15.2: Isolation and identification of Staphylococci
- Page ID
- 122719
\( \newcommand{\vecs}[1]{\overset { \scriptstyle \rightharpoonup} {\mathbf{#1}} } \)
\( \newcommand{\vecd}[1]{\overset{-\!-\!\rightharpoonup}{\vphantom{a}\smash {#1}}} \)
\( \newcommand{\dsum}{\displaystyle\sum\limits} \)
\( \newcommand{\dint}{\displaystyle\int\limits} \)
\( \newcommand{\dlim}{\displaystyle\lim\limits} \)
\( \newcommand{\id}{\mathrm{id}}\) \( \newcommand{\Span}{\mathrm{span}}\)
( \newcommand{\kernel}{\mathrm{null}\,}\) \( \newcommand{\range}{\mathrm{range}\,}\)
\( \newcommand{\RealPart}{\mathrm{Re}}\) \( \newcommand{\ImaginaryPart}{\mathrm{Im}}\)
\( \newcommand{\Argument}{\mathrm{Arg}}\) \( \newcommand{\norm}[1]{\| #1 \|}\)
\( \newcommand{\inner}[2]{\langle #1, #2 \rangle}\)
\( \newcommand{\Span}{\mathrm{span}}\)
\( \newcommand{\id}{\mathrm{id}}\)
\( \newcommand{\Span}{\mathrm{span}}\)
\( \newcommand{\kernel}{\mathrm{null}\,}\)
\( \newcommand{\range}{\mathrm{range}\,}\)
\( \newcommand{\RealPart}{\mathrm{Re}}\)
\( \newcommand{\ImaginaryPart}{\mathrm{Im}}\)
\( \newcommand{\Argument}{\mathrm{Arg}}\)
\( \newcommand{\norm}[1]{\| #1 \|}\)
\( \newcommand{\inner}[2]{\langle #1, #2 \rangle}\)
\( \newcommand{\Span}{\mathrm{span}}\) \( \newcommand{\AA}{\unicode[.8,0]{x212B}}\)
\( \newcommand{\vectorA}[1]{\vec{#1}} % arrow\)
\( \newcommand{\vectorAt}[1]{\vec{\text{#1}}} % arrow\)
\( \newcommand{\vectorB}[1]{\overset { \scriptstyle \rightharpoonup} {\mathbf{#1}} } \)
\( \newcommand{\vectorC}[1]{\textbf{#1}} \)
\( \newcommand{\vectorD}[1]{\overrightarrow{#1}} \)
\( \newcommand{\vectorDt}[1]{\overrightarrow{\text{#1}}} \)
\( \newcommand{\vectE}[1]{\overset{-\!-\!\rightharpoonup}{\vphantom{a}\smash{\mathbf {#1}}}} \)
\( \newcommand{\vecs}[1]{\overset { \scriptstyle \rightharpoonup} {\mathbf{#1}} } \)
\(\newcommand{\longvect}{\overrightarrow}\)
\( \newcommand{\vecd}[1]{\overset{-\!-\!\rightharpoonup}{\vphantom{a}\smash {#1}}} \)
\(\newcommand{\avec}{\mathbf a}\) \(\newcommand{\bvec}{\mathbf b}\) \(\newcommand{\cvec}{\mathbf c}\) \(\newcommand{\dvec}{\mathbf d}\) \(\newcommand{\dtil}{\widetilde{\mathbf d}}\) \(\newcommand{\evec}{\mathbf e}\) \(\newcommand{\fvec}{\mathbf f}\) \(\newcommand{\nvec}{\mathbf n}\) \(\newcommand{\pvec}{\mathbf p}\) \(\newcommand{\qvec}{\mathbf q}\) \(\newcommand{\svec}{\mathbf s}\) \(\newcommand{\tvec}{\mathbf t}\) \(\newcommand{\uvec}{\mathbf u}\) \(\newcommand{\vvec}{\mathbf v}\) \(\newcommand{\wvec}{\mathbf w}\) \(\newcommand{\xvec}{\mathbf