15.1: Types of Environmental Hazards
- Page ID
- 31632
\( \newcommand{\vecs}[1]{\overset { \scriptstyle \rightharpoonup} {\mathbf{#1}} } \)
\( \newcommand{\vecd}[1]{\overset{-\!-\!\rightharpoonup}{\vphantom{a}\smash {#1}}} \)
\( \newcommand{\id}{\mathrm{id}}\) \( \newcommand{\Span}{\mathrm{span}}\)
( \newcommand{\kernel}{\mathrm{null}\,}\) \( \newcommand{\range}{\mathrm{range}\,}\)
\( \newcommand{\RealPart}{\mathrm{Re}}\) \( \newcommand{\ImaginaryPart}{\mathrm{Im}}\)
\( \newcommand{\Argument}{\mathrm{Arg}}\) \( \newcommand{\norm}[1]{\| #1 \|}\)
\( \newcommand{\inner}[2]{\langle #1, #2 \rangle}\)
\( \newcommand{\Span}{\mathrm{span}}\)
\( \newcommand{\id}{\mathrm{id}}\)
\( \newcommand{\Span}{\mathrm{span}}\)
\( \newcommand{\kernel}{\mathrm{null}\,}\)
\( \newcommand{\range}{\mathrm{range}\,}\)
\( \newcommand{\RealPart}{\mathrm{Re}}\)
\( \newcommand{\ImaginaryPart}{\mathrm{Im}}\)
\( \newcommand{\Argument}{\mathrm{Arg}}\)
\( \newcommand{\norm}[1]{\| #1 \|}\)
\( \newcommand{\inner}[2]{\langle #1, #2 \rangle}\)
\( \newcommand{\Span}{\mathrm{span}}\) \( \newcommand{\AA}{\unicode[.8,0]{x212B}}\)
\( \newcommand{\vectorA}[1]{\vec{#1}} % arrow\)
\( \newcommand{\vectorAt}[1]{\vec{\text{#1}}} % arrow\)
\( \newcommand{\vectorB}[1]{\overset { \scriptstyle \rightharpoonup} {\mathbf{#1}} } \)
\( \newcommand{\vectorC}[1]{\textbf{#1}} \)
\( \newcommand{\vectorD}[1]{\overrightarrow{#1}} \)
\( \newcommand{\vectorDt}[1]{\overrightarrow{\text{#1}}} \)
\( \newcommand{\vectE}[1]{\overset{-\!-\!\rightharpoonup}{\vphantom{a}\smash{\mathbf {#1}}}} \)
\( \newcommand{\vecs}[1]{\overset { \scriptstyle \rightharpoonup} {\mathbf{#1}} } \)
\( \newcommand{\vecd}[1]{\overset{-\!-\!\rightharpoonup}{\vphantom{a}\smash {#1}}} \)
\(\newcommand{\avec}{\mathbf a}\) \(\newcommand{\bvec}{\mathbf b}\) \(\newcommand{\cvec}{\mathbf c}\) \(\newcommand{\dvec}{\mathbf d}\) \(\newcommand{\dtil}{\widetilde{\mathbf d}}\) \(\newcommand{\evec}{\mathbf e}\) \(\newcommand{\fvec}{\mathbf f}\) \(\newcommand{\nvec}{\mathbf n}\) \(\newcommand{\pvec}{\mathbf p}\) \(\newcommand{\qvec}{\mathbf q}\) \(\newcommand{\svec}{\mathbf s}\) \(\newcommand{\tvec}{\mathbf t}\) \(\newcommand{\uvec}{\mathbf u}\) \(\newcommand{\vvec}{\mathbf v}\) \(\newcommand{\wvec}{\mathbf w}\) \(\newcommand{\xvec}{\mathbf x}\) \(\newcommand{\yvec}{\mathbf y}\) \(\newcommand{\zvec}{\mathbf z}\) \(\newcommand{\rvec}{\mathbf r}\) \(\newcommand{\mvec}{\mathbf m}\) \(\newcommand{\zerovec}{\mathbf 0}\) \(\newcommand{\onevec}{\mathbf 1}\) \(\newcommand{\real}{\mathbb R}\) \(\newcommand{\twovec}[2]{\left[\begin{array}{r}#1 \\ #2 \end{array}\right]}\) \(\newcommand{\ctwovec}[2]{\left[\begin{array}{c}#1 \\ #2 \end{array}\right]}\) \(\newcommand{\threevec}[3]{\left[\begin{array}{r}#1 \\ #2 \\ #3 \end{array}\right]}\) \(\newcommand{\cthreevec}[3]{\left[\begin{array}{c}#1 \\ #2 \\ #3 \end{array}\right]}\) \(\newcommand{\fourvec}[4]{\left[\begin{array}{r}#1 \\ #2 \\ #3 \\ #4 \end{array}\right]}\) \(\newcommand{\cfourvec}[4]{\left[\begin{array}{c}#1 \\ #2 \\ #3 \\ #4 \end{array}\right]}\) \(\newcommand{\fivevec}[5]{\left[\begin{array}{r}#1 \\ #2 \\ #3 \\ #4 \\ #5 \\ \end{array}\right]}\) \(\newcommand{\cfivevec}[5]{\left[\begin{array}{c}#1 \\ #2 \\ #3 \\ #4 \\ #5 \\ \end{array}\right]}\) \(\newcommand{\mattwo}[4]{\left[\begin{array}{rr}#1 \amp #2 \\ #3 \amp #4 \\ \end{array}\right]}\) \(\newcommand{\laspan}[1]{\text{Span}\{#1\}}\) \(\newcommand{\bcal}{\cal B}\) \(\newcommand{\ccal}{\cal C}\) \(\newcommand{\scal}{\cal S}\) \(\newcommand{\wcal}{\cal W}\) \(\newcommand{\ecal}{\cal E}\) \(\newcommand{\coords}[2]{\left\{#1\right\}_{#2}}\) \(\newcommand{\gray}[1]{\color{gray}{#1}}\) \(\newcommand{\lgray}[1]{\color{lightgray}{#1}}\) \(\newcommand{\rank}{\operatorname{rank}}\) \(\newcommand{\row}{\text{Row}}\) \(\newcommand{\col}{\text{Col}}\) \(\renewcommand{\row}{\text{Row}}\) \(\newcommand{\nul}{\text{Nul}}\) \(\newcommand{\var}{\text{Var}}\) \(\newcommand{\corr}{\text{corr}}\) \(\newcommand{\len}[1]{\left|#1\right|}\) \(\newcommand{\bbar}{\overline{\bvec}}\) \(\newcommand{\bhat}{\widehat{\bvec}}\) \(\newcommand{\bperp}{\bvec^\perp}\) \(\newcommand{\xhat}{\widehat{\xvec}}\) \(\newcommand{\vhat}{\widehat{\vvec}}\) \(\newcommand{\uhat}{\widehat{\uvec}}\) \(\newcommand{\what}{\widehat{\wvec}}\) \(\newcommand{\Sighat}{\widehat{\Sigma}}\) \(\newcommand{\lt}{<}\) \(\newcommand{\gt}{>}\) \(\newcommand{\amp}{&}\) \(\definecolor{fillinmathshade}{gray}{0.9}\)Environmental health is a field that focuses on how the natural and human-built surroundings as well as behaviors affect human well-being. The field is concerned with preventing disease, death, and disability by reducing exposure to environmental hazards and promoting behavioral change. Environmental hazards are threats to human health and well-being (table \(\PageIndex{a}\)).
| Underlying Determinants | Possible Adverse Health and Safety Consequences |
|---|---|
| Inadequate water (quantity and quality), sanitation and solid waste disposal, improper hygein (handwashing) | Diarrhea and vector-related diseases (for example, malaria, schistosomiasis, and dengue) |
| Improper water resource managment, uncluding poor drainage | Vector-related diseases |
| Crowded housing and poor ventialtion of smoke | Acute and chronic respiratory disease, including lung cancer from coal and tobacco inhalation |
| Exposures to vehicular and industrial air pollution | Respiratory diseases, some cancers, and loss of IQ in children |
| Population movement and encroachment and construction, which affect feeding and breeding grounds of vectors, such as mosquitoes |
Vector-related diseases May also spread other infectious diseases (for example, HIV/AIDS, Ebola) |
| Exposure to naturally ocurring toxic substances | Poising from substances such as arsenic, manganese, and fluorides |
| Natural resources degradation (for example, landslides, poor drainage, erosion) | Injury and death from landslides and flooding |
| Climate change, partly from combustion of fossil fuel and release of greenhouse gases in transportation, industry, and poor energy conservation in housing, fuel, commerce, and industry |
