19.7B: Translation
- Page ID
- 3435
\( \newcommand{\vecs}[1]{\overset { \scriptstyle \rightharpoonup} {\mathbf{#1}} } \)
\( \newcommand{\vecd}[1]{\overset{-\!-\!\rightharpoonup}{\vphantom{a}\smash {#1}}} \)
\( \newcommand{\id}{\mathrm{id}}\) \( \newcommand{\Span}{\mathrm{span}}\)
( \newcommand{\kernel}{\mathrm{null}\,}\) \( \newcommand{\range}{\mathrm{range}\,}\)
\( \newcommand{\RealPart}{\mathrm{Re}}\) \( \newcommand{\ImaginaryPart}{\mathrm{Im}}\)
\( \newcommand{\Argument}{\mathrm{Arg}}\) \( \newcommand{\norm}[1]{\| #1 \|}\)
\( \newcommand{\inner}[2]{\langle #1, #2 \rangle}\)
\( \newcommand{\Span}{\mathrm{span}}\)
\( \newcommand{\id}{\mathrm{id}}\)
\( \newcommand{\Span}{\mathrm{span}}\)
\( \newcommand{\kernel}{\mathrm{null}\,}\)
\( \newcommand{\range}{\mathrm{range}\,}\)
\( \newcommand{\RealPart}{\mathrm{Re}}\)
\( \newcommand{\ImaginaryPart}{\mathrm{Im}}\)
\( \newcommand{\Argument}{\mathrm{Arg}}\)
\( \newcommand{\norm}[1]{\| #1 \|}\)
\( \newcommand{\inner}[2]{\langle #1, #2 \rangle}\)
\( \newcommand{\Span}{\mathrm{span}}\) \( \newcommand{\AA}{\unicode[.8,0]{x212B}}\)
\( \newcommand{\vectorA}[1]{\vec{#1}} % arrow\)
\( \newcommand{\vectorAt}[1]{\vec{\text{#1}}} % arrow\)
\( \newcommand{\vectorB}[1]{\overset { \scriptstyle \rightharpoonup} {\mathbf{#1}} } \)
\( \newcommand{\vectorC}[1]{\textbf{#1}} \)
\( \newcommand{\vectorD}[1]{\overrightarrow{#1}} \)
\( \newcommand{\vectorDt}[1]{\overrightarrow{\text{#1}}} \)
\( \newcommand{\vectE}[1]{\overset{-\!-\!\rightharpoonup}{\vphantom{a}\smash{\mathbf {#1}}}} \)
\( \newcommand{\vecs}[1]{\overset { \scriptstyle \rightharpoonup} {\mathbf{#1}} } \)
\( \newcommand{\vecd}[1]{\overset{-\!-\!\rightharpoonup}{\vphantom{a}\smash {#1}}} \)
Define translation. Briefly describethe function of the following in terms of translation: - 30S ribosomal subunit
- ribosome binding site
- start codon
- initiation complex
- 50S ribosomal subunit
- tRNA
- aminoacyl-tRNA
- anticodon
- P-site of ribosome
- A-site of ribosome
- E-site of ribosome
- peptidyl transferase
- nonsense (stop) codon
- release factors
- 30S ribosomal subunit
- ribosome binding site
- start codon
- initiation complex
- 50S ribosomal subunit
- tRNA
- aminoacyl-tRNA
- anticodon
- P-site of ribosome
- A-site of ribosome
- E-site of ribosome
- peptidyl transferase
- nonsense (stop) codon
- release factors

A 50S ribosomal subunit then attaches to the initiation complex and the initiation factors leave. This forms the 70S ribosome. (see Figure \(\PageIndex{4}\)).
