1.6: Molecules of Life
- Page ID
- 93951
\( \newcommand{\vecs}[1]{\overset { \scriptstyle \rightharpoonup} {\mathbf{#1}} } \)
\( \newcommand{\vecd}[1]{\overset{-\!-\!\rightharpoonup}{\vphantom{a}\smash {#1}}} \)
\( \newcommand{\dsum}{\displaystyle\sum\limits} \)
\( \newcommand{\dint}{\displaystyle\int\limits} \)
\( \newcommand{\dlim}{\displaystyle\lim\limits} \)
\( \newcommand{\id}{\mathrm{id}}\) \( \newcommand{\Span}{\mathrm{span}}\)
( \newcommand{\kernel}{\mathrm{null}\,}\) \( \newcommand{\range}{\mathrm{range}\,}\)
\( \newcommand{\RealPart}{\mathrm{Re}}\) \( \newcommand{\ImaginaryPart}{\mathrm{Im}}\)
\( \newcommand{\Argument}{\mathrm{Arg}}\) \( \newcommand{\norm}[1]{\| #1 \|}\)
\( \newcommand{\inner}[2]{\langle #1, #2 \rangle}\)
\( \newcommand{\Span}{\mathrm{span}}\)
\( \newcommand{\id}{\mathrm{id}}\)
\( \newcommand{\Span}{\mathrm{span}}\)
\( \newcommand{\kernel}{\mathrm{null}\,}\)
\( \newcommand{\range}{\mathrm{range}\,}\)
\( \newcommand{\RealPart}{\mathrm{Re}}\)
\( \newcommand{\ImaginaryPart}{\mathrm{Im}}\)
\( \newcommand{\Argument}{\mathrm{Arg}}\)
\( \newcommand{\norm}[1]{\| #1 \|}\)
\( \newcommand{\inner}[2]{\langle #1, #2 \rangle}\)
\( \newcommand{\Span}{\mathrm{span}}\) \( \newcommand{\AA}{\unicode[.8,0]{x212B}}\)
\( \newcommand{\vectorA}[1]{\vec{#1}} % arrow\)
\( \newcommand{\vectorAt}[1]{\vec{\text{#1}}} % arrow\)
\( \newcommand{\vectorB}[1]{\overset { \scriptstyle \rightharpoonup} {\mathbf{#1}} } \)
\( \newcommand{\vectorC}[1]{\textbf{#1}} \)
\( \newcommand{\vectorD}[1]{\overrightarrow{#1}} \)
\( \newcommand{\vectorDt}[1]{\overrightarrow{\text{#1}}} \)
\( \newcommand{\vectE}[1]{\overset{-\!-\!\rightharpoonup}{\vphantom{a}\smash{\mathbf {#1}}}} \)
\( \newcommand{\vecs}[1]{\overset { \scriptstyle \rightharpoonup} {\mathbf{#1}} } \)
\(\newcommand{\longvect}{\overrightarrow}\)
\( \newcommand{\vecd}[1]{\overset{-\!-\!\rightharpoonup}{\vphantom{a}\smash {#1}}} \)
\(\newcommand{\avec}{\mathbf a}\) \(\newcommand{\bvec}{\mathbf b}\) \(\newcommand{\cvec}{\mathbf c}\) \(\newcommand{\dvec}{\mathbf d}\) \(\newcommand{\dtil}{\widetilde{\mathbf d}}\) \(\newcommand{\evec}{\mathbf e}\) \(\newcommand{\fvec}{\mathbf f}\) \(\newcommand{\nvec}{\mathbf n}\) \(\newcommand{\pvec}{\mathbf p}\) \(\newcommand{\qvec}{\mathbf q}\) \(\newcommand{\svec}{\mathbf s}\) \(\newcommand{\tvec}{\mathbf t}\) \(\newcommand{\uvec}{\mathbf u}\) \(\newcommand{\vvec}{\mathbf v}\) \(\newcommand{\wvec}{\mathbf w}\) \(\newcommand{\xvec}{\mathbf x}\) \(\newcommand{\yvec}{\mathbf y}\) \(\newcommand{\zvec}{\mathbf z}\) \(\newcommand{\rvec}{\mathbf r}\) \(\newcommand{\mvec}{\mathbf m}\) \(\newcommand{\zerovec}{\mathbf 0}\) \(\newcommand{\onevec}{\mathbf 1}\) \(\newcommand{\real}{\mathbb R}\) \(\newcommand{\twovec}[2]{\left[\begin{array}{r}#1 \\ #2 \end{array}\right]}\) \(\newcommand{\ctwovec}[2]{\left[\begin{array}{c}#1 \\ #2 \end{array}\right]}\) \(\newcommand{\threevec}[3]{\left[\begin{array}{r}#1 \\ #2 \\ #3 \end{array}\right]}\) \(\newcommand{\cthreevec}[3]{\left[\begin{array}{c}#1 \\ #2 \\ #3 \end{array}\right]}\) \(\newcommand{\fourvec}[4]{\left[\begin{array}{r}#1 \\ #2 \\ #3 \\ #4 \end{array}\right]}\) \(\newcommand{\cfourvec}[4]{\left[\begin{array}{c}#1 \\ #2 \\ #3 \\ #4 \end{array}\right]}\) \(\newcommand{\fivevec}[5]{\left[\begin{array}{r}#1 \\ #2 \\ #3 \\ #4 \\ #5 \\ \end{array}\right]}\) \(\newcommand{\cfivevec}[5]{\left[\begin{array}{c}#1 \\ #2 \\ #3 \\ #4 \\ #5 \\ \end{array}\right]}\) \(\newcommand{\mattwo}[4]{\left[\begin{array}{rr}#1 \amp #2 \\ #3 \amp #4 \\ \end{array}\right]}\) \(\newcommand{\laspan}[1]{\text{Span}\{#1\}}\) \(\newcommand{\bcal}{\cal B}\) \(\newcommand{\ccal}{\cal C}\) \(\newcommand{\scal}{\cal S}\) \(\newcommand{\wcal}{\cal W}\) \(\newcommand{\ecal}{\cal E}\) \(\newcommand{\coords}[2]{\left\{#1\right\}_{#2}}\) \(\newcommand{\gray}[1]{\color{gray}{#1}}\) \(\newcommand{\lgray}[1]{\color{lightgray}{#1}}\) \(\newcommand{\rank}{\operatorname{rank}}\) \(\newcommand{\row}{\text{Row}}\) \(\newcommand{\col}{\text{Col}}\) \(\renewcommand{\row}{\text{Row}}\) \(\newcommand{\nul}{\text{Nul}}\) \(\newcommand{\var}{\text{Var}}\) \(\newcommand{\corr}{\text{corr}}\) \(\newcommand{\len}[1]{\left|#1\right|}\) \(\newcommand{\bbar}{\overline{\bvec}}\) \(\newcommand{\bhat}{\widehat{\bvec}}\) \(\newcommand{\bperp}{\bvec^\perp}\) \(\newcommand{\xhat}{\widehat{\xvec}}\) \(\newcommand{\vhat}{\widehat{\vvec}}\) \(\newcommand{\uhat}{\widehat{\uvec}}\) \(\newcommand{\what}{\widehat{\wvec}}\) \(\newcommand{\Sighat}{\widehat{\Sigma}}\) \(\newcommand{\lt}{<}\) \(\newcommand{\gt}{>}\) \(\newcommand{\amp}{&}\) \(\definecolor{fillinmathshade}{gray}{0.9}\)- Identify that all matter is composed of atoms and that atoms build molecules.
- Define organic molecules and inorganic molecules and be able to identify whether a molecule is organic or inorganic based on its chemical structure.
- List the four types of biological molecules, their monomers, and their functions.
- Define dehydration synthesis and hydrolysis and describe the importance of these reactions for biological molecules.
- Describe a burn test and interpret results of a burn test.
- Successfully build molecular molecules biological molecule monomers and conduct dehydration synthesis and hydrolysis reactions with these models.
- Test food items for a significant presence of proteins, carbohydrates, and lipids.