Injury/death from extreeme heat/cold, sotres, floods, and fires Indirect effects spread of vectorbrone diseases, aggravation of respriatory diseases, population dislocation, water pollution from sea level rise, etc. |
| Ozone depletion from industrial and commercial actvitiy |
Skin cancer, cataracts Indirect effect: compromised food production |
Table based on Lvovsky/World Bank (CC-BY)
Traditional versus Modern Environmental Hazards
Environmental hazards can be classified as traditional or modern. Traditional hazards are related to poverty and mostly affect low-income people and those in developing countries. Modern hazards, caused by technological development, prevail in industrialized countries where exposure to traditional hazards is low.
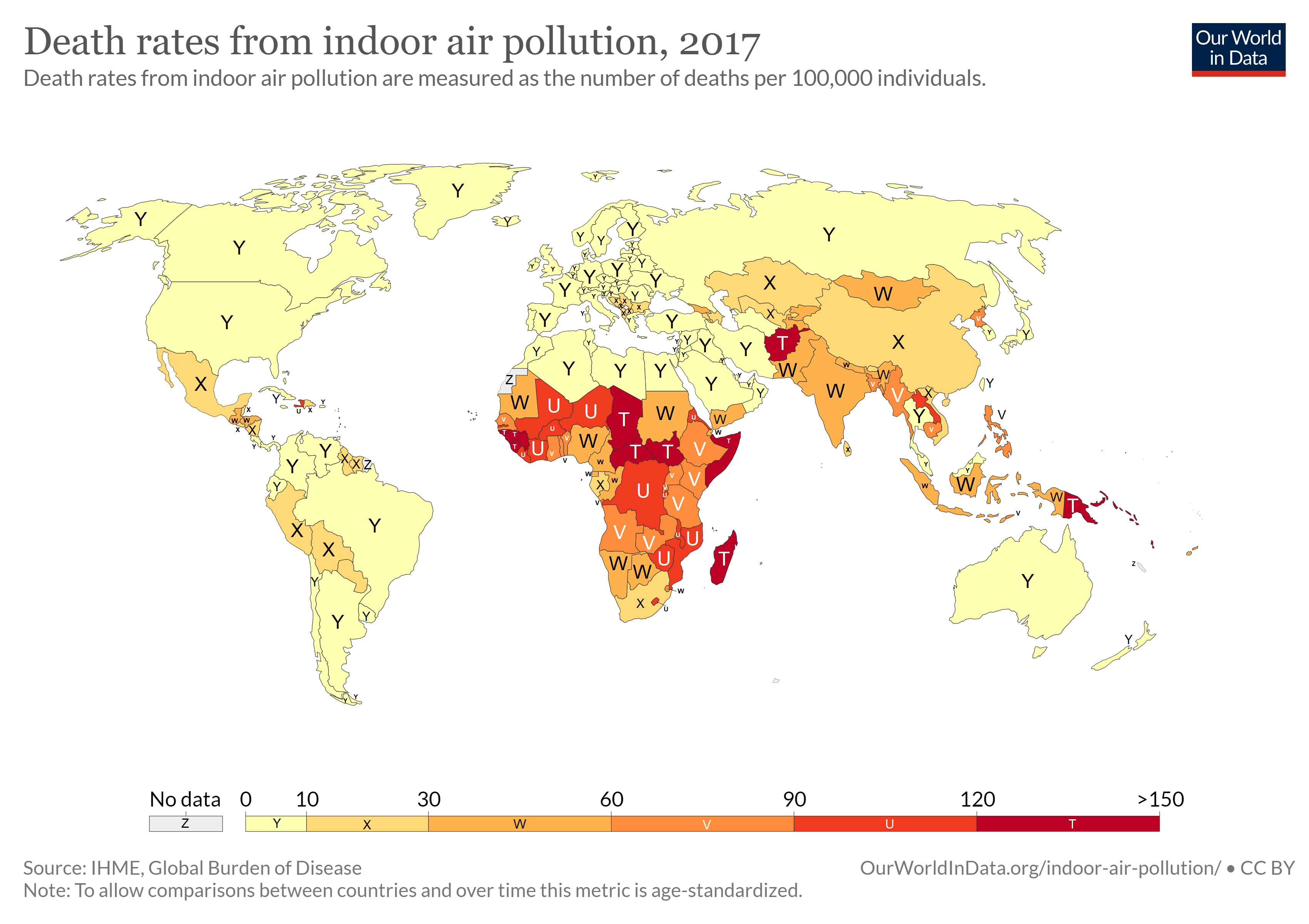
The impact of traditional hazards exceeds that of modern hazards by 10 times in Africa, five times in Asian countries (except for China), and 2.5 times in Latin America and the Middle East (figure \(\PageIndex{a}\)). Water-related diseases caused by inadequate water supply and sanitation impose an especially large health burden in Africa, Asia, and the Pacific region. In India alone, 90,000 children under five died diarrhea in 2017. Globally, 409,000 people died of malaria in 2019 with 94% of these deaths occurring in African countries. In 2016, approximately one third of the world’s households used unprocessed solid fuels, particularly biomass (crop residues, wood, and dung) for cooking and heating in inefficient stoves without proper ventilation. This exposes people—mainly low-income women and children—to high levels of indoor air pollution, the cause of about 1.6 million deaths in each year (figure \(\PageIndex{b}\)).
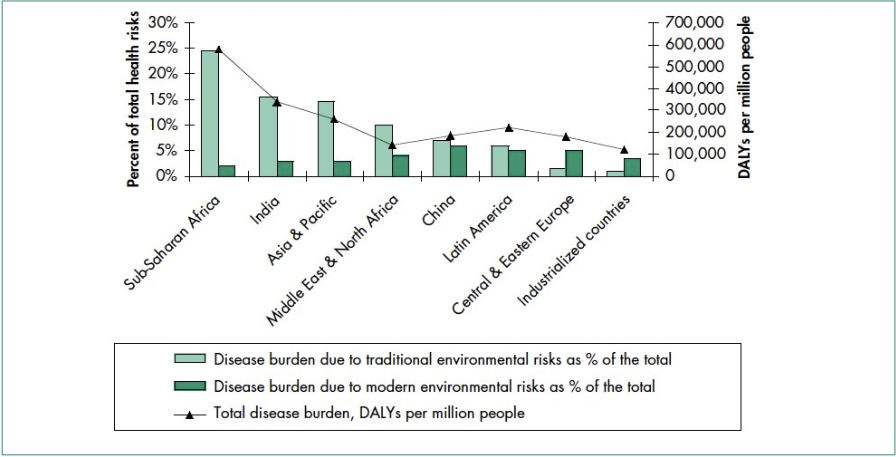
The contribution of modern environmental risks to the disease burden in most developing countries is similar to – and in quite a few countries, greater than – that in rich countries (figure \(\PageIndex{a}\)). Urban air pollution, for example, is highest in parts of China, India, and some cities in Asia and Latin America. Low-income people increasingly experience a “double burden” of traditional and modern environmental health risks. In rich countries, they experience twice the burden of illness and death from all causes and 10 times greater disease burden from environmental risks.
Biological, Chemical, and Physical Environmental Hazards
Environmental hazards can also be classified into three interrelated categories (biological, chemical, and physical) based on the properties of their causes. These categories are not mutually exclusive with traditional versus modern hazards. For example, indoor air pollution is both a traditional and chemical hazard. Different hazards can interact and exacerbate one another. For example, a flood is primarily a physical hazard, but it can lead to the spread of waterborne disease (a biological hazard). Similarly, air pollution (a chemical hazard) can damage respiratory tissue, making the body more vulnerable to a respiratory infection (a biological hazard). Infectious diseases (biological hazards) can also weaken the immune system, making an individual more vulnerable to chemical hazards.
