6.3: Capsule Staining Procedure
( \newcommand{\kernel}{\mathrm{null}\,}\)
Many bacteria secrete a slimy, viscous covering called a capsule or glycocalyx (see Fig. 1). This is usually composed of polysaccharide, polypeptide, or both
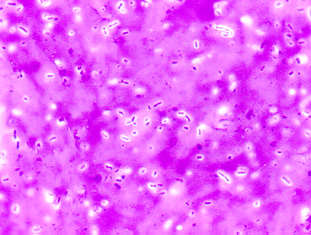
Figure 6.3.1: Capsule stain of Klebsiella aerogenes (formerly known as Enterobacter aerogenes). Note colorless capsules surrounding purple bacilli. ( Copyright; Gary E. Kaiser, Ph.D. The Community College of Baltimore County, Catonsville Campus CC-BY-3.0 )
The ability to produce a capsule is an inherited property of the organism, but the capsule is not an absolutely essential cellular component. Capsules are often produced only under specific growth conditions. Even though not essential for life, capsules probably help bacteria to survive in nature. Capsules help many pathogenic and normal flora bacteria to initially resist phagocytosis by the host's phagocytic cells. In soil and water, capsules help prevent bacteria from being engulfed by protozoans. Capsules also help many bacteria to adhere to surfaces and thus resist flushing. It also enables many bacteria to form biofilms. A biofilm consists layers of bacterial populations adhering to host cells and embedded in a common capsular mass.
For further information on the bacterial capsules, see the following in your CourseArc Lectures:
ORGANISM
Skim Milk broth culture of Klebsiella aerogenes (formerly known as Enterobacter aerogenes). The skim milk supplies essential nutrients for capsule production and provides a slightly stainable background.
PROCEDURE (to be done individually)
1. Stir up the Skim Milk broth culture with your loop and place 2-3 loops of Klebsiella aerogenes on a microscope slide.
2. Using your inoculating loop, spread the sample out to cover about one inch of the slide.
3. Let it completely air dry. Do not heat fix. Capsules stick well to glass, and heat may destroy the capsule.
4. Stain with crystal violet for one minute.
5. Wash off the excess dye with 20% copper sulfate solution.
6. Shake off the excess copper sulfate solution and immediately blot dry.
7. Observe using oil immersion microscopy. The organism and the milk dried on the slide will pick up the purple dye while the capsule will remain colorless.
8 . Make sure you carefully pour the used dye in your staining tray into the waste dye collection container, not down the sink.
9. Observe the demonstration capsule stain of Streptococcus lactis , an encapsulated bacterium that is normal microbiota in milk (see Fig. 2).
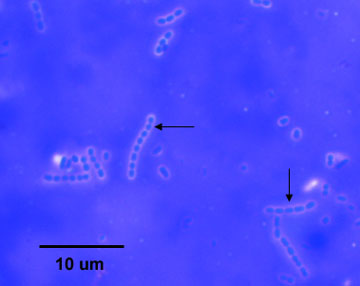
Figure 6.3.1: Capsule stain of Streptococcus lactis. Note colorless capsules surrounding purple bacilli. ( Copyright; Gary E. Kaiser, Ph.D. The Community College of Baltimore County, Catonsville Campus CC-BY-3.0 )
Results
Make a drawing of your capsule stain preparation of Enterobacter aerogenes and the demonstration capsule stain of Streptococcus pneumoniae.
|
Capsule stain of Capsule stain of |
Capsule stain of
|
Contributors and Attributions
Dr. Gary Kaiser (COMMUNITY COLLEGE OF BALTIMORE COUNTY, CATONSVILLE CAMPUS)