Atoms & Molecules
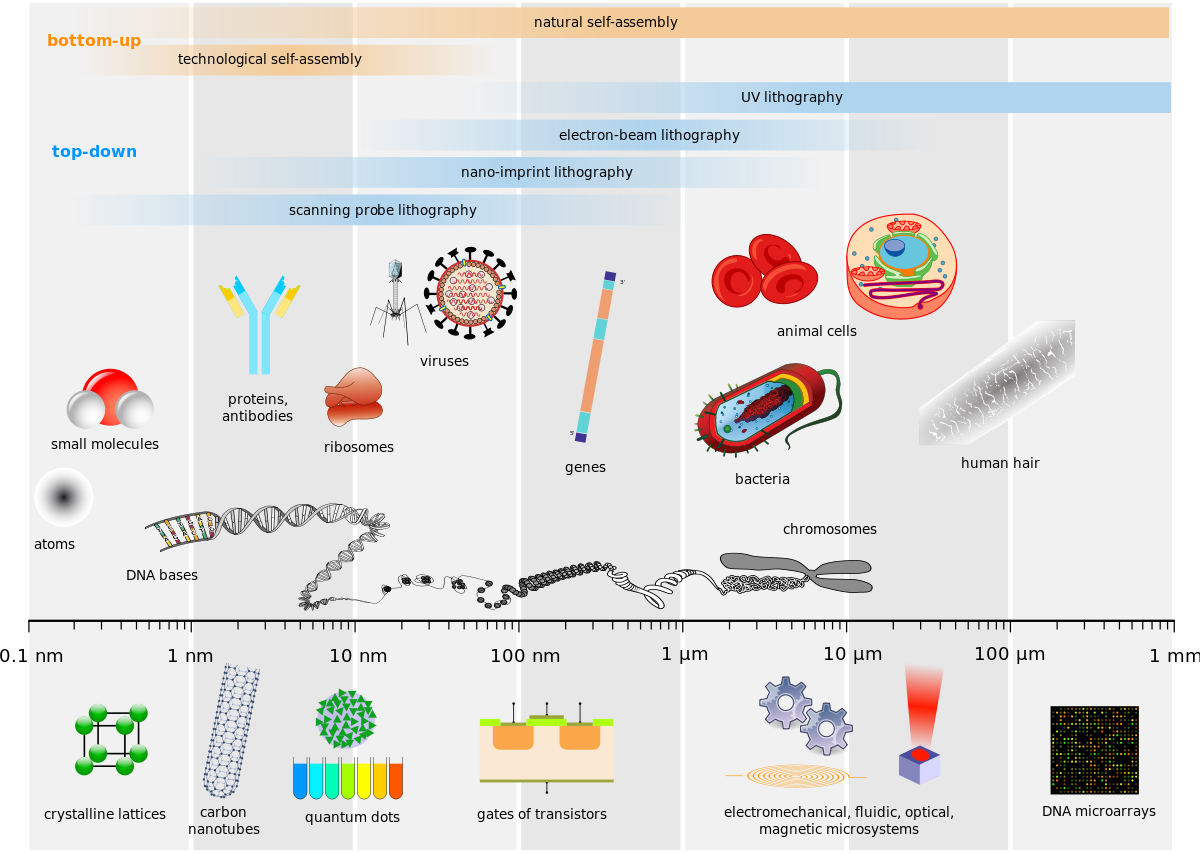
Figure 1: Anything in the universe that has physical substance is composed of atoms. Molecules are built from atoms. The molecules of life (biological molecules) are usually larger molecules (e.g. DNA, proteins). Biological molecules are the fundamental structures that make up living things (e.g. bacteria, animal cells) and non-living biological particles (e.g. viruses).
All matter (anything with physical substance) is made atoms; this includes all living things from microorganisms to humans to blue whales. Atoms, composed of protons, neutrons, and electrons, are the smallest structures that makes up elements of the periodic table. Therefore, the periodic table lists different types of atoms (also known as elements). Each type of element is different because the number of protons it has (the number of protons is the same as the atomic number).
*microbes in this interactive tool include: amoeba proteus, paramecium, baker's yeast, E. coli bacterium, measles virus, hiv, phage, influenza virus, hepatitis virus, and rhinovirus
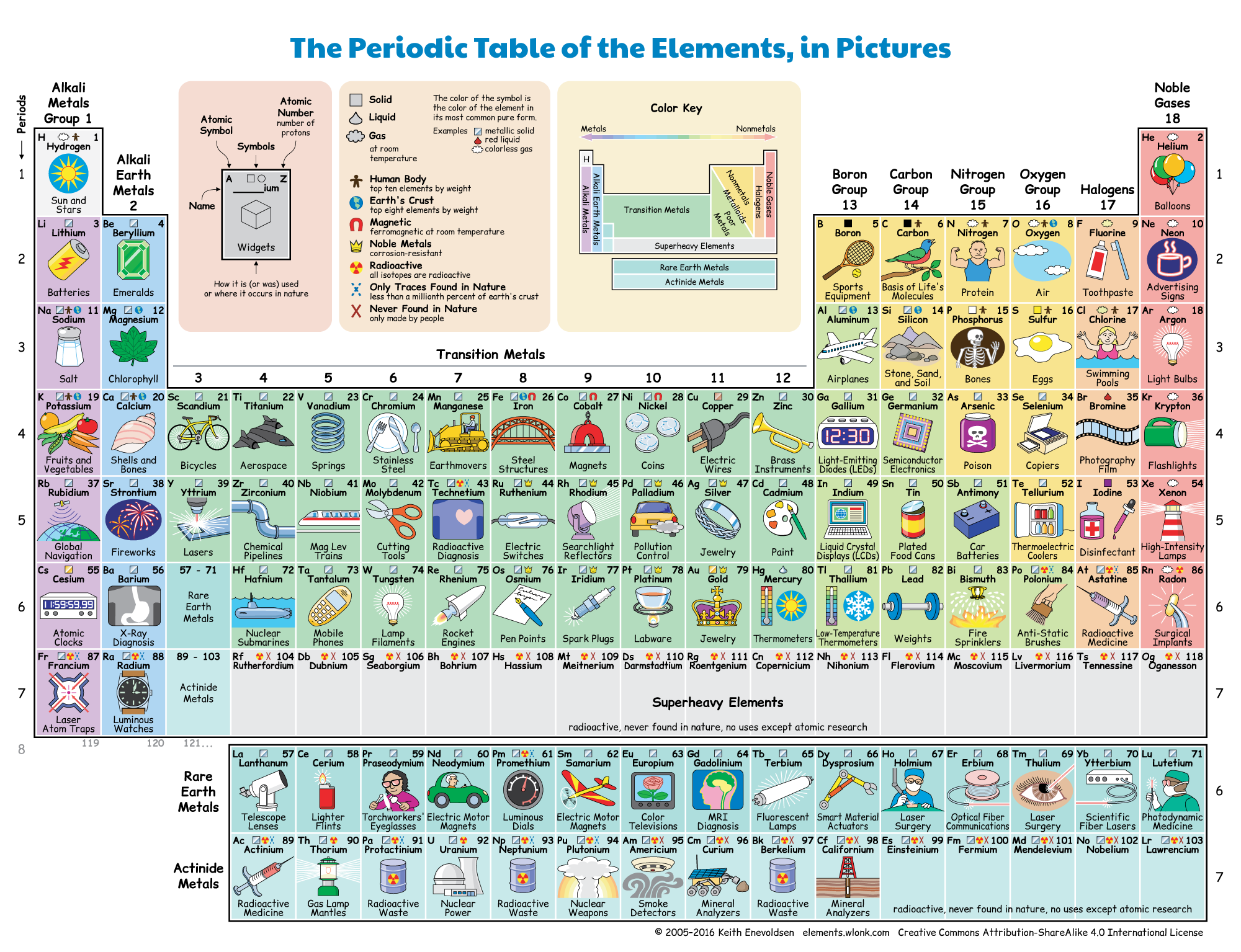
Figure 2: The periodic table of elements. Each element is a different type of atom that is defined based on the number of protons within that atom (the atomic number). For example, the atomic number of carbon (C) is 6. Carbon has six protons. Carbon will always have six protons. If you have an element that does not have six protons, it is not carbon.
