11.3: Gene Therapy - Methods and Prospects
- Page ID
- 4939
\( \newcommand{\vecs}[1]{\overset { \scriptstyle \rightharpoonup} {\mathbf{#1}} } \)
\( \newcommand{\vecd}[1]{\overset{-\!-\!\rightharpoonup}{\vphantom{a}\smash {#1}}} \)
\( \newcommand{\dsum}{\displaystyle\sum\limits} \)
\( \newcommand{\dint}{\displaystyle\int\limits} \)
\( \newcommand{\dlim}{\displaystyle\lim\limits} \)
\( \newcommand{\id}{\mathrm{id}}\) \( \newcommand{\Span}{\mathrm{span}}\)
( \newcommand{\kernel}{\mathrm{null}\,}\) \( \newcommand{\range}{\mathrm{range}\,}\)
\( \newcommand{\RealPart}{\mathrm{Re}}\) \( \newcommand{\ImaginaryPart}{\mathrm{Im}}\)
\( \newcommand{\Argument}{\mathrm{Arg}}\) \( \newcommand{\norm}[1]{\| #1 \|}\)
\( \newcommand{\inner}[2]{\langle #1, #2 \rangle}\)
\( \newcommand{\Span}{\mathrm{span}}\)
\( \newcommand{\id}{\mathrm{id}}\)
\( \newcommand{\Span}{\mathrm{span}}\)
\( \newcommand{\kernel}{\mathrm{null}\,}\)
\( \newcommand{\range}{\mathrm{range}\,}\)
\( \newcommand{\RealPart}{\mathrm{Re}}\)
\( \newcommand{\ImaginaryPart}{\mathrm{Im}}\)
\( \newcommand{\Argument}{\mathrm{Arg}}\)
\( \newcommand{\norm}[1]{\| #1 \|}\)
\( \newcommand{\inner}[2]{\langle #1, #2 \rangle}\)
\( \newcommand{\Span}{\mathrm{span}}\) \( \newcommand{\AA}{\unicode[.8,0]{x212B}}\)
\( \newcommand{\vectorA}[1]{\vec{#1}} % arrow\)
\( \newcommand{\vectorAt}[1]{\vec{\text{#1}}} % arrow\)
\( \newcommand{\vectorB}[1]{\overset { \scriptstyle \rightharpoonup} {\mathbf{#1}} } \)
\( \newcommand{\vectorC}[1]{\textbf{#1}} \)
\( \newcommand{\vectorD}[1]{\overrightarrow{#1}} \)
\( \newcommand{\vectorDt}[1]{\overrightarrow{\text{#1}}} \)
\( \newcommand{\vectE}[1]{\overset{-\!-\!\rightharpoonup}{\vphantom{a}\smash{\mathbf {#1}}}} \)
\( \newcommand{\vecs}[1]{\overset { \scriptstyle \rightharpoonup} {\mathbf{#1}} } \)
\(\newcommand{\longvect}{\overrightarrow}\)
\( \newcommand{\vecd}[1]{\overset{-\!-\!\rightharpoonup}{\vphantom{a}\smash {#1}}} \)
\(\newcommand{\avec}{\mathbf a}\) \(\newcommand{\bvec}{\mathbf b}\) \(\newcommand{\cvec}{\mathbf c}\) \(\newcommand{\dvec}{\mathbf d}\) \(\newcommand{\dtil}{\widetilde{\mathbf d}}\) \(\newcommand{\evec}{\mathbf e}\) \(\newcommand{\fvec}{\mathbf f}\) \(\newcommand{\nvec}{\mathbf n}\) \(\newcommand{\pvec}{\mathbf p}\) \(\newcommand{\qvec}{\mathbf q}\) \(\newcommand{\svec}{\mathbf s}\) \(\newcommand{\tvec}{\mathbf t}\) \(\newcommand{\uvec}{\mathbf u}\) \(\newcommand{\vvec}{\mathbf v}\) \(\newcommand{\wvec}{\mathbf w}\) \(\newcommand{\xvec}{\mathbf x}\) \(\newcommand{\yvec}{\mathbf y}\) \(\newcommand{\zvec}{\mathbf z}\) \(\newcommand{\rvec}{\mathbf r}\) \(\newcommand{\mvec}{\mathbf m}\) \(\newcommand{\zerovec}{\mathbf 0}\) \(\newcommand{\onevec}{\mathbf 1}\) \(\newcommand{\real}{\mathbb R}\) \(\newcommand{\twovec}[2]{\left[\begin{array}{r}#1 \\ #2 \end{array}\right]}\) \(\newcommand{\ctwovec}[2]{\left[\begin{array}{c}#1 \\ #2 \end{array}\right]}\) \(\newcommand{\threevec}[3]{\left[\begin{array}{r}#1 \\ #2 \\ #3 \end{array}\right]}\) \(\newcommand{\cthreevec}[3]{\left[\begin{array}{c}#1 \\ #2 \\ #3 \end{array}\right]}\) \(\newcommand{\fourvec}[4]{\left[\begin{array}{r}#1 \\ #2 \\ #3 \\ #4 \end{array}\right]}\) \(\newcommand{\cfourvec}[4]{\left[\begin{array}{c}#1 \\ #2 \\ #3 \\ #4 \end{array}\right]}\) \(\newcommand{\fivevec}[5]{\left[\begin{array}{r}#1 \\ #2 \\ #3 \\ #4 \\ #5 \\ \end{array}\right]}\) \(\newcommand{\cfivevec}[5]{\left[\begin{array}{c}#1 \\ #2 \\ #3 \\ #4 \\ #5 \\ \end{array}\right]}\) \(\newcommand{\mattwo}[4]{\left[\begin{array}{rr}#1 \amp #2 \\ #3 \amp #4 \\ \end{array}\right]}\) \(\newcommand{\laspan}[1]{\text{Span}\{#1\}}\) \(\newcommand{\bcal}{\cal B}\) \(\newcommand{\ccal}{\cal C}\) \(\newcommand{\scal}{\cal S}\) \(\newcommand{\wcal}{\cal W}\) \(\newcommand{\ecal}{\cal E}\) \(\newcommand{\coords}[2]{\left\{#1\right\}_{#2}}\) \(\newcommand{\gray}[1]{\color{gray}{#1}}\) \(\newcommand{\lgray}[1]{\color{lightgray}{#1}}\) \(\newcommand{\rank}{\operatorname{rank}}\) \(\newcommand{\row}{\text{Row}}\) \(\newcommand{\col}{\text{Col}}\) \(\renewcommand{\row}{\text{Row}}\) \(\newcommand{\nul}{\text{Nul}}\) \(\newcommand{\var}{\text{Var}}\) \(\newcommand{\corr}{\text{corr}}\) \(\newcommand{\len}[1]{\left|#1\right|}\) \(\newcommand{\bbar}{\overline{\bvec}}\) \(\newcommand{\bhat}{\widehat{\bvec}}\) \(\newcommand{\bperp}{\bvec^\perp}\) \(\newcommand{\xhat}{\widehat{\xvec}}\) \(\newcommand{\vhat}{\widehat{\vvec}}\) \(\newcommand{\uhat}{\widehat{\uvec}}\) \(\newcommand{\what}{\widehat{\wvec}}\) \(\newcommand{\Sighat}{\widehat{\Sigma}}\) \(\newcommand{\lt}{<}\) \(\newcommand{\gt}{>}\) \(\newcommand{\amp}{&}\) \(\definecolor{fillinmathshade}{gray}{0.9}\)Many human diseases are caused by defective genes. A few common examples are tabulate in Table \(\PageIndex{1}\); all of these diseases are caused by a defect at a single gene locus. (The inheritance is recessive so both the maternal and paternal copies of the gene must be defective.) Is there any hope of introducing functioning genes into these patients to correct their disorder? Probably. Other diseases also have a genetic basis, but it appears that several genes must act in concert to produce the disease phenotype. The prospects of gene therapy in these cases seems far more remote.
| Disease | Genetic defect |
|---|---|
| hemophilia A | absence of clotting factor VIII |
| cystic fibrosis | defective chloride channel protein |
| muscular dystrophy | defective muscle protein (dystrophin) |
| sickle-cell disease | defective beta globin |
| hemophilia B | absence of clotting factor IX |
| severe combined immunodeficiency (SCID) | any one of several genes fail to make a protein essential for T and B cell function |
SCID is a disease in which the patient has neither cell-mediated immune responses nor is able to make antibodies. It is a disease of young children because, until recently, the absence of an immune system left them prey to infections that ultimately killed them. About 25% of the cases of SCID are the result of the child being homozygous for a defective gene encoding the enzyme adenosine deaminase (ADA). The normal catabolism of purines is deficient, and this is particularly toxic for T cells and B cells.