Biological Hazards
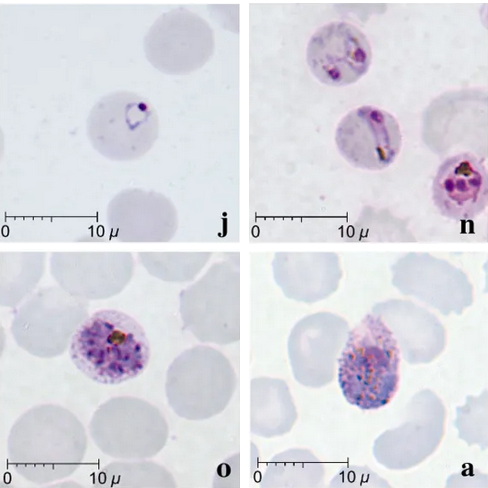
For most of human history, biological hazards were the most significant factor in health. Biological hazards are infectious (communicable) diseases caused by pathogens (disease-causing organisms or infectious particles) such as bacteria, fungi, parasitic worms, protozoa, viruses, and prions. Bacteria are single-celled organisms with small, simple cells. Examples of bacterial diseases include tuberculosis, cholera, bacterial pneumonia, and dysentery. Fungi may have one or multiple cells and have a more complex cell type than bacteria. Fungal diseases include minor infections like candidiasis (yeast infection) or athlete's foot, but they can also causes severe respiratory infections (histoplasmosis, coccidioidomycosis, etc.) particularly in individuals with compromised immune systems. Parasitic worms are animals from several phyla (groups) that siphon nutrients from their hosts. Examples include tapeworms, commonly acquired through consuming undercooked meat, and blood flukes (Schistosoma). Like fungi, protozoa have larger, more complex cells than bacteria, but they are single celled and lack the rigid cell wall that surrounds fungal cells. Malaria (figure \(\PageIndex{c}\)), African trypanosomiasis (sleeping sickness), and giardiasis are caused by protozoa. Viruses are infectious particles with genetic information surrounded by a protein coat, but they are not technically considered organisms in part because they do not consist fo cells. COVID-19, influenza, measles, the common cold, ebola viral disease (Ebola hemorrhagic fever), and human immunodeficiency virus (HIV)/acquired immune deficiency syndrome (AIDS) are all caused by viruses. Prions (proteinaceous infectious particles) are even simpler than viruses because they lack genetic material and only contain protein.
While the proportion of deaths caused by in infectious diseases has overall decreased (with a higher proportion of deaths caused by noncommunicable diseases such as cancer and cardiovascular disease), infectious diseases still caused about one in five deaths in 2017. These deaths occurred at the highest rates in developing countries and many were in children. Malnutrition, unclean water, poor sanitary conditions and lack of proper medical care all play roles in transmission and high death rates from infectious diseases Compounding the problems of infectious diseases are factors such as antibiotic-resistant pathogens, pesticide-resistant disease vectors, and overpopulation.
Chemical Hazards
Chemical hazards are toxic substances, which cause damage to living organisms. Air pollutants (such as secondhand smoke or carbon monoxide), heavy metals, and pesticides are a few examples. We can be exposed to these contaminants from a variety of residential, commercial, and industrial sources. Sometimes harmful environmental contaminants occur biologically, such as those from mold or a toxic algae bloom. Toxins can be classified based on their origin, purpose, chemical structure and properties, or effects. Table \(\PageIndex{b}\) describes a few categories of toxins based on their effects and provides examples. A few of these examples are discussed more specifically below.
| Contaminant | Definition |
|---|---|
| Carcinogen | An agent which may produce cancer (uncontrolled cell growth), either by itself or in conjunction with another substance. Examples include formaldehyde, asbestos, radon, vinyl chloride, and tobacco. |
| Teratogen |
A substance which can cause physical defects in a developing embryo. Examples include alcohol and cigarette smoke. |
| Mutagen | A material that induces genetic changes (mutations) in the DNA. Examples include radioactive substances (such as radon and nuclear fuel and waste) and nitrous acid. Some forms of radiation (see Physical Hazards) are also mutagens. |
| Neurotoxin |
A substance that can cause an adverse effect on the chemistry, structure or function of the nervous system. Examples include lead and mercury. |
|
Endocrine disruptor |
A chemical that may interfere with the body’s endocrine (hormonal) system and produce adverse developmental, reproductive, neurological, and immune effects in both humans and wildlife. A wide range of substances, both natural and man-made, are thought to cause endocrine disruption, including pharmaceuticals, dioxin and dioxin-like compounds, arsenic, polychlorinated biphenyls (PCBs), DDT and other pesticides, per- and polyfluoroalkyl substances (PFAS), pthalates, and plasticizers such as bisphenol A (BPA). |
Formaldehyde
Formaldehyde is a colorless, flammable gas or liquid that has a pungent, suffocating odor. It is a volatile organic compound, which is a compound containing carbon and hydrogen that easily becomes a vapor or gas. It is also naturally produced in small, harmless amounts in the human body. The primary way we can be exposed to formaldehyde is by breathing air containing it. Formaldehyde is released into the air by industries using or manufacturing formaldehyde, wood products (such as particle-board, plywood, and furniture), automobile exhaust, cigarette smoke, paints and varnishes, and carpets and permanent press fabrics. Nail polish and commercially applied floor finish emit formaldehyde (figure \(\PageIndex{d}\)).

In general, indoor environments consistently have higher concentrations than outdoor environments because many building materials, consumer products, and fabrics emit formaldehyde. Levels of formaldehyde measured in indoor air range from 0.02–4 parts per million (ppm). Formaldehyde levels in outdoor air range from 0.001 to 0.02 ppm in urban areas.