Organic vs. Inorganic Molecules
All molecules can be classified as either organic or inorganic. Organic molecules are types of molecules typically found in living things and molecules that came from living things. In contrast, inorganic molecules are considered to be molecules not produced by living things. While organic molecules are considered the molecules of life/biological molecules, inorganic molecules can still be necessary for life. For example, water (H2O) is a molecule that is required by all living things, yet it is considered inorganic. A straight-forward way to tell determine if a molecule is organic or inorganic is based on the types of elements it has:
- organic molecules always contain carbon atoms (C) and hydrogen atoms (H).
- inorganic molecules can contain carbon atoms (C) and can contain hydrogen atoms (H), but inorganic molecules will not contain both carbon atoms (C) and hydrogen atoms (H).
Inorganic molecules usually do not contain carbon atoms (C). Also, organic molecules are formed only by covalent bonds, whereas inorganic molecules can be formed by covalent bonds or ionic bonds.
What if we have a sample of a substance (and perhaps do not know the elements it contains) and would like to determine if the substance is organic or inorganic?

A simple laboratory test that can determine if a sample is organic or inorganic is called the burn test. This test is very simple: place the substance over heat - if it burns/turns black, it is likely organic. If it does not burn/does not turn black, it is likely inorganic.
Molecules that Make up Living Things
Biological molecules are classified into four types:
- proteins
- carbohydrates
- lipids
- nucleic acids
Each category of biological molecule has distinct features, including their building blocks and the types of functions they can serve. Biological molecules tend to be very large and are built of repeating units of smaller molecules. These small subunits that make up larger biological molecules are called monomers. The monomers of proteins are called amino acids, the monomers of carbohydrates are called monosaccharides, the monomers of lipids are called fatty acids, and the monomers of nucleic acids are called nucleotides.
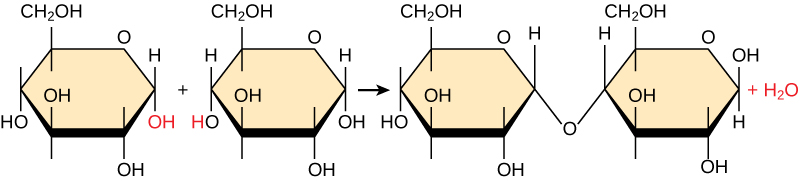
Figure 3: In the dehydration synthesis reaction depicted above, two molecules of glucose are linked together to form the disaccharide maltose. In the process, a water molecule is formed. The arrow represents the chemical reaction. At the butt end of the arrow are the reactants. Notice that there are two separate molecules that are not touching each other on the reactant side. These are two small monomers. On the pointy end of the arrow are the products. Notice that there is one larger molecule on the product side. The reaction changed two monomers into a dimer (two monomers bonded together).
Monomers are joined together by covalent bonds through dehydration synthesis reactions to form larger biological molecules made of multiple monomers. The number of monomers bonded together is apparent in the generalized naming scheme for biological molecules:
| number of monomers bonded together | commonly used prefix | protein name | carbohydrate name | lipid name | nucleic acid name |
|---|---|---|---|---|---|
|
1 (no bonds between monomers) |
mono- | amino acid | monosaccharide | fatty acid | nucleotide |
| 2 | di- | dipeptide | disaccharide | diglyceride | dinucleotide |
| 3 | tri- | tripeptide | trisaccharide | triglyceride | trinucleotide |
| "a few" (2, 3, or 4) | oligo- | oligopeptide | oligosaccharide | n/a | oligonucleotide |
|
many (could be hundreds or more) |
poly- | polypeptide | polysaccharide | n/a | polynucleotide |
Lipids are limited to triglycerides and cannot get bigger. This is due to the glycerol structure fatty acids bond with that can only attach to three fatty acids. There are other types of lipids that do not fit this model, such as steroid molecules like cholesterol.

Figure 4: In the hydrolysis reaction shown here, the disaccharide maltose is broken down to form two glucose monomers with the addition of a water molecule. Note that this reaction is the reverse of the synthesis reaction shown in Figure 3.
Polymers and other monomers bonded together can also be broken down into smaller pieces by breaking covalent bonds through hydrolysis reactions.
Proteins
Proteins are one of the most abundant organic molecules in living systems and have the most diverse range of functions of all macromolecules. Proteins may be structural, regulatory, contractile, or protective; they may serve in transport, storage, or membranes; or they may be toxins or enzymes. Each cell in a living system may contain thousands of proteins, each with a unique function. Their structures, like their functions, vary greatly. They are all, however, polymers of amino acids, arranged in a linear sequence.
| Type | Examples | Functions |
|---|---|---|
| digestive enzymes | amylase, lipase, pepsin, trypsin | Help in digestion of food by catabolizing nutrients into monomeric units |
| transport | hemoglobin, albumin | Carry substances in the blood or lymph throughout the body |
| structural | actin, tubulin, keratin | Construct different structures, like the cytoskeleton |
| hormones | insulin, thyroxine | Coordinate the activity of different body systems |
| defense | immunoglobulins | Protect the body from foreign pathogens |
| contractile | actin, myosin | Effect muscle contraction |
| storage | legume storage proteins, egg white (albumin) | Provide nourishment in early development of the embryo and the seedling |
Proteins have different shapes and molecular weights; some proteins are globular in shape whereas others are fibrous in nature. For example, hemoglobin is a globular protein, but collagen, found in our skin, is a fibrous protein. Protein shape is critical to its function, and this shape is maintained by many different types of chemical bonds. Changes in temperature, pH, and exposure to chemicals may lead to permanent changes in the shape of the protein, leading to loss of function, known as denaturation. All proteins are made up of different arrangements of the same 20 types of amino acids.
Amino acids are the monomers that make up proteins. Each amino acid has the same fundamental structure, which consists of a central carbon atom, also known as the alpha (α) carbon, bonded to an amino group (NH2), a carboxyl group (COOH), and to a hydrogen atom. Every amino acid also has another atom or group of atoms bonded to the central atom known as the R group.
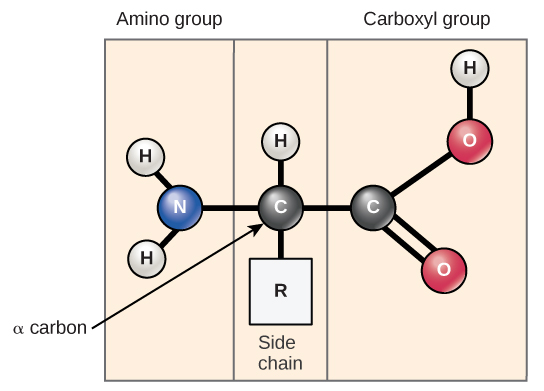
Figure 5: Amino acids have a central asymmetric carbon to which an amino group, a carboxyl group, a hydrogen atom, and a side chain (R group) are attached.
The name "amino acid" is derived from the fact that they contain both amino group (NH2) and carboxylic acid group (COOH) in their basic structure. As mentioned, there are 20 amino acids present in proteins. Ten of these are considered essential amino acids in humans because the human body cannot produce them and they are obtained from the diet. For each amino acid, the R group (or side chain) is different.
