5.1: Energy, Matter, and Enzymes
- Page ID
- 42512
Learning Objectives
- Define and describe metabolism
- Compare and contrast autotrophs and heterotrophs
- Describe the importance of oxidation-reduction reactions in metabolism
- Describe why ATP, FAD, NAD+, and NADP+ are important in a cell
- Differentiate between substrate-level and oxidative phosphorylation
- Identify the structure and structural components of an enzyme and the roles of substrates and products in a reaction
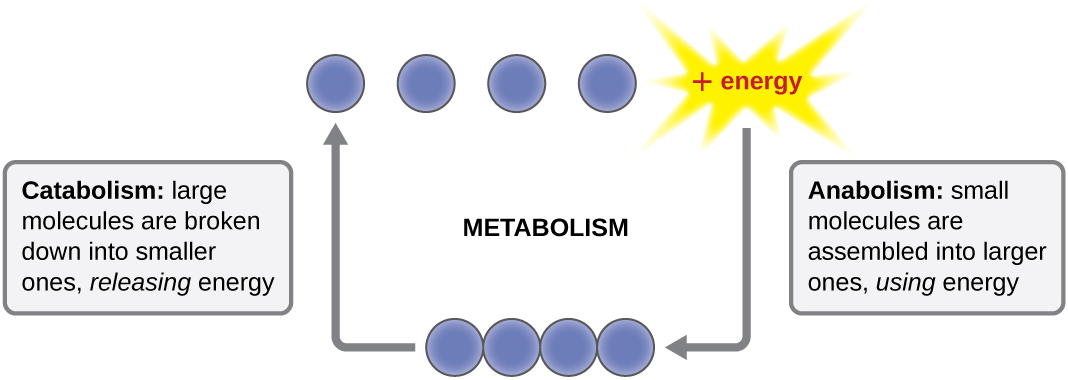
The term used to describe all of the chemical reactions inside a cell is metabolism (Figure \(\PageIndex{1}\)). Cellular processes such as the building or breaking down of complex molecules occur through series of stepwise, interconnected chemical reactions called metabolic pathways. Reactions that are spontaneous and release energy are exergonic reactions, whereas endergonic reactions require energy to proceed. The term anabolism refers to those endergonic metabolic pathways involved in biosynthesis, converting simple molecular building blocks into more complex molecules, and fueled by the use of cellular energy, usually in the form of ATP (energy) and/or NAD(P)H (electrons). Conversely, the term catabolism refers to exergonic pathways that break down complex molecules into simpler ones. Molecular energy stored in the bonds of complex molecules is released in catabolic pathways and harvested in such a way that it can be used to produce high-energy molecules (usually in ATP (energy) and/or NADH (electrons)), which are used to drive anabolic pathways. Thus, in terms of energy and molecules, cells are continually balancing catabolism with anabolism.
Oxidation and Reduction in Metabolism
The transfer of electrons between molecules is important because most of the energy stored in atoms and used to fuel cell functions is in the form of high-energy electrons. The transfer of energy in the form of electrons allows the cell to transfer and use energy incrementally; that is, in small packages rather than a single, destructive burst. Reactions that remove electrons from donor molecules, leaving them oxidized, are oxidation reactions; those that add electrons to acceptor molecules, leaving them reduced, are reduction reactions. Because electrons can move from one molecule to another, oxidation and reduction occur in tandem. These pairs of reactions are called oxidation-reduction reactions, or redox reactions.