x}\) \(\newcommand{\yvec}{\mathbf y}\) \(\newcommand{\zvec}{\mathbf z}\) \(\newcommand{\rvec}{\mathbf r}\) \(\newcommand{\mvec}{\mathbf m}\) \(\newcommand{\zerovec}{\mathbf 0}\) \(\newcommand{\onevec}{\mathbf 1}\) \(\newcommand{\real}{\mathbb R}\) \(\newcommand{\twovec}[2]{\left[\begin{array}{r}#1 \\ #2 \end{array}\right]}\) \(\newcommand{\ctwovec}[2]{\left[\begin{array}{c}#1 \\ #2 \end{array}\right]}\) \(\newcommand{\threevec}[3]{\left[\begin{array}{r}#1 \\ #2 \\ #3 \end{array}\right]}\) \(\newcommand{\cthreevec}[3]{\left[\begin{array}{c}#1 \\ #2 \\ #3 \end{array}\right]}\) \(\newcommand{\fourvec}[4]{\left[\begin{array}{r}#1 \\ #2 \\ #3 \\ #4 \end{array}\right]}\) \(\newcommand{\cfourvec}[4]{\left[\begin{array}{c}#1 \\ #2 \\ #3 \\ #4 \end{array}\right]}\) \(\newcommand{\fivevec}[5]{\left[\begin{array}{r}#1 \\ #2 \\ #3 \\ #4 \\ #5 \\ \end{array}\right]}\) \(\newcommand{\cfivevec}[5]{\left[\begin{array}{c}#1 \\ #2 \\ #3 \\ #4 \\ #5 \\ \end{array}\right]}\) \(\newcommand{\mattwo}[4]{\left[\begin{array}{rr}#1 \amp #2 \\ #3 \amp #4 \\ \end{array}\right]}\) \(\newcommand{\laspan}[1]{\text{Span}\{#1\}}\) \(\newcommand{\bcal}{\cal B}\) \(\newcommand{\ccal}{\cal C}\) \(\newcommand{\scal}{\cal S}\) \(\newcommand{\wcal}{\cal W}\) \(\newcommand{\ecal}{\cal E}\) \(\newcommand{\coords}[2]{\left\{#1\right\}_{#2}}\) \(\newcommand{\gray}[1]{\color{gray}{#1}}\) \(\newcommand{\lgray}[1]{\color{lightgray}{#1}}\) \(\newcommand{\rank}{\operatorname{rank}}\) \(\newcommand{\row}{\text{Row}}\) \(\newcommand{\col}{\text{Col}}\) \(\renewcommand{\row}{\text{Row}}\) \(\newcommand{\nul}{\text{Nul}}\) \(\newcommand{\var}{\text{Var}}\) \(\newcommand{\corr}{\text{corr}}\) \(\newcommand{\len}[1]{\left|#1\right|}\) \(\newcommand{\bbar}{\overline{\bvec}}\) \(\newcommand{\bhat}{\widehat{\bvec}}\) \(\newcommand{\bperp}{\bvec^\perp}\) \(\newcommand{\xhat}{\widehat{\xvec}}\) \(\newcommand{\vhat}{\widehat{\vvec}}\) \(\newcommand{\uhat}{\widehat{\uvec}}\) \(\newcommand{\what}{\widehat{\wvec}}\) \(\newcommand{\Sighat}{\widehat{\Sigma}}\) \(\newcommand{\lt}{<}\) \(\newcommand{\gt}{>}\) \(\newcommand{\amp}{&}\) \(\definecolor{fillinmathshade}{gray}{0.9}\)1. Blood agar with a novobiocin (NB) disc
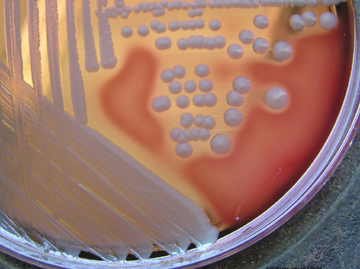
To isolate staphylococci, clinical specimens are usually grown on Blood agar (described in Lab 14). Staphylococci produce round, raised, opaque colonies 1-2mm in diameter. The novobiocin disc is used to detect sensitivity or resistance to the antibiotic novobiocin.