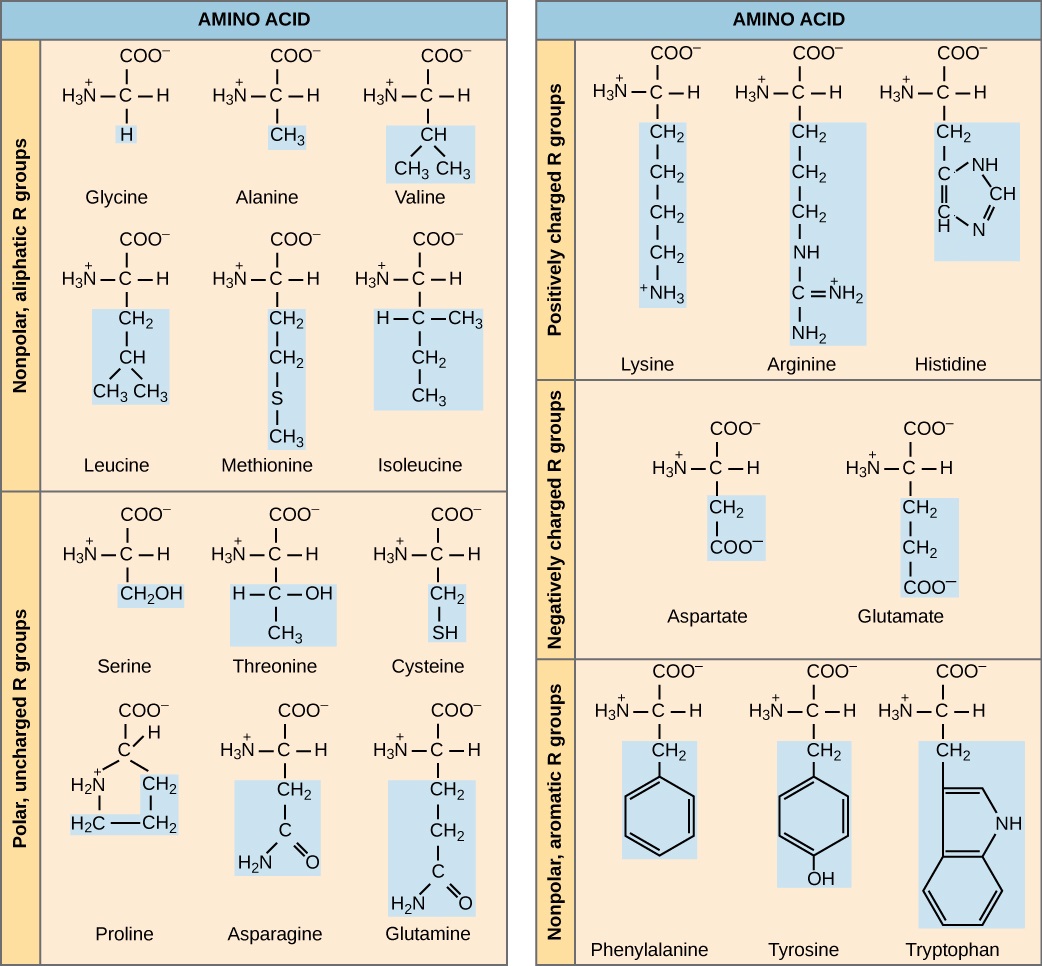
Figure 6: There are 20 common amino acids commonly found in proteins, each with a different R group (variant group) that determines its chemical nature.
The chemical nature of the side chain determines the nature of the amino acid (that is, whether it is acidic, basic, polar, or nonpolar). For example, the amino acid glycine has a hydrogen atom as the R group. Amino acids such as valine, methionine, and alanine are nonpolar or hydrophobic in nature, while amino acids such as serine, threonine, and cysteine are polar and have hydrophilic side chains. The side chains of lysine and arginine are positively charged, and therefore these amino acids are also known as basic amino acids. Proline has an R group that is linked to the amino group, forming a ring-like structure. Proline is an exception to the standard structure of an animo acid since its amino group is not separate from the side chain.
The sequence and the number of amino acids ultimately determine the protein's shape, size, and function. Each amino acid is attached to another amino acid by a covalent bond, known as a peptide bond, which is formed by a dehydration reaction. The carboxyl group of one amino acid and the amino group of the incoming amino acid combine, releasing a molecule of water. The resulting bond is the peptide bond.

Figure 7: Peptide bond formation is a dehydration synthesis reaction. The carboxyl group of one amino acid is linked to the amino group of the incoming amino acid. In the process, a molecule of water is released.
The products formed by such linkages are called peptides. As more amino acids join to this growing chain, the resulting chain is known as a polypeptide. Each polypeptide has a free amino group at one end. This end is called the N terminal, or the amino terminal, and the other end has a free carboxyl group, also known as the C or carboxyl terminal. While the terms polypeptide and protein are sometimes used interchangeably, a polypeptide is technically a polymer of amino acids, whereas the term protein is used for a polypeptide or polypeptides that have combined together, often have bound non-peptide prosthetic groups, have a distinct shape, and have a unique function. After protein synthesis (translation), most proteins are modified. These are known as post-translational modifications. They may undergo cleavage, phosphorylation, or may require the addition of other chemical groups. Only after these modifications is the protein completely functional.
Carbohydrates
Carbohydrates are a group of macromolecules that are a vital energy source for the cell and provide structural support
Most people are familiar with carbohydrates, one type of macromolecule, especially when it comes to what we eat. To lose weight, some individuals adhere to “low-carb” diets. Athletes, in contrast, often “carb-load” before important competitions to ensure that they have enough energy to compete at a high level. Carbohydrates are, in fact, an essential part of our diet; grains, fruits, and vegetables are all natural sources of carbohydrates. Carbohydrates provide energy to the body, particularly through glucose, a simple sugar that is a component of starch and an ingredient in many staple foods. Carbohydrates also have other important functions in humans, animals, and plants.
Carbohydrates can be represented by the stoichiometric formula (CH2O)n, where n is the number of carbons in the molecule. In other words, the ratio of carbon to hydrogen to oxygen is 1:2:1 in carbohydrate molecules. This formula also explains the origin of the term “carbohydrate”: the components are carbon (“carbo”) and the components of water (hence, “hydrate”). Carbohydrates are classified into three subtypes: monosaccharides, disaccharides, and polysaccharides.
Monosaccharides (mono- = “one”; sacchar- = “sweet”) are simple sugars, the most common of which is glucose. In monosaccharides, the number of carbons usually ranges from three to seven. Most monosaccharide names end with the suffix -ose. If the sugar has an aldehyde group (the functional group with the structure R-CHO), it is known as an aldose, and if it has a ketone group (the functional group with the structure RC(=O)R'), it is known as a ketose. Depending on the number of carbons in the sugar, they also may be known as trioses (three carbons), pentoses (five carbons), and or hexoses (six carbons).
Disaccharides (di- = “two”) form when two monosaccharides undergo a dehydration reaction (also known as a condensation reaction or dehydration synthesis). During this process, the hydroxyl group of one monosaccharide combines with the hydrogen of another monosaccharide, releasing a molecule of water and forming a covalent bond. A covalent bond formed between a carbohydrate molecule and another molecule (in this case, between two monosaccharides) is known as a glycosidic bond. Glycosidic bonds (also called glycosidic linkages) can be of the alpha or the beta type.

Figure 8: Sucrose is formed when a monomer of glucose and a monomer of fructose are joined in a dehydration reaction to form a glycosidic bond. In the process, a water molecule is lost. By convention, the carbon atoms in a monosaccharide are numbered from the terminal carbon closest to the carbonyl group. In sucrose, a glycosidic linkage is formed between carbon 1 in glucose and carbon 2 in fructose.
Common disaccharides include lactose, maltose, and sucrose. Lactose is a disaccharide consisting of the monomers glucose and galactose. It is found naturally in milk. Maltose, or malt sugar, is a disaccharide formed by a dehydration reaction between two glucose molecules. The most common disaccharide is sucrose, or table sugar, which is composed of the monomers glucose and fructose.
A long chain of monosaccharides linked by glycosidic bonds is known as a polysaccharide (poly- = “many”). The chain may be branched or unbranched, and it may contain different types of monosaccharides. The molecular weight may be 100,000 daltons or more depending on the number of monomers joined. Starch, glycogen, cellulose, and chitin are primary examples of polysaccharides.
Lipids
Lipids include a diverse group of compounds that are largely nonpolar in nature. This is because they are hydrocarbons that include mostly nonpolar carbon–carbon or carbon–hydrogen bonds. Non-polar molecules are hydrophobic (“water fearing”), or insoluble in water. Lipids perform many different functions in a cell. Cells store energy for long-term use in the form of fats. Lipids are also the building blocks of many hormones and are an important constituent of all cellular membranes. Lipids include fats, oils, waxes, phospholipids, and steroids.
A fat molecule consists of two main components—glycerol and fatty acids. Glycerol is an organic compound (alcohol) with three carbons, five hydrogens, and three hydroxyl (OH) groups. Fatty acids have a long chain of hydrocarbons to which a carboxyl group is attached, hence the name “fatty acid.” The number of carbons in the fatty acid may range from 4 to 36; most common are those containing 12–18 carbons. In a fat molecule, the fatty acids are attached to each of the three carbons of the glycerol molecule with an ester bond through an oxygen atom.