Heavy Metals
Heavy metals are chemical elements of high density that form a special type of bond (called metallic bonds, in which electrons are shared but in a less constricted way than in covalent bonds). Arsenic, mercury, lead, and cadmium are examples of heavy metals.
Arsenic (As) is a naturally occurring element that is normally present throughout our environment in water, soil, dust, air, and food. Levels of arsenic can regionally vary due to farming and industrial activity as well as natural geological processes. The arsenic from farming and smelting tends to bind strongly to soil and is expected to remain near the surface of the land for hundreds of years as a long-term source of exposure. Wood that has been treated with chromated copper arsenate (CCA) is commonly found in decks and railings in existing homes and outdoor structures such as playground equipment. Some underground aquifers are located in rock or soil that has naturally high arsenic content.
Most arsenic gets into the body through ingestion of food or water. Arsenic in drinking water is a problem in many countries around the world, including Bangladesh, Chile, China, Vietnam, Taiwan, India, and the United States. Arsenic may also be found in foods, including rice and some fish, where it is present due to uptake from soil and water. It can also enter the body by breathing dust containing arsenic.
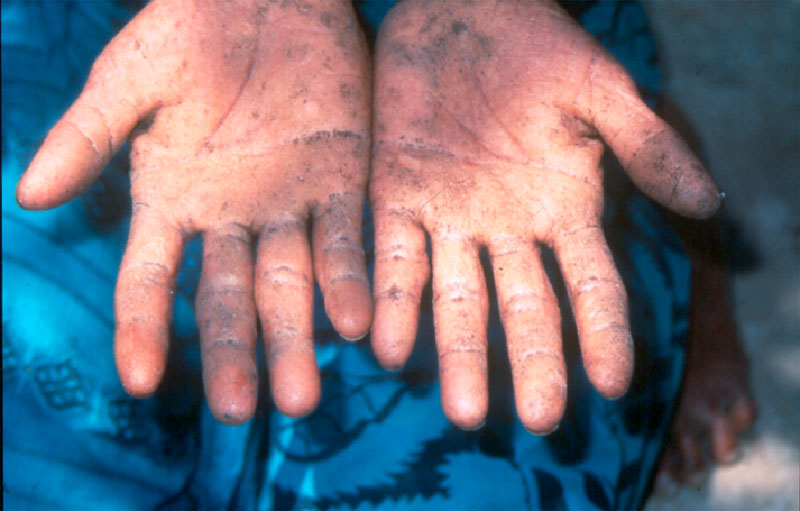
Arsenic poisoning causes a variety of symptoms and serious health conditions (figure \(\PageIndex{e}\)). Researchers are finding that arsenic, even at low levels, can interfere with the body’s endocrine system. Arsenic is also a known human carcinogen associated with skin, lung, bladder, kidney, and liver cancer.
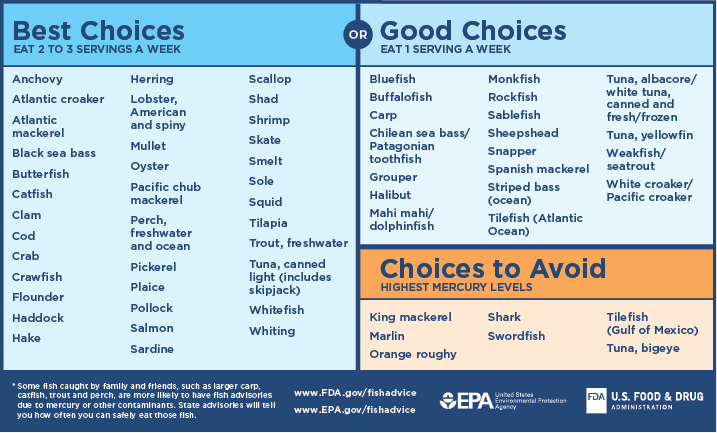
Mercury (Hg) is a naturally occurring metal, a useful chemical in some products, and a potential health risk. Mercury exists in several forms; the types people are usually exposed to are methylmercury and elemental mercury. Elemental mercury at room temperature is a shiny, silver-white liquid which can produce a harmful odorless vapor. Methylmercury, an organic compound, can build up in the bodies of long-living, predatory fish (Biomagnification). Although fish and shellfish have many nutritional benefits, consuming large quantities of fish increases a person’s exposure to mercury. Pregnant women who eat fish high in mercury on a regular basis run the risk of permanently damaging their developing fetuses. Children born to these mothers may exhibit motor difficulties, sensory problems and cognitive deficits. The United States Environmental Protection Agency thus recommends that pregnant women and young children should not consume any swordfish, shark, king mackerel, or tilefish because of their high mercury content. These individuals are advised to eat fish low in mercury such as salmon, shrimp, pollock, and catfish (figure \(\PageIndex{f}\)). To keep mercury out of the fish we eat and the air we breathe, it’s important to take mercury-containing products to a hazardous waste facility for disposal. Common products sold today that contain small amounts of mercury include fluorescent lights and button-cell batteries (figure \(\PageIndex{g}\)).

Lead (Pb) is a metal that occurs naturally in the rocks and soil of the Earth’s crust. It is also released from mining, manufacturing, and the combustion (burning) fossil fuels such as coal, oil, gasoline, and natural gas. Lead has no distinctive taste or smell. Lead is used to produce batteries, pipes, roofing, scientific electronic equipment, military tracking systems, medical devices, and products to shield X-rays and nuclear radiation. It is used in ceramic glazes and crystal glassware. Because of health concerns, lead and lead compounds were banned from house paint in 1978; from solder used on water pipes in 1986; from gasoline in 1995; from solder used on food cans in 1996; and from tin-coated foil on wine bottles in 1996. The U.S. Food and Drug Administration has set a limit on the amount of lead that can be used in ceramics.
