15.11.8: Circadian Rhythms in Drosophila and Mammals
- Page ID
- 5770
All eukaryotes and some microbes (e.g., cyanobacteria) display changes in gene activity, biochemistry, physiology, and behavior that wax and wane through the cycle of days and nights.
Examples:
- The level of the hormone melatonin that rises in your body during the night and falls during the day.
- Fruit flies (Drosophila melanogaster) hatch in greatest numbers just at dawn.
Even when the organism is placed in constant conditions (e.g., continuous darkness), these rhythms persist. However, without environmental cues, they tend to be somewhat longer or somewhat shorter than 24 hours giving rise to the name circadian rhythms (L. circa = about; dies = day).
The genetics and molecular biology of circadian rhythms have been studied in several model organisms including
- some unicellular eukaryotes
- fungi
- plants (Arabidopsis)
- invertebrates (Drosophila)
- mammals (mice, rats, and humans)
What has emerged are some remarkable similarities in mechanisms across these various groups. Let us take a detailed look at the mechanism in Drosophila.
The Circadian Clock in Drosophila
A number of genes in Drosophila are turned on when the animal is exposed to light:
- effector genes whose products mediate the animal's responses (e.g. hatching or molting)
- clock genes whose products regulate the circadian clock. Two key members of this group are:
- period (per)
- timeless (tim)
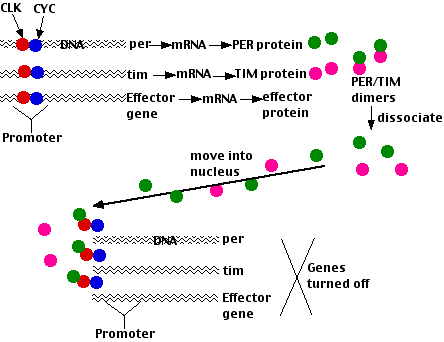
Activation of all of these genes requires that their promoters are bound by the protein transcription factors
- CLOCK encoded by the gene clock (clk)
- CYCLE encoded by the gene cycle (cyc)
(The names of proteins will be designated with capitalized Roman letters; the genes that encode them indicated in lower case italics.)
The Mechanism
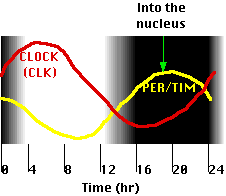
Fig.15.11.8.2 Mechanism
- The PER and TIM proteins (synthesized on ribosomes in the cytoplasm) form dimers.
- When the concentration of these gets high enough (early evening), they dissociate and are transported into the nucleus.
- Here PER
- binds to the CLK/CYC transcription factors, removing them from the promoters of the genes they activate; thus shutting off transcription. Because these genes include per and tim, the result is a negative feedback loop; that is, the product of the per gene inhibit its own synthesis (as well as that of tim). Just as the heat of a furnace turns through the thermostat - its own production off, so a rising level of PER/TIM dimers turns off further synthesis of them. As the level then falls, this inhibition is lifted and PER/TIM activity begins anew.
- turns on clock gene expression.
- The time required for the different effects results in the levels of PER/TIM and CLOCK oscillating in opposite phases with a circadian (~24 hr) rhythm (figure).
Setting the Clock
Even without any external cues (e.g., alternating light and dark), the cycles persist although they tend to drift away from environmental time.
- Under natural conditions, the clocks are precise.
- This is because they are "set" (synchronized) by environmental cues, of which light is one of the most important.
In Drosophila, it works like this.
- Light (blue) is absorbed by the protein cryptochrome (CRY).
- This causes an allosteric change in its conformation enabling it to bind to TIM and PER.
- This causes TIM and PER to break down (in proteasomes) ending their inhibition of gene transcription.
- If this happens when PER/TIM levels are rising (late in the "day"), it sets the clock back.
- If it happens when PER/TIM levels are declining (late in the "night"), it sets the clock ahead.
The Circadian Clock in Mammals
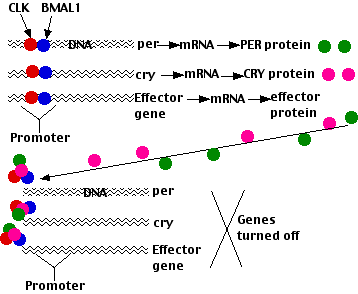
The circadian clock in mammals resembles that in Drosophila in a number of ways with many of the participating genes being homologous. However, there are some differences:
- The transcription factors that turn on the light-induced promoters are dimers of the CLOCK protein and a protein designated BMAL1. These dimers turn on
- three Per genes
- two Cry genes, the genes encoding cryptochrome
- hundreds of effector genes whose products control a wide variety of metabolic functions (e.g., cellular respiration, glycolysis, gluconeogenesis, lipid metabolism)
- The PER and CRY mRNAs are exported to the cytoplasm where they are translated.