Figure 9: Triacylglycerol (also known as a triglyceride) is formed by the joining of three fatty acids to a glycerol backbone in a dehydration reaction. Three molecules of water are released in the process.
Phospholipids are major constituents of the plasma membrane, the outermost layer of animal cells. Like fats, they are composed of fatty acid chains attached to a glycerol or sphingosine backbone. Instead of three fatty acids attached as in triglycerides, however, there are two fatty acids forming diacylglycerol, and the third carbon of the glycerol backbone is occupied by a modified phosphate group. A phosphate group alone attached to a diaglycerol does not qualify as a phospholipid; it is phosphatidate (diacylglycerol 3-phosphate), the precursor of phospholipids. The phosphate group is modified by an alcohol. Phosphatidylcholine and phosphatidylserine are two important phospholipids that are found in plasma membranes.
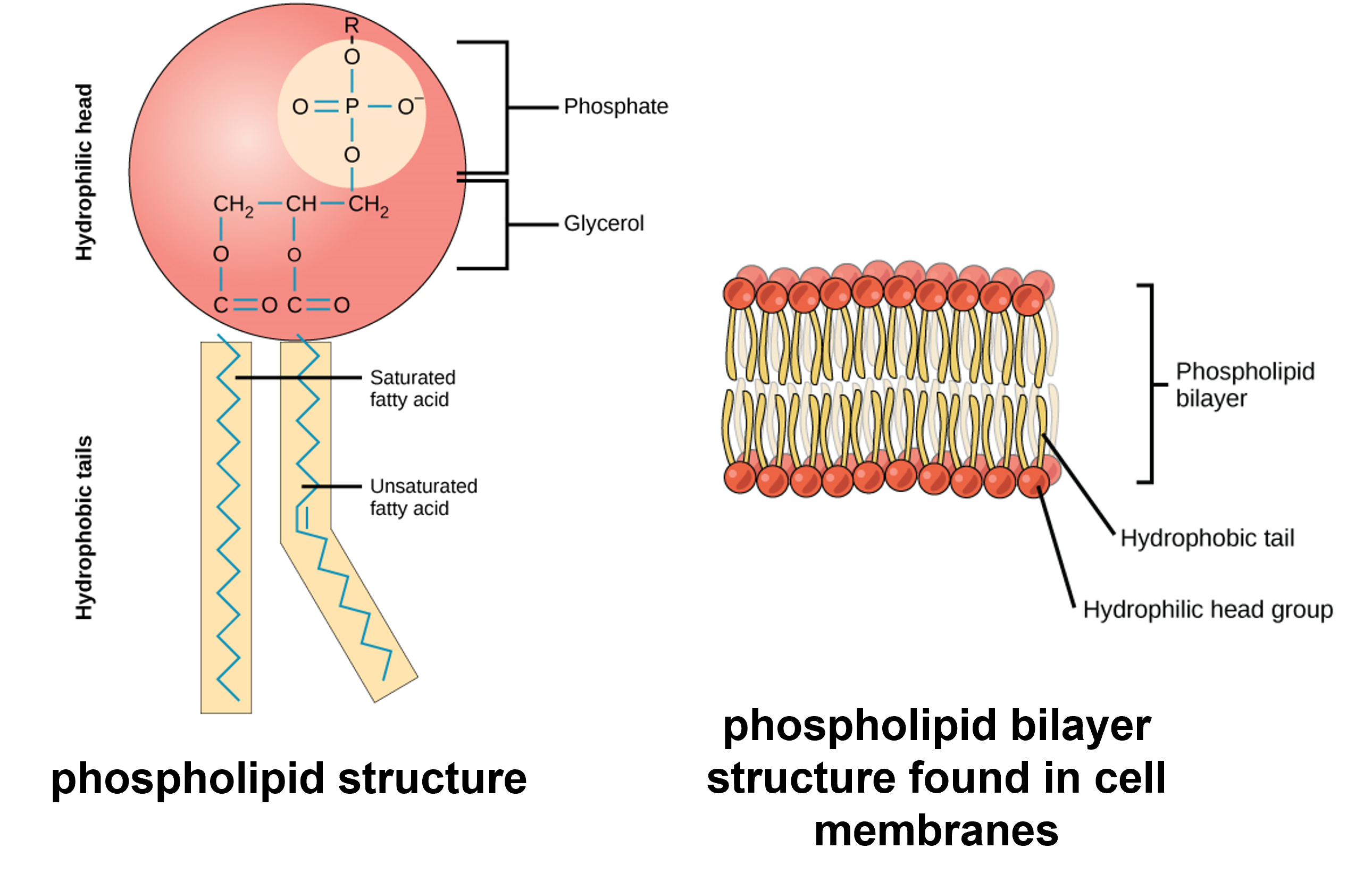
Figure 10: (Left) A phospholipid is a molecule with two fatty acids and a modified phosphate group attached to a glycerol backbone. The phosphate may be modified by the addition of charged or polar chemical groups. Two chemical groups that may modify the phosphate, choline and serine, are shown here. Both choline and serine attach to the phosphate group at the position labeled R via the hydroxyl group indicated in green. (Right) The phospholipid bilayer is the major component of all cellular membranes. The hydrophilic head groups of the phospholipids face the aqueous solution. The hydrophobic tails are sequestered in the middle of the bilayer.
Unlike the phospholipids and fats discussed earlier, steroids have a fused ring structure. Although they do not resemble the other lipids, they are grouped with them because they are also hydrophobic and insoluble in water. All steroids have four linked carbon rings and several of them, like cholesterol, have a short tail.
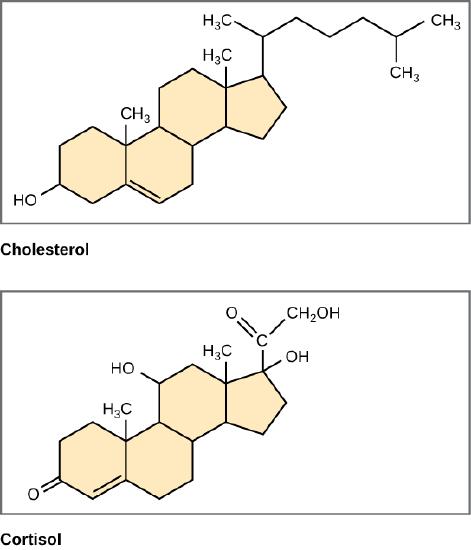
Figure 11: Steroids such as cholesterol and cortisol are composed of four fused hydrocarbon rings.
Nucleic Acids
Nucleic acids are the most important macromolecules for the continuity of life. They carry the genetic blueprint of a cell and carry instructions for the functioning of the cell.
The two main types of nucleic acids are deoxyribonucleic acid (DNA) and ribonucleic acid (RNA). DNA is the genetic material found in all living organisms, ranging from single-celled bacteria to multicellular mammals. It is found in the nucleus of eukaryotes and in the organelles, chloroplasts, and mitochondria. In prokaryotes, the DNA is not enclosed in a membranous envelope.
DNA and RNA are made up of monomers known as nucleotides. The nucleotides combine with each other to form a polynucleotide, DNA or RNA. Each nucleotide is made up of three components: a nitrogenous base, a pentose (five-carbon) sugar, and a phosphate group. Each nitrogenous base in a nucleotide is attached to a sugar molecule, which is attached to one or more phosphate groups.

Figure 12: A nucleotide is made up of three components: a nitrogenous base, a pentose sugar, and one or more phosphate groups. Carbon residues in the pentose are numbered 1′ through 5′ (the prime distinguishes these residues from those in the base, which are numbered without using a prime notation). The base is attached to the 1′ position of the ribose, and the phosphate is attached to the 5′ position. When a polynucleotide is formed, the 5′ phosphate of the incoming nucleotide attaches to the 3′ hydroxyl group at the end of the growing chain. Two types of pentose are found in nucleotides, deoxyribose (found in DNA) and ribose (found in RNA). Deoxyribose is similar in structure to ribose, but it has an H instead of an OH at the 2′ position. Bases can be divided into two categories: purines and pyrimidines. Purines have a double ring structure, and pyrimidines have a single ring.
