pyruvate oxidation via the TCA cycle 4 additional essential precursors are formed: acetyle~CoA, alpha-ketoglutarate, oxaloacetate, and succinyl~CoA. Three molecules of CO2 are lost and this represents a net loss of mass for the cell. These precursors, however, are substrates for a variety of catabolic reactions including the production of amino acids, fatty acids, and various co-factors, such as heme. This means that the rate of reaction through the TCA cycle will be sensitive to the concentrations of each metabolic intermediate (more on the thermodynamics in class). A metabolic intermediate is a compound that is produced by one reaction (a product) and then acts as a substrate for the next reaction. This also means that metabolic intermediates, in particular the 4 essential precursors, can be removed at any time for catabolic reactions, if there is a demand.
Not all cells have a functional TCA cycle
Since all cells require the ability of make these precursor molecules, one might expect that all organisms would have a fully functional TCA cycle. In fact, the cells of many organisms DO NOT have a the enzymes to form a complete cycle - all cells, however, DO have the capability of making the 4 TCA cycle precursors noted in the previous paragraph. How can the cells make precursors and not have a full cycle? Remember that most of these reactions are freely reversible, so, if NAD+ is required to for the oxidation of pyruvate or acetyl~CoA, then the reverse reactions would require NADH. This process is often referred to as the reductive TCA cycle. To drive these reactions in reverse (with respect to the direction discussed above) requires energy, in this case carried by ATP and NADH. If you get ATP and NADH driving a pathway one direction, it stands to reason that driving it in reverse will require ATP and NADH as "inputs".
Suggested discussion
Why might some organisms not have evolved a fully oxidative TCA cycle? Remember, cells need to keep a balance in the NAD+ to NADH ratio as well as the [ATP]/[AMP]/[ADP] ratios.
Additional Links
Here are some additional links to videos and pages that you may find useful.