Treatment Options for SCID include:
- Raise the child in a strictly germfree environment: all food, water, and air to be sterilized. David, the "bubble boy" from Houston, survived this way until he was 12 years old.
- Give the child a transplant of bone marrow from a normal, histocompatible, donor. Ideally, this would give the child a continuous source of ADA+ T and B cells. However, even though the child cannot reject the transplant (the child has no immune system), T cells in the transplant (unless the donor was an identical twin) can attack the cells of the child producing graft-versus-host disease. Moreover, the donor cells may be infected with a virus which could overwhelm the recipient before his or her immune system was restored. (David received a bone marrow transplant from his sister, but she, like many people, had been infected earlier with the Epstein-Barr virus (the cause of "mono")). The virus was still present in the cells she donated, and killed her brother.
- Give injections of ADA (the enzyme is currently extracted from cows). When conjugated with polyethylene glycol (PEG) to delay its breakdown in the blood, ADA-PEG injections have kept SCID patients reasonably healthy. But just like the insulin injections of a diabetic, they must be repeated at frequent intervals. So,
- Giving the patient functioning ADA genes - gene therapy
Gene Therapy Requirements
The gene must be identified and cloned. This has been done for the ADA gene. It must be inserted in cells that can take up long-term residence in the patient. So far, this means removing the patient's own cells, treating them in tissue culture, and then returning them to the patient. It must be inserted in the DNA so that it will be expressed adequately; that is, transcribed and translated with sufficient efficiency that worthwhile amounts of the enzyme are produced. All these requirements seem to have been met for SCID therapy using a retrovirus as the gene vector. Retroviruses have several advantages for introducing genes into human cells.


- Their envelope protein enables the virus to infect human cells.
- RNA copies of the human ADA gene can be incorporated into the retroviral genome using a packaging cell.
Packaging cells are treated so they express an RNA copy of the human ADA gene along with a packaging signal (P) needed for the assembly of fresh virus particles. They also needs inverted repeats ("R") at each end to aid insertion of the DNA copies into the DNA of the target cell. They need an RNA copy of the retroviral gag, pol, and env genes but with no packaging signal (so these genes cannot be incorporated in fresh viral particles).
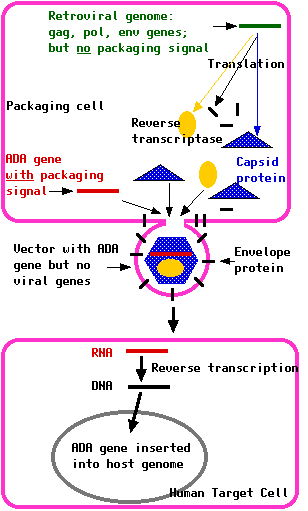
Treated with these two genomes, the packaging cell produces a crop of retroviruses with:
- the envelope protein needed to infect the human target cells
- an RNA copy of the human ADA gene, complete with R sequences at each end
- reverse transcriptase, needed to make a DNA copy of the ADA gene that can be inserted into the DNA of the target cell
- none of the genes (gag, pol, env) that would enable the virus to replicate in its new host.
Once the virus has infected the target cells, this RNA is reverse transcribed into DNA and inserted into the chromosomal DNA of the host.