Energy Carriers: NAD+, NADP+, FAD, and ATP
The energy released from the breakdown of the chemical bonds within nutrients can be stored either through the reduction of electron carriers or in the bonds of adenosine triphosphate (ATP). In living systems, a small class of compounds functions as mobile electron carriers, molecules that bind to and shuttle high-energy electrons between compounds in pathways. The principal electron carriers we will consider originate from the B vitamin group and are derivatives of nucleotides; they are nicotinamide adenine dinucleotide, nicotine adenine dinucleotide phosphate, and flavin adenine dinucleotide. These compounds can be easily reduced or oxidized. Nicotinamide adenine dinucleotide (NAD+/NADH) is the most common mobile electron carrier used in catabolism. NAD+ is the oxidized form of the molecule; NADH is the reduced form of the molecule. Nicotine adenine dinucleotide phosphate (NADP+), the oxidized form of an NAD+ variant that contains an extra phosphate group, is another important electron carrier; it forms NADPH when reduced. The oxidized form of flavin adenine dinucleotide is FAD, and its reduced form is FADH2. Both NAD+/NADH and FAD/FADH2 are extensively used in energy extraction from sugars during catabolism in chemoheterotrophs, whereas NADP+/NADPH plays an important role in anabolic reactions and photosynthesis. Collectively, FADH2, NADH, and NADPH are often referred to as having reducing power due to their ability to donate electrons to various chemical reactions.
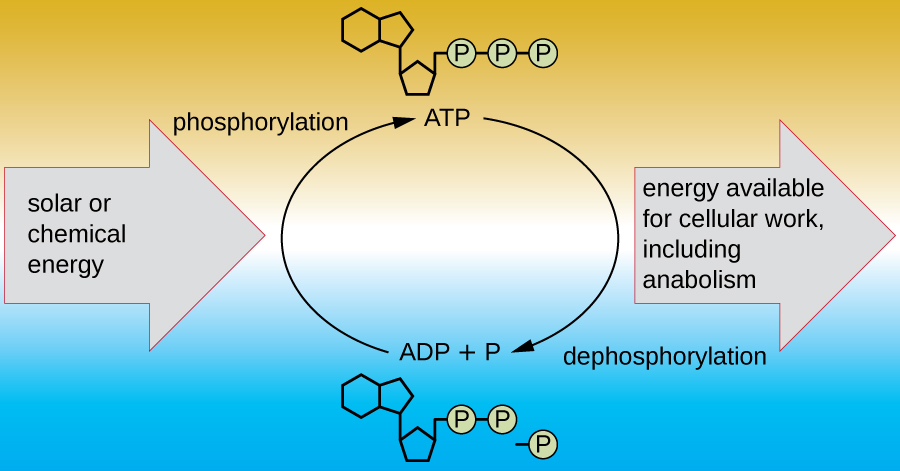
A living cell must be able to handle the energy released during catabolism in a way that enables the cell to store energy safely and release it for use only as needed. Living cells accomplish this by using the compound adenosine triphosphate (ATP). ATP is often called the “energy currency” of the cell, and, like currency, this versatile compound can be used to fill any energy need of the cell. At the heart of ATP is a molecule of adenosine monophosphate (AMP), which is composed of an adenine molecule bonded to a ribose molecule and a single phosphate group. Ribose is a five-carbon sugar found in RNA, and AMP is one of the nucleotides in RNA. The addition of a second phosphate group to this core molecule results in the formation of adenosine diphosphate (ADP); the addition of a third phosphate group forms ATP (Figure \(\PageIndex{2}\)). Adding a phosphate group to a molecule, a process called phosphorylation, requires energy. Phosphate groups are negatively charged and thus repel one another when they are arranged in series, as they are in ADP and ATP. This repulsion makes the ADP and ATP molecules inherently unstable. Thus, the bonds between phosphate groups (one in ADP and two in ATP) are called high-energy phosphate bonds. When these high-energy bonds are broken to release one phosphate (called inorganic phosphate [Pi]) or two connected phosphate groups (called pyrophosphate [PPi]) from ATP through a process called dephosphorylation, energy is released to drive endergonic reactions (Figure \(\PageIndex{3}\)).