Lead and lead compounds are listed as “reasonably anticipated to be a human carcinogen”. It can affect almost every organ and system in your body. It can be equally harmful if breathed or swallowed. The part of the body most sensitive to lead exposure is the central nervous system, especially in children, who are more vulnerable to lead poisoning than adults. A child who swallows large amounts of lead can develop brain damage that can cause convulsions and death; the child can also develop blood anemia, kidney damage, colic, and muscle weakness. Repeated low levels of exposure to lead can alter a child’s normal mental and physical growth and result in learning or behavioral problems. Exposure to high levels of lead for pregnant women can cause miscarriage, premature births, and smaller babies. Repeated or chronic exposure can cause lead to accumulate in your body, leading to lead poisoning.
The video below explains how Flint, Michigan's water supply was polluted with lead in 2014 and the problematic government response that followed. This video was made in 2016. As of 2020, Flint has a clean water source, and the city of Flint is in the process of compensating affected residents for the damages. (Click here to read a 2020 update.)
Asbestos
Asbestos is a mineral fiber that occurs in rock and soil. Because of its fiber strength and heat resistance asbestos has been used in a variety of building construction materials for insulation and as a fire retardant. Asbestos has also been used in a wide range of manufactured goods, mostly in building materials (roofing shingles, ceiling and floor tiles, paper products, and asbestos cement products), friction products (automobile clutch, brake, and transmission parts), heat-resistant fabrics, packaging, gaskets, and coatings. Exposure to asbestos is associated with cancers (lung cancer and mesothelioma) and another lung disease called asbestosis. In the United States, certain uses of asbestos, including in corrugated paper, flooring, and building insulation, are banned under the Toxic Substances Control Act and Clean Air Act (figure \(\PageIndex{h}\)). In contrast, asbestos is fully banned in 67 countries as of 2019.

Per- and polyfluoroalkyl substances (PFAS)
Per- and polyfluoroalkyl substances (PFAS) are a group of manufactured organic chemicals used in a variety of industries (figure \(\PageIndex{i}\)). They can be found in food packaging, stain- and water-repellent fabrics, nonstick products (such as Teflon), polishes, waxes, paints, cleaning products, and fire-fighting foams.
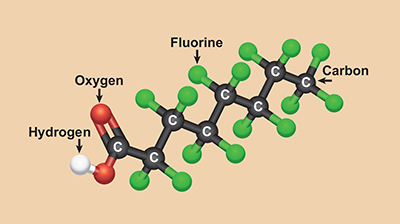
Studies indicate that some PFAS can cause reproductive and developmental, liver and kidney, and immunological effects in laboratory animals. More limited findings associate some PFAS with low infant birth weights, effects on the immune system, cancer, and thyroid hormone disruption in humans.
Eight major chemical manufacturers in the U.S. phased out the use certain PFAS (called perfluorooctanoic acid, PFOA, and perfluorooctane sulfonate, PFOS) and related chemicals in their products and as emissions from their facilities. However, these PFAS can still be imported and other PFAS are still manufactured in the U.S.
Polychlorinated Biphenyls (PCBs)
Polychlorinated biphenyls (PCBs) are a group of manufactured organic chemicals. They belong to a broad family of chemicals known as chlorinated hydrocarbons, which consisting of carbon, hydrogen and chlorine atoms (figure \(\PageIndex{j}\)). The number of chlorine atoms and their location in a PCB molecule determine many of its physical and chemical properties. PCBs have no known taste or smell, and range in consistency from an oil to a waxy solid.
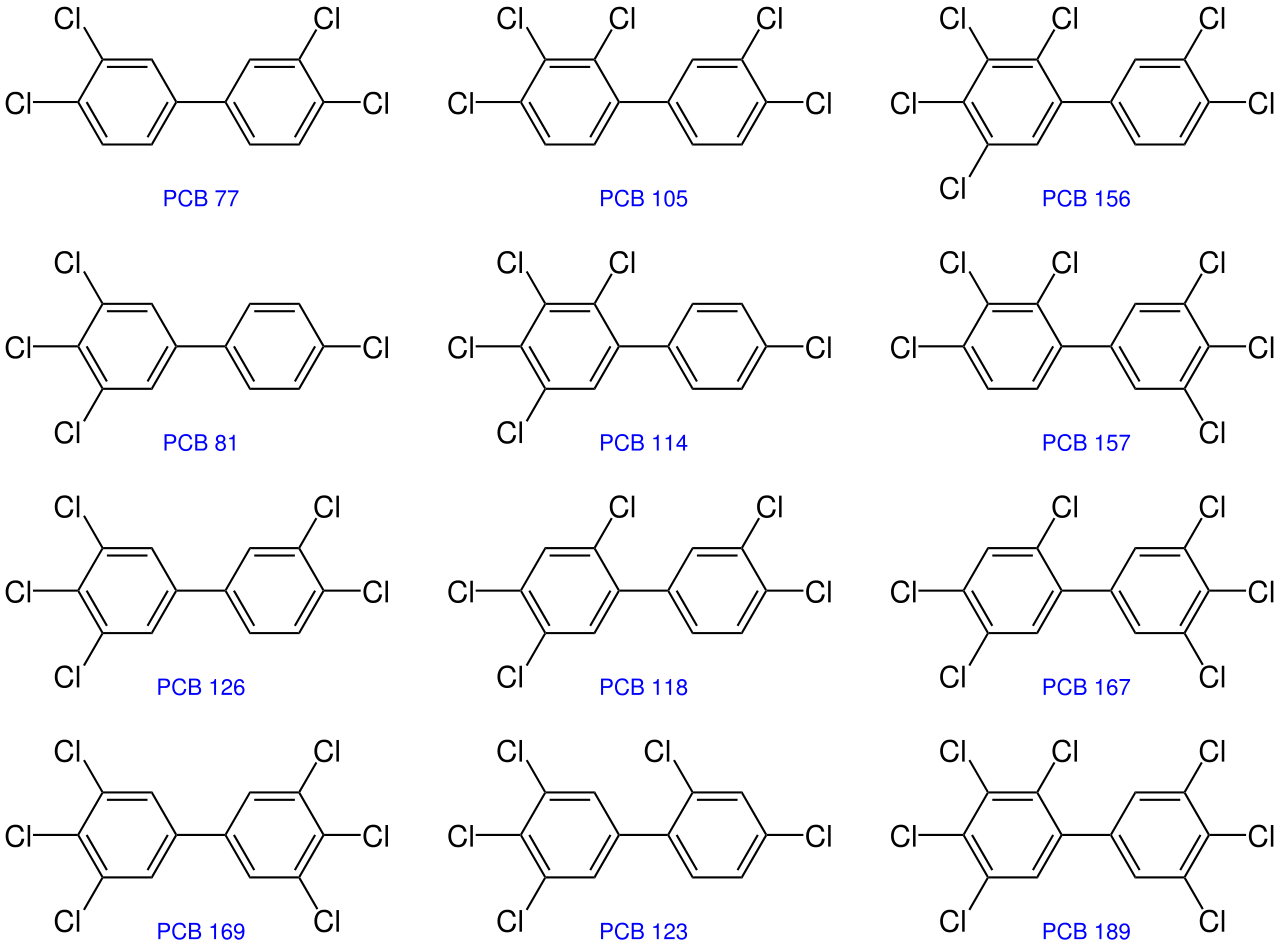