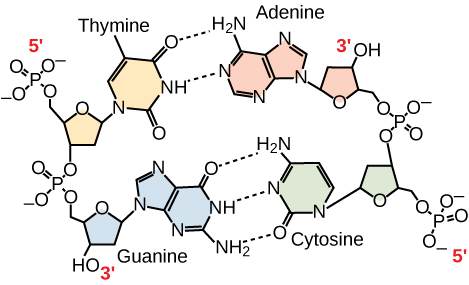
Figure 13: In a double stranded DNA molecule, the two strands run antiparallel to one another so that one strand runs 5′ to 3′ and the other 3′ to 5′. The phosphate backbone is located on the outside, and the bases are in the middle. Adenine forms hydrogen bonds (or base pairs) with thymine, and guanine base pairs with cytosine.
Overview Video of Biological Molecules
Laboratory Instructions
Burn Test
Conduct the Burn Test
- Put safety goggles on.
- Put a small amount of baking soda into a test tube.
- Hold the test tube with a test tube holder.
- Ignite a Bunsen burner.
- Angle the test tube so the open end is not facing yourself or any other person. Place the bottom of the test tube into the flame to heat the baking soda.
- Continue burning until the baking soda is clearly very hot and smoking.
- Observe the baking soda color.
- Write results in the table below.
- Repeat steps 2-8 for each of the following substances (with new test tubes each time): hair, salt, starch, sugar, vegetable oil, water.
Alternatively, your instructor may have completed these burn tests for you. Observe the results and record the results in the table below.
Burn Test Results & Questions
| Substance Tested in the Burn Test | Appearance of Substance after Burn Test | Is the Substance Organic or Inorganic? | If Substance is Organic, Give the Type of Biological Molecule (protein, carbohydrate, lipid, or nucleic acid) |
|---|---|---|---|
| baking soda | |||
| hair | |||
| salt | |||
| starch | |||
| sugar | |||
| vegetable oil | |||
| water |
- Complete the table above based on results of the burn test.
- What type(s) of biological molecules were not tested using the burn test? Give examples of these types of biological molecules.
- True or False: Every type of molecule found within a living thing is organic. Explain your answer.
- True or False: It is possible for inorganic molecules to be essential for life.
- What is the burn test and what can it show us?
Building Monomers of Biological Molecules
To become familiar with the molecular modeling kits, examine the colored balls that represent different elements common in biological molecules. Each colored ball has one or more holes. These holes represent the ability of these elements to form bonds:
| element | element abbreviation | molecular model ball color | bonding capacity |
|---|---|---|---|
| carbon | C | black | |
| oxygen | O | red | |
| hydrogen | H | white | |
| nitrogen | N | blue | |
| phosphorus | P | orange |
Based on the holes in each of the model element balls, fill in the column that indicates the bonding capacity of the element. Bonding capacity is based on the number of protons the element has and the number of valence electrons the element has.
Protein Monomers: Amino Acids
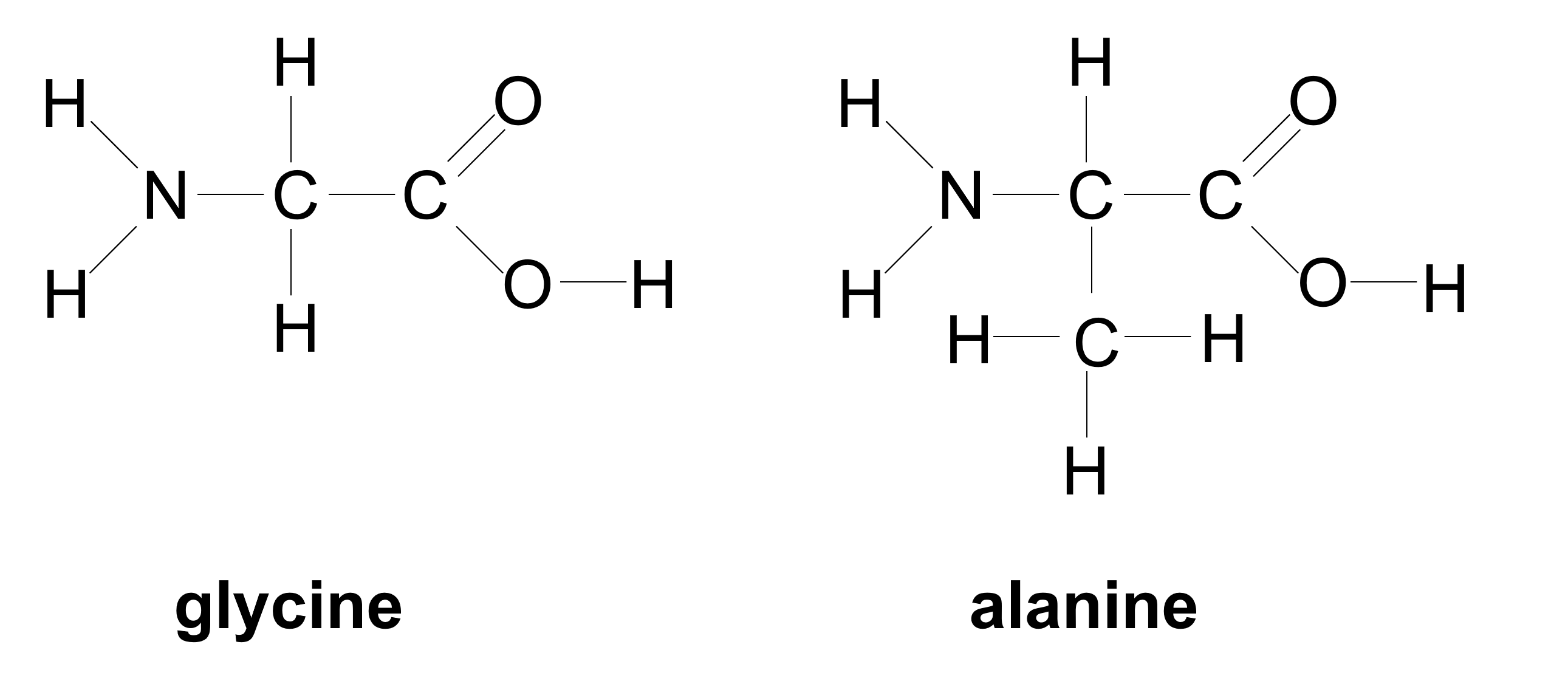
- Your instructor will assign you to build one of the two amino acids shown above.
- Use the diagram above and the table indicating the molecular model element ball colors to build the amino acid you are assigned.
- With your partner, conduct a dehydration synthesis reaction as directed by your instructor to form a dipeptide from your amino acids. This will form a molecule of water.
- Conduct a hydrolysis reaction by breaking the water molecule to break down the dipeptide into the two amino acids.