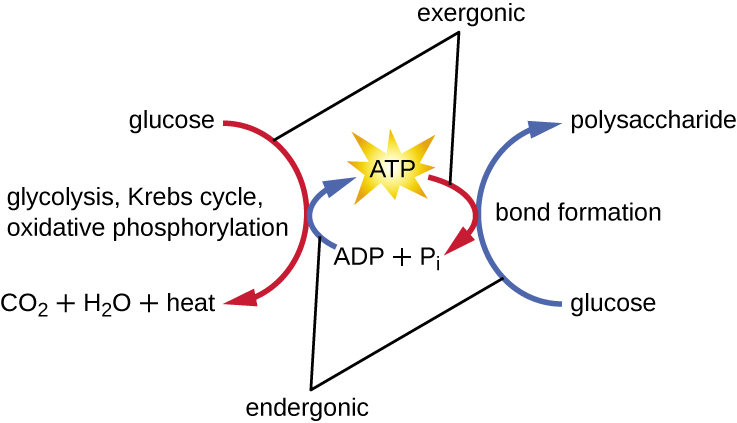
There are two general mechanisms by which ATP is formed in cells: substrate-level phosphorylation and oxidative phosphorylation (Figure \(\PageIndex{4}\)). Remember that a substrate is a molecule on which an enzyme acts. In substrate-level phosphorylation, ADP and a phosphorylated metabolite are both substrates in the same enzymatic reaction. During the exergonic enzymatic reaction, the phosphate on the metabolite is transferred to ADP to form the products ATP and an unphosphorylated metabolite. Any ATP produced in a typical metabolic pathway is through substrate-level phosphorylation.
Oxidative phosphorylation, on the other hand, is specifically the addition of inorganic phosphate to ADP by ATP synthase powered by a proton gradient across a membrane. This almost always occurs in conjunction with an electron transport chain in respiration.
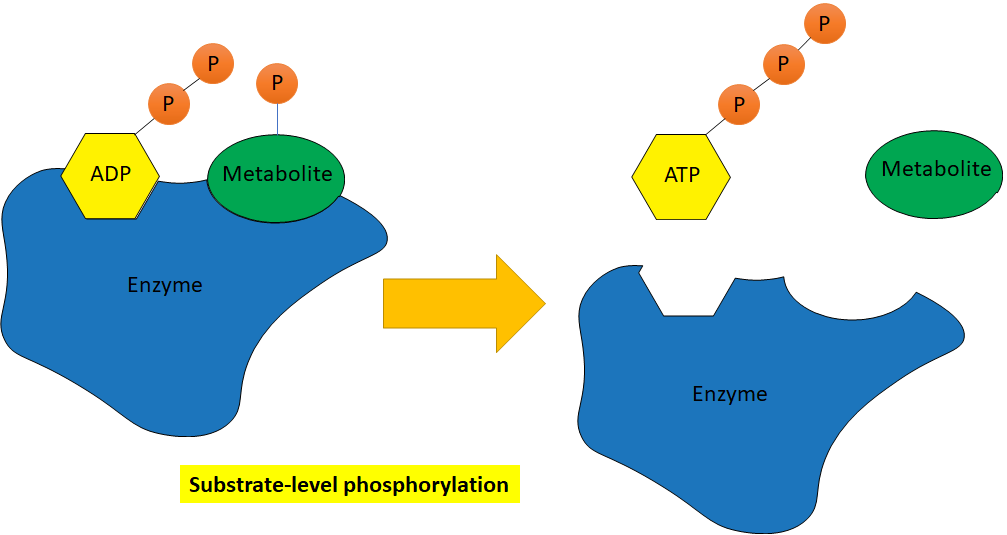
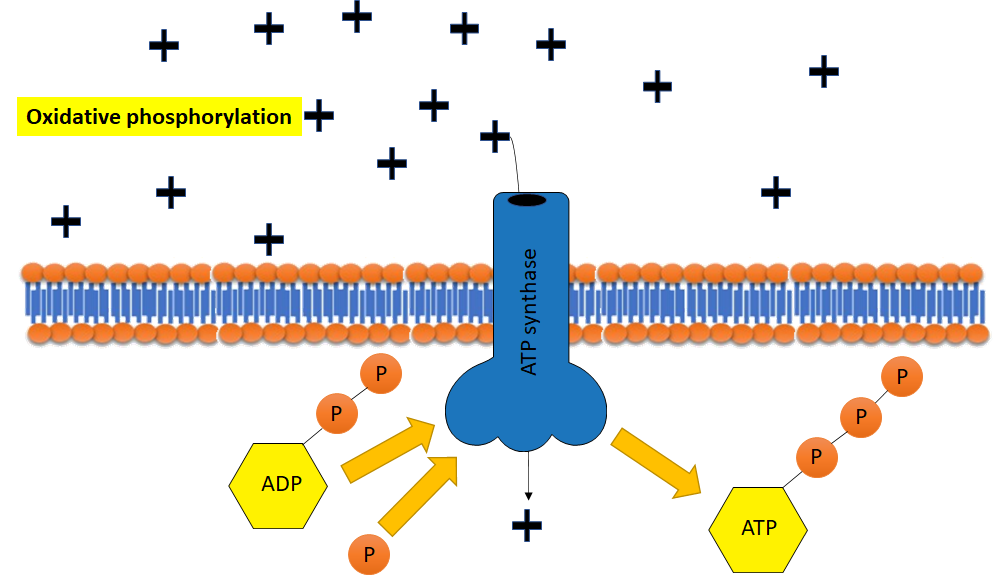
Figure \(\PageIndex{4}\): Substrate-level (top) and oxidative (bottom) phosphorylation (2021; Jeanne Kagle)
Exercise \(\PageIndex{3}\)
What is the function of an electron carrier?
Enzyme Structure and Function
A substance that helps speed up a chemical reaction is a catalyst. Catalysts are not used or changed during chemical reactions and, therefore, are reusable. Whereas inorganic molecules may serve as catalysts for a wide range of chemical reactions, proteins called enzymes serve as catalysts for biochemical reactions inside cells. Enzymes thus play an important role in controlling cellular metabolism.