The joining of individual amino acids to form a protein or polypeptide is known as the elongation phase of translation. There are three sites on the 70S ribosome. The A or acceptor or aminoacyl site is where an aminoacyl-tRNA first attaches. The P or peptide site is where a tRNA is temporarily holding the growing amino acid chain as the next codon in the mRNA is being read. The E or exit site is where the uncharged tRNA that has released its amino acid exits the ribosome. During peptide bond formation, the amino acid chain or peptide moves from the tRNA at the P-site and forms a peptide bond with the new amino acid attached to the tRNA at the A-site. The peptide bond is formed by a ribozyme , an enzyme composed of the 23S rRNA itself, called peptidyl transferase. The now uncharged tRNA at the P-site leaves the ribosome through the E-site to eventually pick up a new amino acid and be recycled. Meanwhile, the 70S ribosome moves a distance of one codon down the mRNA through a process called translocation to allow decoding of the next codon in the message (see Figure \(\PageIndex{5}\)A - 5F). The growing polypeptide chain actually passes through a tunnel in the 50S ribosomal subunit.
This process continues over and over again in the 5' to 3' direction until the ribosome hits a stop codon. A stop codon is a series of three mRNA bases coding for no amino acid and thus terminates the protein chain. UAA, UAG, UGA are the three stop codons in the genetic code. Stop codons do not code for an amino acid because a tRNA cannot recognize them.
Proteins called release factors free the protein from the tRNA and the two ribosomal subunits come apart to be recycled (see Figure \(\PageIndex{5}\)F). During this elongation process, the protein has assumed its three-dimensional functional shape. Proteins called chaperonins assist in the protein folding.
Once the ribosome is clear of the ribosome binding site and the AUG start codon, another 30S ribosomal subunit attaches to the ribosome binding site of the mRNA to initiate another round of translation. In this way, multiple copies of a protein can be produced from a single molecule of mRNA. A mRNA with multiple ribosomes attached is known as a polyribosome or polysome .
Summary
- During translation, specific tRNAs pick up specific amino acids, transfer those amino acids to the ribosomes, and insert them in their proper place according to the mRNA genetic "message."
- This is done by the anticodon portion of the tRNA molecules complementary base pairing with the codons along the mRNA.
- Transfer RNA (tRNA) is a three-dimensional, inverted cloverleaf-shaped molecule of RNA.
- At the top, or 3' end, a specific amino acid can be attached to a specific tRNA by means of specific enzymes called aminoacyl-tRNA synthetases.
- At the bottom loop of the cloverleaf is a series of three unpaired tRNA bases called the anticodon. An anticodon is a series of three tRNA bases complementary to a mRNA codon.
- The anticodons of some tRNAs are able to recognize more than one codon because the tRNA's recognition of the third nucleotide of the codon is not always precise, however, the right amino acid is still inserted because there are 61 codons that code for the 22 different amino acids.
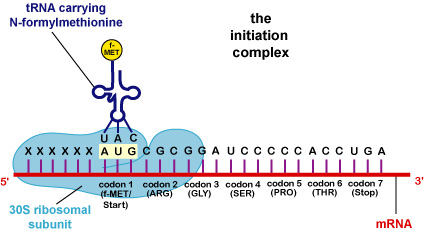
- To initiate translation in prokaryotic cells, a 30S ribosomal subunit binds to a short nucleotide sequence on the mRNA called the ribosome binding site.
- AUG is known as the start codon. At this point, an initiation complex composed of the 30S subunit, a tRNA having the anticodon UAC and carrying an altered form of the amino acid methionine (N-formylmethionine or f-Met), and proteins called initiation factors is formed. A 50S ribosomal subunit then attaches to the initiation complex and the initiation factors leave. This forms the 70S ribosome.
- The A or acceptor or aminoacyl site of the ribosome is where an aminoacyl-tRNA first attaches.
- The P or peptide site of the ribosome is where a tRNA is temporarily holding the growing amino acid chain as the next codon in the mRNA is being read.
- The E or exit site of the ribosome is where the uncharged tRNA that has released its amino acid exits the ribosome.
- During peptide bond formation, the amino acid chain or peptide moves from the tRNA at the P-site and forms a peptide bond with the new amino acid attached to the tRNA at the A-site.
- A stop codon is a series of three mRNA bases coding for no amino acid and thus terminates the protein chain. UAA, UAG, UGA are the three stop codons in the genetic code. (Stop codons do not code for an amino acid because a tRNA cannot recognize them.)
- A mRNA with multiple ribosomes attached is known as a polyribosome or polysome.