Polychlorinated biphenyls have been shown to cause cancer, cause birth defects, and affect the immune, reproductive, nervous, and endocrine systems in animals. Studies in humans support evidence for potential carcinogenic and non-carcinogenic effects of PCBs. The different health effects of PCBs may be interrelated. Alterations in one system may have significant implications for the other systems of the body.
The manufacture of PCBs in the U.S. began in 1929 until it was banned in 1979 under the Toxic Substances Control Act. Due to their non-flammability, chemical stability, high boiling point and electrical insulating properties, PCBs were used in hundreds of industrial and commercial applications including in electrical equipment, coolants paints, plastics, rubber products, pigments, and dyes.
Bisphenol A (BPA)
Bisphenol A (BPA) is a chemical synthesized in large quantities for use primarily in the production of polycarbonate plastics and epoxy resins. Polycarbonate plastics have many applications including use in some food and drink packaging such as water and infant bottles, impact-resistant safety equipment, and medical devices (figure \(\PageIndex{k}\)). Epoxy resins are used as lacquers to coat metal products such as food cans, bottle tops, and water supply pipes. Some dental sealants and composites may also contribute to BPA exposure. The primary source of exposure to BPA for most people is through the diet. Bisphenol A can leach into food from the protective internal epoxy resin coatings of canned foods and from consumer products such as polycarbonate tableware, food storage containers, water bottles, and baby bottles. The degree to which BPA leaches from polycarbonate bottles into liquid may depend more on the temperature of the liquid or bottle, than the age of the container. It has also be found in breast milk.
Some animal studies suggest that infants and children may be the most vulnerable to the effects of BPA. It disrupts signaling by estrogen, a naturally produced hormone, and the U.S. National Toxicology Program (NTP) documented concerns about its effects on the behavior, brain, and prostate in young children and developing fetuses.
The following personal choices can reduce exposure to BPA:
- Avoid microwaving polycarbonate plastic food containers. Polycarbonate is strong and durable, but over time it may break down from over use at high temperatures.
- Note recycle codes on the bottom of plastic containers. Some, but not all, plastics that are marked with recycle codes 3 or 7 may be made with BPA.
- Reduce your use of canned foods.
- When possible, opt for glass, porcelain or stainless steel containers, particularly for hot food or liquids.
While BPA is not banned in the U.S., the Food and Drug Adminsitration banned its use in baby bottles and sippy cups in 2012 and its use in the coating of infant formula containers in 2013. However, similar compounds such as bisphenol S (BPS) are now used as replacements.
Phthalates
Phthalates are a group of synthetic chemicals used to soften and increase the flexibility of plastic and vinyl. Polyvinyl chloride is made softer and more flexible by the addition of phthalates. Phthalates are used in hundreds of consumer products. Phthalates are used in cosmetics and personal care products, including perfume, hair spray, soap, shampoo, nail polish, and skin moisturizers (figure \(\PageIndex{l}\)). They are used in consumer products such as flexible plastic and vinyl toys, shower curtains, wallpaper, vinyl miniblinds, food packaging, and plastic wrap. Exposure to low levels of phthalates may come from eating food packaged in plastic that contains phthalates or breathing dust in rooms with vinyl miniblinds, wallpaper, or recently installed flooring that contain phthalates. We can be exposed to phthalates by drinking water that contains phthalates.