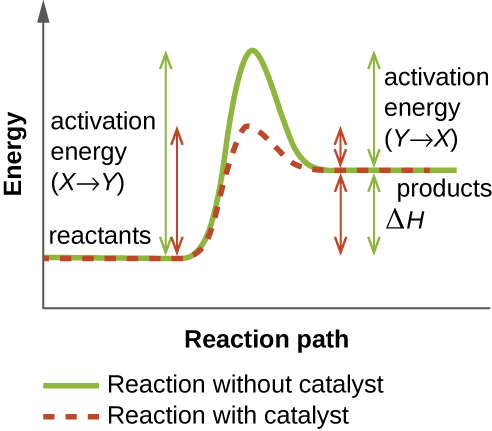
An enzyme functions by lowering the activation energy of a chemical reaction inside the cell. Activation energy is the energy needed to form or break chemical bonds and convert reactants to products (Figure \(\PageIndex{5}\)). Enzymes lower the activation energy by binding to the reactant molecules and holding them in such a way as to speed up the reaction.
The chemical reactants to which an enzyme binds are called substrates, and the location within the enzyme where the substrate binds is called the enzyme’s active site. The characteristics of the amino acids near the active site create a very specific chemical environment within the active site that induces suitability to binding, albeit briefly, to a specific substrate (or substrates). Due to this jigsaw puzzle-like match between an enzyme and its substrates, enzymes are known for their specificity. In fact, as an enzyme binds to its substrate(s), the enzyme structure changes slightly to find the best fit between the transition state (a structural intermediate between the substrate and product) and the active site, just as a rubber glove molds to a hand inserted into it. This active-site modification in the presence of substrate, along with the simultaneous formation of the transition state, is called induced fit (Figure \(\PageIndex{6}\)). Overall, there is a specifically matched enzyme for each substrate and, thus, for each chemical reaction; however, there is some flexibility as well. Some enzymes have the ability to act on several different structurally related substrates.