| Test |
Fig. \(\PageIndex{1A}\): Staphylococcus aureus (pigmented strain) on Blood Agar Fig. \(\PageIndex{1B}\): Staphylococcus aureus (non-pigmented strain) on Blood Agar |
Fig. \(\PageIndex{1C}\): Staphylococcus epidermidis on Blood Agar | Fig. \(\PageIndex{1D}\): Staphylococcus saprophyticuson Blood Agar |
|---|---|---|---|
| Hemolysis (*) | Usually beta(1) | Usually gamma(2) | Usually gamma(2) |
| Pigment | Often creamy gold(1) | Usually white(2) | Usually white(2) |
| Novobiocin test | Sensitive | Sensitive | Resistant |
(*) See Lab 14 for descriptions of hemolysis
(1) some strains do not show hemolysis and/or pigment
(2) some strains do show hemolysis and/or pigment
sensitive = zone of inhibition around disc
resistant = no zone of inhibition around disc
|
Fig. \(\PageIndex{1A}\): Staphylococcus aureus (Pigmented Strain) Growing on Blood Agar |
Fig. \(\PageIndex{1B}\): Staphylococcus aureus (Non-pigmented Strain) Growing on Blood Agar |
Fig. \(\PageIndex{1C}\): Staphylococcus epidermidis Growing on Blood Agar |
Fig. \(\PageIndex{1D}\): Staphylococcus saprophyticus Growing on Blood Agar |
|---|---|---|---|
|
|
|
|
|
| Note beta hemolysis (complete lysis of the red blood cells around the colonies; see arrows) on the blood agar and the organism is sensitive to the antibiotic novobiocin in the Taxo NB® disk. | Note the non-pigmented colonies surrounded by a zone of beta hemolysis (complete lysis of the red blood cells around the colonies) on the blood agar. | Note there is no hemolysis (gamma reaction) on the blood agar and the organism is sensitive to the antibiotic novobiocin as shown by the zone of inhibition around the Taxo NB® disk. | Note there is no hemolysis (gamma reaction) on the blood agar and the organism is resistant to the antibiotic novobiocin in the Taxo NB® disk. |
|
Copyright; Gary E. Kaiser, Ph.D. The Community College of Baltimore County, Catonsville Campus CC-BY-3.0 |
Image: Blood agar culture of Staphylococcus aureus © Rebecca Buxton, author. Licensed for use, ASM MicrobeLibrary. |
Copyright; Gary E. Kaiser, Ph.D. The Community College of Baltimore County, Catonsville Campus CC-BY-3.0 |
Copyright; Gary E. Kaiser, Ph.D. The Community College of Baltimore County, Catonsville Campus CC-BY-3.0 |
2. Gram stain
All staphylococci appear as Gram-positive cocci, usually in irregular, often grape-like clusters (see Fig. \(\PageIndex{1}\) above).
3. Mannitol fermentation on Mannitol Salt agar (MSA)
Staphylococci are able to tolerate the high salt concentration found in Mannitol Salt agar and thus grow readily. If mannitol is fermented, the acid produced turns the phenol red pH indicator from red (alkaline) to yellow (acid).
| Test | Fig. \(\PageIndex{2A}\): Staphylococcus aureus Growing on Mannitol Salt Agar | Fig. \(\PageIndex{2B}\): Staphylococcus epidermidis Growing on Mannitol Salt Agar | Fig. \(\PageIndex{2C}\): Staphylococcus saprophyticus Growing on Mannitol Salt Agar |
|---|---|---|---|
| Mannitol fermentation | Positive | Negative | Usually positive |
positive = acid end products turn the phenol red pH indicator from red to yellow
negative = prenol red remains red
|
Fig. \(\PageIndex{2A}\): Staphylooccus aureus Growing on Mannitol Salt Agar. |
Fig. \(\PageIndex{2B}\): Staphylooccus epidermidis Growing on Mannitol Salt Agar. |
Fig. \(\PageIndex{2C}\): Staphylooccus saprophyticus Growing on Mannitol Salt Agar. |
|---|---|---|
|
|
||
| Mannitol salt agar is selective for staphylococci because of the high salt concentration. Acid from mannitol fermentation causes the pH indicator phenol red to turn from red (alkaline) to yellow (acid) | Mannitol salt agar is selective for staphylococci because of the high salt concentration. There is no mannitol fermentation and, therefore, no acid production. The pH indicator phenol red remains red (alkaline) | Mannitol salt agar is selective for staphylococci because of the high salt concentration. Acid from mannitol fermentation causes the pH indicator phenol red to turn from red (alkaline) to yellow (acid) |
| Copyright; Gary E. Kaiser, Ph.D. The Community College of Baltimore County, Catonsville Campus CC-BY-3.0 | ||
4. Production of coagulase
The staphylococcal enzyme coagulase will cause inoculated citrated rabbit plasma to gel or coagulate. The coagulase converts soluble fibrinogen in the plasma into insoluble fibrin.