- The PER and CRY proteins then enter the nucleus where they inhibit CLOCK-BMAL1 thus turning OFF transcription of Per and Cry, and are then degraded in proteasomes.
In due course these actions allow CLOCK and BMAL1 to once again stimulate transcription of Per and Cry. Thus this negative feedback loop causes the levels of BMAL1 and PER/CRY to oscillate in opposite phases (as CLOCK and PER/TIM do in Drosophila).
Many tissues in mammals, e.g., liver, skeletal muscle, and the beta cells of the pancreas have endogenous clocks. But all of these are under the control of a "master clock", the suprachiasmatic nucleus (SCN) - clusters of neurons in the hypothalamus.
The blood levels of many hormones have strong circadian rhythms. Examples:
- hormones synthesized in the hypothalamus, e.g. vasopressin
- whose secretion is controlled by the hypothalamus such as growth hormone and cortisol
- insulin
Setting the Clock
By light
Mice who are totally blind (lacking both rods and cones) have no trouble keeping their circadian clock on time.
They are able to do this because
- Some 1–2% of the ganglion cells in their retina - instead of depending on signals arriving from rods and/or cones detect light directly.
- These ganglion cells have an extensive network of dendrites that contain the pigment melanopsin.
- When exposed to light (diffuse light is fine), these ganglion cells become depolarized and send their signals back to the suprachiasmatic nucleus (SCN).
By food
In mice, the SCN clock, set by light/dark cycles, is the master clock as long as food is available all the time (the normal situation in the laboratory). However, if food is offered for only a 4-hour period when the mice would normally be asleep, they shift several circadian activities so that, for example, once a day they begin running about just before they expect food to be given to them. This rhythm continues even if the mice are kept in constant darkness.
The clock mechanism is the same as the light/dark-driven clock in the SCN, but the machinery that sets the clock by food is located in a different part of the hypothalamus, the dorsomedial hypothalamic nuclei (DMH).
Mice with both copies of the Bmal1 gene knocked out, are unable to establish circadian rhythms to either light or food. However, injections of an adeno-associated virus vector (AAV) containing the Bmal1 gene
- into the SCN restores the light clock but not the food-set clock
- into the DMH restores the food but not the light-set clock.
Sleep Disorders
Unlike mice, people who are totally blind cannot set the clock in their SCN. As a result, their circadian rhythm drifts out of phase with the actual cycle of day and night. These people often are bothered by feeling sleepy during the day and wide awake when they want to be asleep at night. A report in the 12 October 2000 issue of the New England Journal of Medicine tells of a group of blind people who were able to set their clocks with the help of a dose (10 mg) of melatonin at bedtime. However, this treatment worked only when the subject's circadian rhythm had drifted so that the normal rise in melatonin from the pineal gland was occurring in the early evening; that is, the dose of melatonin had to be given when it could boost the endogenous level of the hormone.
Some people suffer from a disorder called familial advanced sleep-phase syndrome (FASPS). As the name suggests, it is inherited ("familial") and their circadian clocks run fast ("advanced"). Those afflicted tend to wake up several (up to four) hours earlier than normal.
One cause of the disorder turns out to be a point mutation in the human PER2 gene. Exactly how this mutation causes shortening of the circadian cycle is still under investigation.
Photoperiodism
Many plants and animals not only engage in a cycle of daily activities (opening of flowers, waking, feeding, etc.) but also in seasonal activities.
- In plants, such things as production of flowers and making buds dormant in preparation for winter.
- In animals, such things as preparing to migrate and entering and leaving hibernation.
The most reliable clue to the change of season is length of day (temperature is far less reliable!). The farther a plant or animal lives north or south of the equator, the more pronounced the changing ratio of daytime hours to nighttime hours with the changing seasons.
It is not surprising then that both plants and animals mainly depend on photoperiod to prepare for changes in seasonal activities. And what better way to measure the relative length of day and night than by enlisting the machinery by which circadian rhythms are entrained?
As for animals, recent work with Drosophila suggests that this animal uses two circadian clocks to monitor the changing length of day and night.
- An "evening" clock that takes over in the long days of summer.
- A"morning" clock that is inhibited by light but takes over when the nights are getting longer;
The molecular machinery (Cry, Tim, Per, etc.) for each clock is confined to separate neurons in two different parts of the brain.
In these experiments, Drosophila is using the two clocks to adapt daily — not seasonal — cycles of activity to the changing seasons. But this machinery for measuring photoperiod could enable them to prepare for seasonal changes in activity, e.g., to stop forming eggs at the end of the summer. However, other studies examining such seasonal changes in Drosophila find that the photoperiodic response is independent of circadian responses. So we must await more experiments to resolve the question.