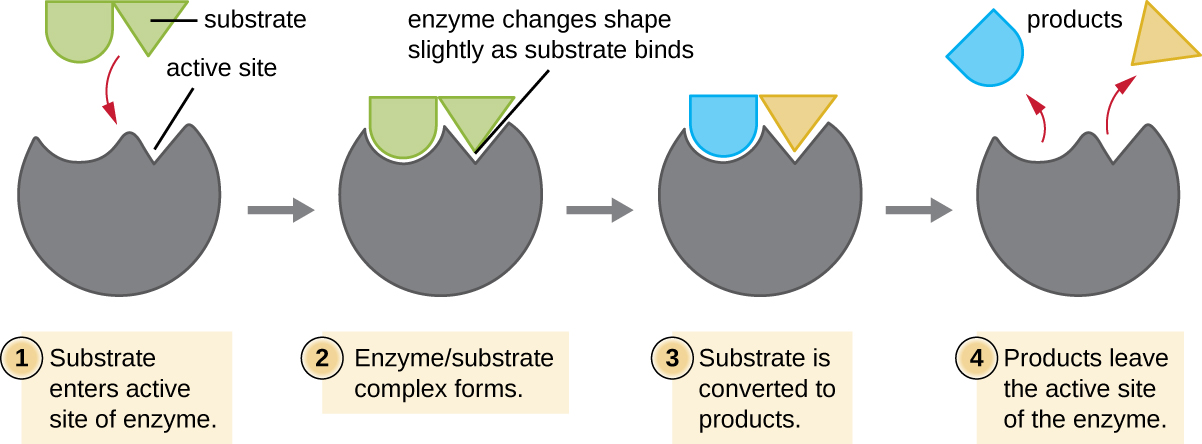
Enzymes are subject to influences by local environmental conditions such as pH, substrate concentration, and temperature. Although increasing the environmental temperature generally increases reaction rates, enzyme catalyzed or otherwise, increasing or decreasing the temperature outside of an optimal range can affect chemical bonds within the active site, making them less well suited to bind substrates. High temperatures will eventually cause enzymes, like other biological molecules, to denature, losing their three-dimensional structure and function. Enzymes are also suited to function best within a certain pH range, and, as with temperature, extreme environmental pH values (acidic or basic) can cause enzymes to denature. Active-site amino-acid side chains have their own acidic or basic properties that are optimal for catalysis and, therefore, are sensitive to changes in pH.
Another factor that influences enzyme activity is substrate concentration: Enzyme activity is increased at higher concentrations of substrate until it reaches a saturation point at which the enzyme can bind no additional substrate. Overall, enzymes are optimized to work best under the environmental conditions in which the organisms that produce them live. For example, while microbes that inhabit hot springs have enzymes that work best at high temperatures, human pathogens have enzymes that work best at 37°C. Similarly, while enzymes produced by most organisms work best at a neutral pH, microbes growing in acidic environments make enzymes optimized to low pH conditions, allowing for their growth at those conditions.
Many enzymes do not work optimally, or even at all, unless bound to other specific nonprotein helper molecules, either temporarily through ionic or hydrogen bonds or permanently through stronger covalent bonds. Binding to these molecules promotes optimal conformation and function for their respective enzymes. Two types of helper molecules are cofactors and coenzymes. Cofactors are inorganic ions such as iron (Fe2+) and magnesium (Mg2+) that help stabilize enzyme conformation and function. One example of an enzyme that requires a metal ion as a cofactor is the enzyme that builds DNA molecules, DNA polymerase, which requires a bound zinc ion (Zn2+) to function.
Coenzymes are organic helper molecules that are required for enzyme action. Like enzymes, they are not consumed and, hence, are reusable. The most common sources of coenzymes are dietary vitamins. Some vitamins are precursors to coenzymes and others act directly as coenzymes.