Carbohydrate Monomers: Monosaccharides
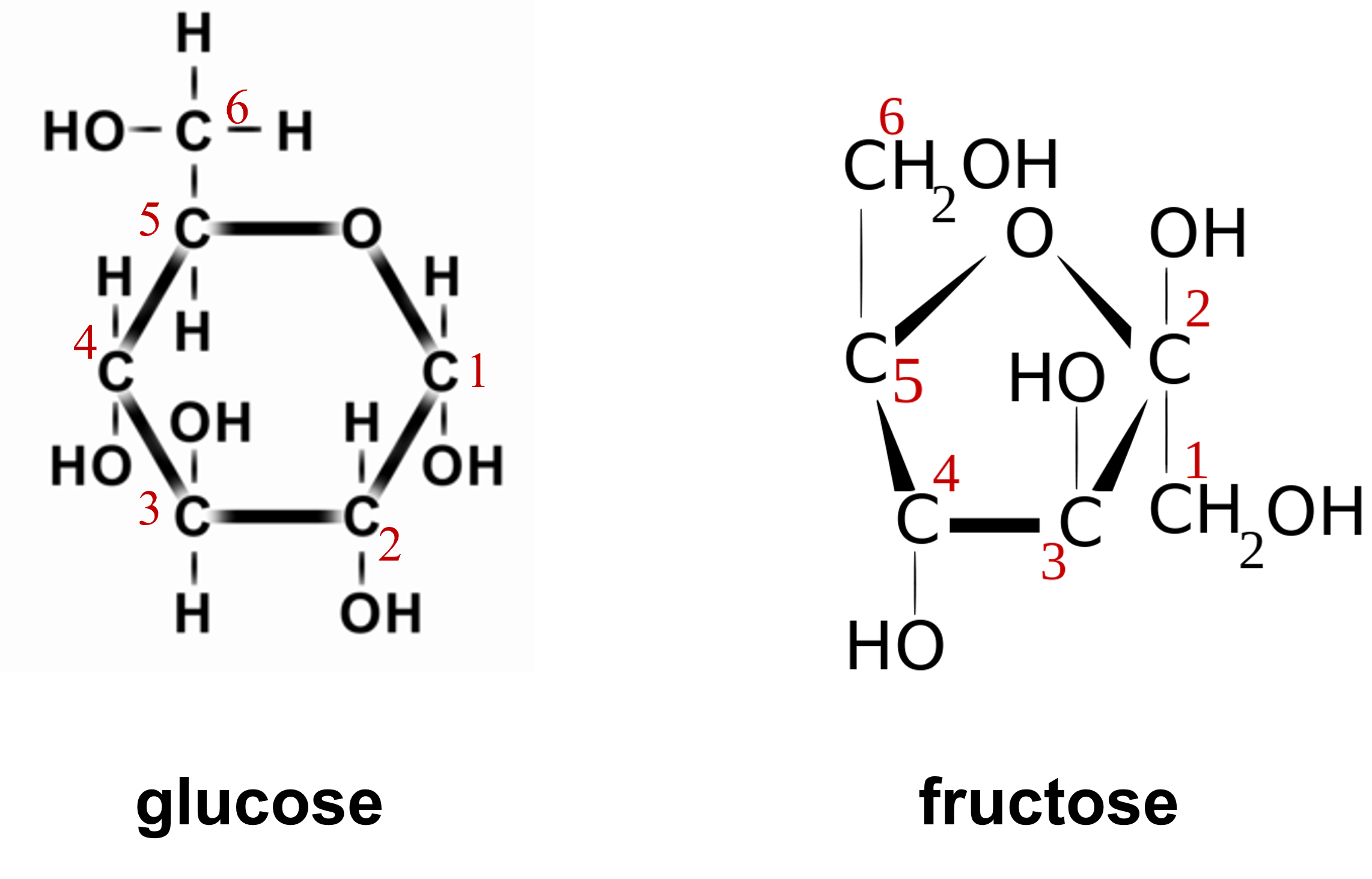
- Your instructor will assign you to build one of the two monosaccharides shown above.
- Use the diagram above and the table indicating the molecular model element ball colors to build the monosaccharide you are assigned.
- With your partner, conduct a dehydration synthesis reaction as directed by your instructor to form a disaccharide from your monosaccharides. This will form a molecule of water.
- Conduct a hydrolysis reaction by breaking the water molecule to break down the disaccharide into the two monosaccharides.
Start this structure by building the circle of carbons (they have red numbers 1-5 in glucose and 2-5 in fructose) with the one oxygen that helps make up the ring. Once you have the circle, then build off of that circle.
Lipids: Fatty Acids
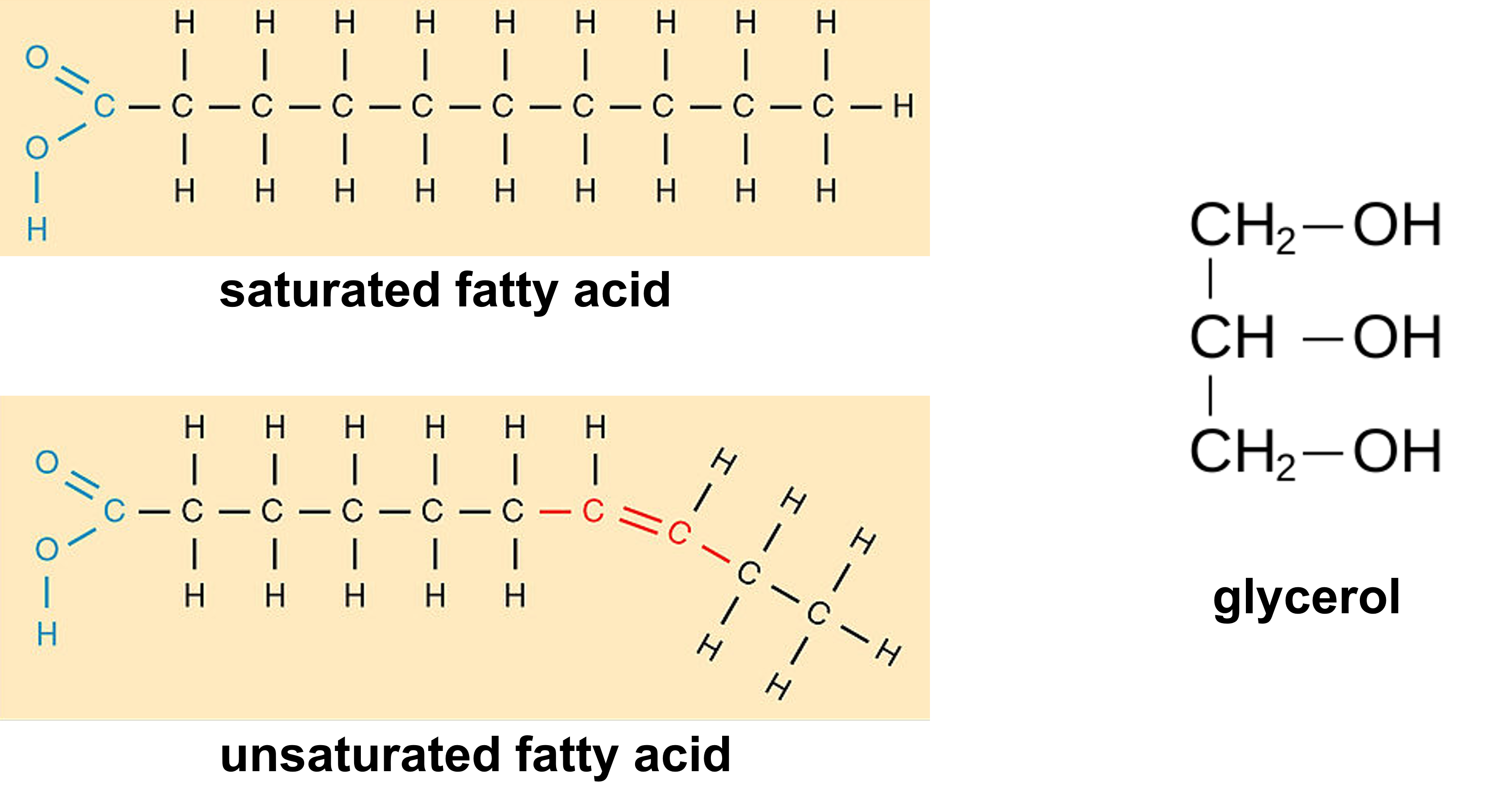
- Build the fatty acid shown above.
- Work with your partner to build one molecule of glycerol.
- With your partner, conduct two dehydration synthesis reactions as directed by your instructor to form a diglyceride from your two fatty acids and the glycerol. This will form two molecules of water.
- Conduct a hydrolysis reaction by breaking the water molecules to break down the diglycerides into the two fatty acids and one glycerol.