Target Cells: T cells
The first attempts at gene therapy for SCID children (in 1990), used their own T cells (produced following ADA-PEG therapy) as the target cells. The T cells were:
- placed in tissue culture
- stimulated to proliferate (by treating them with the lymphokine, Interleukin 2 (IL-2)
- infected with the retroviral vector
- returned, in a series of treatments, to the child
The children developed improved immune function but the injections had to be repeated because T cells live for only 6–12 months in the blood. Moreover, the children also continued to receive ADA-PEG so the actual benefit of the gene therapy was unclear
Target Cells: Stem cells
Blood ("hematopoietic") stem cells:
- produce (by mitosis) all the types of blood cells, including T and B lymphocytes
- produce (by mitosis) more stem cells, thus ensuring an inexhaustible supply
In June of 2002, a team of Italian and Israeli doctors reported on two young SCID patients that were treated with their own blood stem cells that had been transformed in vitro with a retroviral vector carrying the ADA gene. After a year, both children had fully-functioning immune systems (T, B, and NK cells) and were able to live normal lives without any need for treatment with ADA-PEG or immune globulin (IG). The doctors attribute their success to first destroying some of the bone marrow cells of their patients to "make room" for the transformed cells. Nine years later (August 2011) these two patients are still thriving and have been joined by 28 other successfully-treated children most of whom no longer need to take ADA-PEG.
Gene therapy has also succeeded for 20 baby boys who suffered from another form of severe combined immunodeficiency called X-linked SCID because it is caused by a mutated X-linked gene encoding a subunit — called γc (gamma-c) — of the receptor for several interleukins, including interleukin-7 (IL-7). IL-7 is essential for converting blood stem cells into the progenitors of T cells. Boys with X-linked SCID can make normal B cells, but because B cells need T-helper cells to function, these boys could make neither cell-mediated nor antibody-mediated immune responses and had to live in a sterile bubble before their treatment.
Their doctors
- isolated blood stem cells from the bone marrow of each infant
- treated the cells with a retroviral vector containing the normal gene for the γc interleukin receptor subunit
- returned the treated cells to each donor
The results: Now after as long as 11 years, 19 of these boys
- are able to live normal lives at home instead of inside a sterile "bubble"
- have normal (with some exceptions*) numbers of T cells of both the CD4 and CD8 subsets
- have responded to several childhood immunizations, including diphtheria, tetanus and polio by producing both T cells and antibodies specific for these agents
- Antibody production is sufficiently good that most of the boys have no need for periodic infusions of immune globulin (IG)
Five of the little boys developed leukemia (one has died):
- in one case caused by a proliferating clone of γδ T cells in which the vector has inserted itself in a gene (on chromosome 11) implicated in some cases of acute lymphoblastic leukemia (ALL)
- in a second case, the leukemia was of αβ T cells
Gene Therapy for β-thalassemia
β-thalassemia is an inherited disease. The most severe cases result from mutations in both copies of the gene encoding the beta chain of hemoglobin. Many causative mutations have been identified, and most lead to a failure to make any beta chains. The resulting hemoglobin functions poorly and the person requires frequent blood transfusions. In 2010, Cavazzana-Calvo (and many colleagues) report a single case of successful gene therapy for this disorder; their patient was an 18-year old male. Their procedure involve harvesting blood stem cells from the patient and exposing him to a retroviral vector that contained
- a human gene for beta-hemoglobin complete with its promoter, enhancer, and other control elements;
- alterations to the vector to make it safe.
- After sufficient chemotherapy to "make room" for them, the patient was injected with these cells.
The result: Almost three years later, the patient is well and no longer requires periodic blood transfusions. One-third of his hemoglobin is now manufactured by the red-cell precursors descended from the gene-altered stem cells. A similar procedure was used on several babies born with an inherited lysosomal storage disease or Wiskott-Aldrich syndrome (another type of immune deficiency). Up to two years after treatment with a retroviral vector containing the intact gene, these babies shown any signs of their disorders (reported in the 23 August 2013 issue of Science).
Adenovirus Vectors
Adenoviruses are human pathogens responsible for some cases of the human "cold". Modified versions of two strains are currently being used as vectors in gene therapy trials. Advantages of adenovirusus: Unlike retroviral vectors, they do not integrate into the host genome and thus should not be able to disrupt host genes (It was such disruption that caused some X-linked SCID patients treated with a retroviral vector to develop leukemia) and they can infect nondividing cells with high efficiency. Disadvantages are that they elicit a powerful immune response, both by T cells and by B cells (antibodies) so repeated doses soon lose their effectiveness. Moreoer, many people already have antibodies against the virus from earlier "colds", and these can inactivate the vector at the outset. A recent trial of an HIV vaccine using an adenovirus as the vector was halted when it was found not only not to be effective but, in people with preexisting high levels of anti-adenovirus antibodies, may have even increased their susceptibility to HIV.