Phthalates are suspected to be endocrine disruptors. Some types of phthalates have affected the reproductive system of laboratory animals. In 2017, The U.S. Consumer Product Safety Commission (CPSC) has banned several pthalates from being used at concentrations greater than 0.1% in toys and products designed to be used by children three years old or younger.
Radon
Radon is a radioactive gas that is naturally-occurring, colorless, and odorless (figure \(\PageIndex{m}\)). It comes from the natural decay of uranium or thorium found in nearly all soils. It typically moves up through the ground and into the home through cracks in floors, walls and foundations. It can also be released from building materials or from well water. Radon breaks down quickly, giving off radioactive particles. Long-term exposure to these particles can lead to lung cancer. Radon is the leading cause of lung cancer among nonsmokers, according to the U.S. Environmental Protection Agency, and the second leading cause behind smoking. To reduce the risk of radon exposure, the Department of Urban and Housing Development recommends testing your home for radon, avoiding smoking to reduce the risk of lung cancer, and ensuring proper ventilation in your home.

Dichlorodiphenyltrichloroethane (DDT)
Dichlorodiphenyltrichloroethane (DDT) was the first of a long line of chlorinated hydrocarbon insecticides (figure \(\PageIndex{n}\)). These compounds are chains of carbon and hydrogen with chlorine atoms replacing some of the hydrogen atoms. Introduced during World War II, DDT, along with penicillin and the sulfa drugs, was responsible for the fact that this was the first war in history where trauma killed more people - combatants and noncombatants alike - than infectious disease.
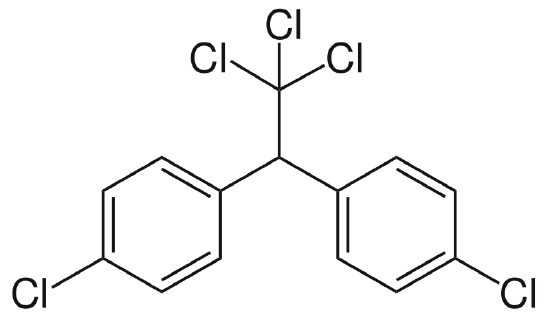

Dichlorodiphenyltrichloroethane is effective against many crop pests as well as vectors of human diseases such as the mosquitoes that spread malaria and yellow fever and fleas, which transmit the plague. Prior to the introduction of DDT, the number of cases of malaria in Ceylon (now Sri Lanka) was more than a million a year. By 1963 the disease had been practically eliminated from the island. However, growing concern about the hazards of DDT led to its abandonment there in the mid-1960s, and soon thereafter malaria became common once again. Because it remains in the environment and is resistant to breakdown, DDT was especially effective against malarial mosquitoes. One or two sprays a year on the walls of homes kept them free of mosquitoes. It was also inexpensive, further adding to its appeal, but DDT has several serious drawbacks.
Because DDT builds up in fatty tissues (bioaccumulation) and becomes more concentrated at the highest levels of the food chain (biomagnification), it is especially harmful to apex predators, such as Bald Eagles (figure \(\PageIndex{o}\)). Classical studies documenting these effects were described in the 1960s bestseller Silent Spring by Rachel Carson. It was discovered that DDT caused the eggshells of birds to become fragile and break, making reproduction impossible. As a result, the Bald Eagle was listed as an endangered species under U.S. law. After DDT was banned in the United States in 1972, affected bird populations made noticeable recoveries, including the iconic Bald Eagle.

Physical Hazards
Physical hazards are additional forces that can imperil humans. Physical hazards may arise naturally such as natural disasters (earthquakes, wildfires, landslides, etc.) or extreme weather (figure \(\PageIndex{p}\)). Others may arise from human structures or activities (traffic accident, building collapse, injury from mechanical equipment, strain on the body from repeated movements, etc.) Some physical hazards, such as explosions or radiation, can arise from natural or human sources.

Radiation is energy given off by matter in the form of rays or high-speed particles, and some types of radiation present a physical hazard. Some familiar forms of radiation are infrared radiation (heat), visible light, ultraviolet (UV) light, radio waves, and microwaves. We are exposed to radiation every day from natural sources. For example, the sun exposes us to UV radiation. We are also exposed to radiation from human-made sources like medical X-rays and smoke detectors. We’re even exposed to low levels of radiation on cross-country flights, from watching television, and even from some construction materials. Some types of radioactive materials are more dangerous than others. Specifically, ionizing radiation, like X rays and gamma rays (one of the forms of radiation emitted from nuclear fuel and waste), have enough energy to break molecular bonds and displace (or remove) electrons from atoms.
Attribution
Modified by Melissa Ha from the following sources:
- Environmental Biology by Mathew R. Fisher (licensed under CC-BY)
- Insecticides from Biology by John W. Kimball (licensed under CC-BY)
- Impact on Human Health from AP Environmental Science by University of California College Prep (licensed under CC-BY)
- Environmental Health at a glance. 2003. World Bank. Washington, DC. (licensed under CC-BY)
- Asbestos. 2018. Environment Protection Agency. Accessed 01-07-2021. (public domain)
- Per- and Polyfluoroalkyl Substances (PFAS). 2018. Environment Protection Agency. Accessed 01-07-2021. (public domain)
- Polychlorinated Biphenyls (PCBs). 2020. Environment Protection Agency. Accessed 01-08-2021. (public domain)
- What are DALYs? National Institute of Mental Health. Accessed 01-06-2021. (public domain)
- Radiation Basics. 2020. U.S. NRC. Accessed 01-07-2021 (public domain)
- Phthalates. 2017. National Biomonitoring Program. Centers for Disease Control. Accessed 01-08-2021. (public domain)