| Test | Fig. \(\PageIndex{3A}\): Staphylococcus aureusCoagulase Test | Fig. \(\PageIndex{3A}\): Staphylococcus epidermidisCoagulase Test | Fig. \(\PageIndex{3C}\): Staphylococcus saprophyticusCoagulase Test |
|---|---|---|---|
| Coagulase production | Positive | Negative | Negative |
positive = plasma will gel or coagulate
negative = plasma will not gel
|
Fig. \(\PageIndex{3A}\): Positive Coagulase Test for Staphylococcus aureus |
Fig. \(\PageIndex{3B}\): Negative Coagulase Test for Staphylococcus epidermidis |
Fig. \(\PageIndex{3C}\): Negative Coagulase Test for Staphylococcus saprophyticus |
|---|---|---|

|

|

|
|
Rabbit plasma has gelled indicating the bacterium is coagulase positive. |
Rabbit plasma has not gelled indicating the bacterium is coagulase negative. |
Rabbit plasma has not gelled indicating the bacterium is coagulase negative. |
| Copyright; Gary E. Kaiser, Ph.D. The Community College of Baltimore County, Catonsville Campus CC-BY-3.0 | ||
5. The Staphyloslide® Latex Test for cell-bound coagulase (clumping factor) and/or Protein A
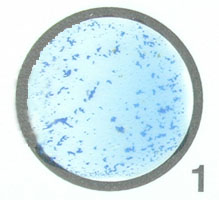
The Staphyloslide® Latex Test is an agglutination test that detects cell-bound coagulase (clumping factor) and/or Protein A. Approximately 97% of human strains of S. aureus possess both bound coagulase and extracellular coagulase. Approximately 95% of human strains of S. aureus possess Protein A on their cell surface (see Fig. 7). This test uses blue latex particles coated with human fibrinogen and the human antibody IgG. Mixing of the latex reagent with colonies of the suspected S. aureus having coagulase and/or Protein A bound to their surface causes agglutination of the latex particles.
| Test | Fig. \(\PageIndex{4A}\): Staphyloside® Latex Test on Staphylococcus aureus | Fig. \(\PageIndex{4B}\): Staphyloside® Latex Test on Staphylococcus epidermidis | Fig. \(\PageIndex{4C}\): Staphyloside® Latex Test on Staphylococcus saprophyticus |
|---|---|---|---|
| Cell-bound coagulase (clumping factor) and/or Protein A | Positive | Negative | Negative |
positive = clumping of latex particles
negative = no clumping of latex particles
|
Fig. \(\PageIndex{4A}\): Staphyloslide® Test on Staphylococcus aureus |
Fig. \(\PageIndex{4B}\): Staphyloslide® Test on Staphylococcus epidermidis |
Fig. \(\PageIndex{4C}\): Staphyloslide® Test on Staphylococcus saprophyticus |
|---|---|---|
|

|

|
|
The blue latex particles coated with human fibrinogen and human IgG have clumped together indicating the bacterium is positive for bound coagulase (clumping factor) and/or Protein A. |
The blue latex particles coated with human fibrinogen and human IgG have not clumped together indicating the bacterium is negative for bound coagulase (clumping factor) and/or Protein A. |
The blue latex particles coated with human fibrinogen and human IgG have not clumped together indicating the bacterium is negative for bound coagulase (clumping factor) and/or Protein A. |
| Copyright; Gary E. Kaiser, Ph.D. The Community College of Baltimore County, Catonsville Campus CC-BY-3.0 | ||
For further information on coagulase and Protein A associated with S. aureus, see the following CourseArc lessons:
Staphylococci are also being identified using chemiluminescent labelled DNA probes complementary to species-specific bacterial ribosomal RNA (rRNA) sequences as well as by other direct DNA techniques.
Contributors and Attributions
Dr. Gary Kaiser (COMMUNITY COLLEGE OF BALTIMORE COUNTY, CATONSVILLE CAMPUS)