Some cofactors and coenzymes, like coenzyme A (CoA), often bind to the enzyme’s active site, aiding in the chemistry of the transition of a substrate to a product (Figure \(\PageIndex{7}\)). In such cases, an enzyme lacking a necessary cofactor or coenzyme is called an apoenzyme and is inactive. Conversely, an enzyme with the necessary associated cofactor or coenzyme is called a holoenzyme and is active. NADH and ATP are also both examples of commonly used coenzymes that provide high-energy electrons or phosphate groups, respectively, which bind to enzymes, thereby activating them.
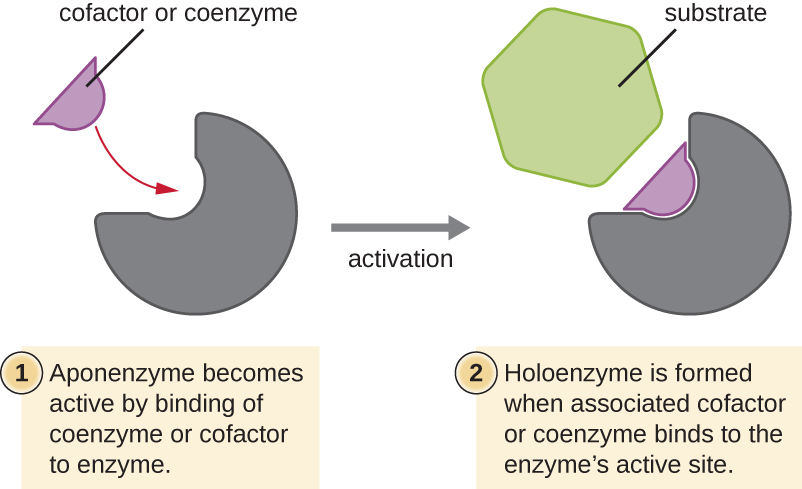
Exercise \(\PageIndex{4}\)
What role do enzymes play in a chemical reaction?
Key Concepts and Summary
- Metabolism includes chemical reactions that break down complex molecules (catabolism) and those that build complex molecules (anabolism).
- Organisms may be classified according to their source of carbon. Autotrophs convert inorganic carbon dioxide into organic carbon; heterotrophs use fixed organic carbon compounds.
- Organisms may also be classified according to their energy source. Phototrophs obtain their energy from light. Chemotrophs get their energy from chemical compounds. Organotrophs use organic molecules, and lithotrophs use inorganic chemicals.
- Cellular electron carriers accept high-energy electrons from foods and later serve as electron donors in subsequent redox reactions. FAD/FADH2, NAD+/NADH, and NADP+/NADPH are important electron carriers.
- Adenosine triphosphate (ATP) serves as the energy currency of the cell, safely storing chemical energy in its two high-energy phosphate bonds for later use to drive processes requiring energy.
- Enzymes are biological catalysts that increase the rate of chemical reactions inside cells by lowering the activation energy required for the reaction to proceed.
- In nature, exergonic reactions do not require energy beyond activation energy to proceed, and they release energy. They may proceed without enzymes, but at a slow rate. Conversely, endergonic reactions require energy beyond activation energy to occur. In cells, endergonic reactions are coupled to exergonic reactions, making the combination energetically favorable.
- Substrates bind to the enzyme’s active site. This process typically alters the structures of both the active site and the substrate, favoring transition-state formation; this is known as induced fit.
- Cofactors are inorganic ions that stabilize enzyme conformation and function. Coenzymes are organic molecules required for proper enzyme function and are often derived from vitamins. An enzyme lacking a cofactor or coenzyme is an apoenzyme; an enzyme with a bound cofactor or coenzyme is a holoenzyme.
Contributors and Attributions
Nina Parker, (Shenandoah University), Mark Schneegurt (Wichita State University), Anh-Hue Thi Tu (Georgia Southwestern State University), Philip Lister (Central New Mexico Community College), and Brian M. Forster (Saint Joseph’s University) with many contributing authors. Original content via Openstax (CC BY 4.0; Access for free at https://openstax.org/books/microbiology/pages/1-introduction)