Nucleic Acids: Nucleotides
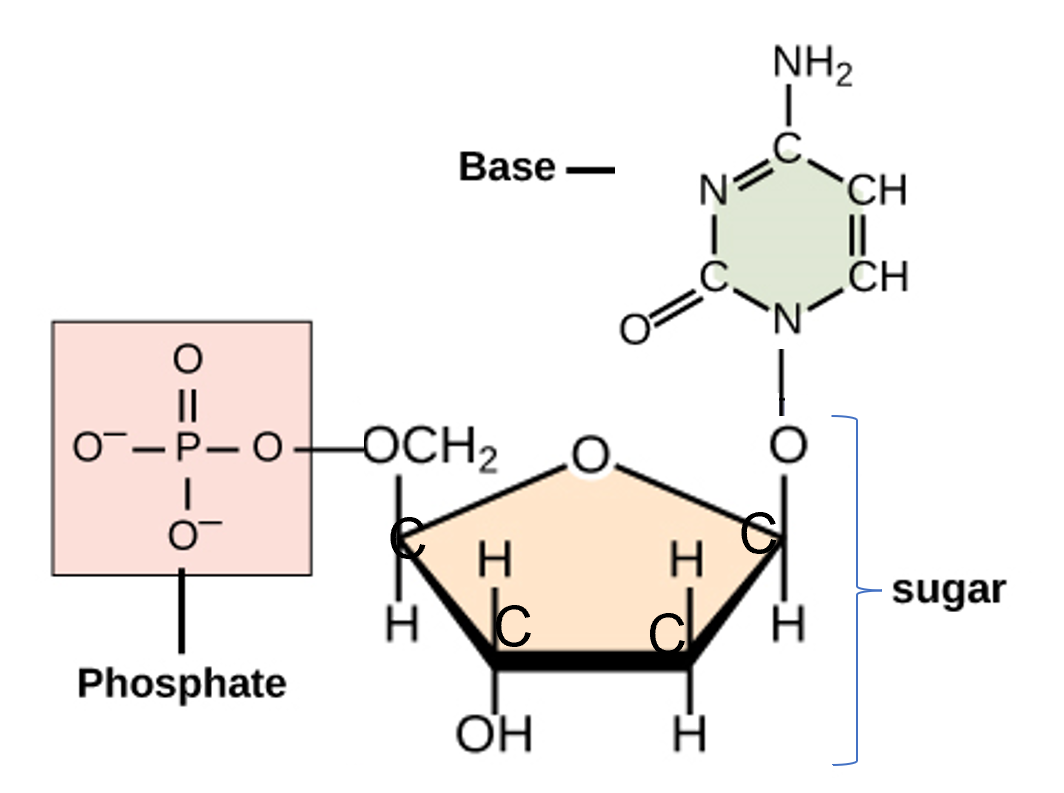
- Work together with your partner to build the nucleotide above.
- You can each build one of the pieces of the nucleotide: phosphate group, sugar, or base.
- When the three parts are completed, form bonds between the parts as shown in the diagram above.
Building Monomers of Biological Molecules Questions
| classification of biological molecule | monomer of this classification of biological molecule | elements found in this type of biological molecule | function(s) of this type of biological molecule | name a specific example of a molecule in this category of biological molecules |
|---|---|---|---|---|
- Fill out the table above to summarize information about the four categories of biological molecules.
- Define the following:
- atom:
- element:
- organic molecule:
- inorganic molecule:
- monomer:
- polymer:
- dehydration synthesis:
- hydrolysis:
- Catabolic reactions break down larger biological molecules into smaller biological molecules. Anabolic reactions form bonds to join together smaller biological molecules into larger biological molecules.
- Fill in the blanks with the bold words above: Dehydration synthesis is a type of reaction that must be a(n) _________________________ and hydrolysis is a type of reaction that must be a(n) ____________________________.
Foods and their Biological Molecules
Foods are Made of Biological Molecules
- Discuss with your group the types of foods you are familiar with that fall into the categories of biological molecules listed above. Fill in the table.
|
proteins (list protein-rich food below) |
carbohydrates (list carbohydrate-rich foods below) |
lipids (fats) (list lipid-rich foods/cooking ingredients below) |
|---|---|---|
Just like humans need food, so do microorganisms. Microorganisms also "eat." They, like us humans, require nutrients to survive.
Pathogenic (disease-causing) microorganisms use their host (e.g. humans) as a source of nutrients.
Test Food Samples for Proteins
- Put on laboratory safety goggles/glasses.
- Label five test tubes with the samples to be tested: green banana, black banana, muscle, egg, DI or distilled water.
- To each test tube, add 5 drops of the samples to be tested as labeled on the test tubes.
- Add 3 drops of biuret reagent and mix well by swirling.
- Wait 2 minutes.
- Blue color is negative for significant protein content; purple/violet is positive for significant protein content
- Record results in the table below.
Test Food Samples for Sugars (Small Carbohydrates)
- Empty and thoroughly rinse the test tubes after the protein test.
- To each test tube, add 5 drops of the samples to be tested as labeled on the test tubes.
- Add 3 drops of Benedict's solution and mix well by swirling.
- Gently place test tubes in a beaker of boiling or nearly boiling water.
- Wait 2 minutes.
- Use a test tube holder to remove the test tubes from the hot water and allow to cool.
- Examine the liquid in the test tubes and interpret the results:
- green to yellow - very low to low levels (positive) of monosaccharides and disaccharides
- yellow-orange to orange - moderate to high levels (positive) of monosaccharides and disaccharides
- orange-red - very high levels (positive) of monosaccharides and disaccharides
- blue, grey, or any other color - negative for detectable monosaccharides and disaccharides
- Record results in the table below.
Test Food Samples for Starch (A Large Carbohydrate)
- Empty and thoroughly rinse the test tubes after the sugars test.
- To each test tube, add 5 drops of the samples to be tested as labeled on the test tubes.
- Add 3 drops of iodine and mix well by swirling.
- Examine the liquid in the test tubes and interpret the results:
- black liquid or amber liquid with black chunks - positive for starch
- amber liquid (no black color) - negative for starch
- Record the color of the test result in the table below.
Test Food Samples for Lipids (Fats)
- On a piece of brown paper, draw five circles and label with with the samples to be tested: green banana, black banana, muscle, egg, DI or distilled water.
- Drop one drop of each substance to be tested into the appropriate circle and allow to dry for at least 15 minutes.
- Examine the dried spots. An oily spot (similar to the oil you would see on a brown napkin after french fries or potato chips) on the paper indicates a positive result for lipids.
- Record your results in the table below.
Foods & their Biological Molecules Results & Questions
| Type of Cells Blended with DI or distilled Water | protein (+/-) | sugar (small carbohydrates) (+/-) | starch (large carbohydrates) (+/-) | lipid (fats) (+/-) |
|---|---|---|---|---|
| green (under-ripe) banana cells | ||||
| black (over-ripe) banana cells | ||||
| muscle cells (chicken breast) | ||||
| egg (chicken egg) | ||||
| DI or distilled water (the control) |
- Complete the table above to summarize results from testing food items for biological molecules.
- Of the four categories of biological molecules, which ones did we test for in these experiments?
- Which category of biological molecule was not tested in these experiments?
- Nucleic acids include DNA molecules. All cells have DNA (with only very few exceptions). If we tested the following, should the be positive or negative for nucleic acids? Briefly explain your answer for each.
- banana cells:
- muscle cells:
- egg cells:
- DI water or distilled water:
- Each of the samples tested were prepared by mixing the food item with DI water or distilled water and mixing it in a blender. Think about the integrity of the experiment. Why did we need to also separately test the DI water or distilled water?
Attributions
- 221 Fatty Acids Shapes-01.jpg by OpenStax is licensed under CC BY 3.0
- Beta-d-fructose.svg by Bufalo 1973 is licensed under CC BY-SA 3.0
- Blausen 0434 Glucose-es.png by BruceBlaus from "Medical gallery of Blausen Medical 2014". WikiJournal of Medicine 1 (2). DOI:10.15347/wjm/2014.010. ISSN 2002-4436 is licensed under CC BY-SA 4.0
- Chapter Image: EF-G, mRNA, and tRNAs in POST state PDB 4W29.gif by Shahid.o is licensed under CC BY-SA 4.0
- Biological and technological scales compared-en.svg by Guillaume Paumier, Philip Ronan, NIH, Artur Jan Fijałkowski, Jerome Walker, Michael David Jones, Tyler Heal, Mariana Ruiz, Science Primer (National Center for Biotechnology Information), Liquid_2003, Arne Nordmann & The Tango! Desktop Project is licensed under CC BY-SA 2.
- Function Follows Structure by Bonnie Waltz, Tracy Rains, Deanna Mayers is licensed under CC BY-NC 4.0
- General Biology by OpenStax is licensed under CC BY 4.0
- Glycerol structure.svg by すじにくシチュー is dedicated to the public domain
- Holding glass test tube.jpg by J.N. Eskra is licensed under CC BY-SA 4.0
- The Periodic Table of the Elements, in pictures.png by Keith Enevoldsen is licensed under CC BY-SA 4.0


