18: Spore Stain
- Page ID
- 3625
\( \newcommand{\vecs}[1]{\overset { \scriptstyle \rightharpoonup} {\mathbf{#1}} } \)
\( \newcommand{\vecd}[1]{\overset{-\!-\!\rightharpoonup}{\vphantom{a}\smash {#1}}} \)
\( \newcommand{\dsum}{\displaystyle\sum\limits} \)
\( \newcommand{\dint}{\displaystyle\int\limits} \)
\( \newcommand{\dlim}{\displaystyle\lim\limits} \)
\( \newcommand{\id}{\mathrm{id}}\) \( \newcommand{\Span}{\mathrm{span}}\)
( \newcommand{\kernel}{\mathrm{null}\,}\) \( \newcommand{\range}{\mathrm{range}\,}\)
\( \newcommand{\RealPart}{\mathrm{Re}}\) \( \newcommand{\ImaginaryPart}{\mathrm{Im}}\)
\( \newcommand{\Argument}{\mathrm{Arg}}\) \( \newcommand{\norm}[1]{\| #1 \|}\)
\( \newcommand{\inner}[2]{\langle #1, #2 \rangle}\)
\( \newcommand{\Span}{\mathrm{span}}\)
\( \newcommand{\id}{\mathrm{id}}\)
\( \newcommand{\Span}{\mathrm{span}}\)
\( \newcommand{\kernel}{\mathrm{null}\,}\)
\( \newcommand{\range}{\mathrm{range}\,}\)
\( \newcommand{\RealPart}{\mathrm{Re}}\)
\( \newcommand{\ImaginaryPart}{\mathrm{Im}}\)
\( \newcommand{\Argument}{\mathrm{Arg}}\)
\( \newcommand{\norm}[1]{\| #1 \|}\)
\( \newcommand{\inner}[2]{\langle #1, #2 \rangle}\)
\( \newcommand{\Span}{\mathrm{span}}\) \( \newcommand{\AA}{\unicode[.8,0]{x212B}}\)
\( \newcommand{\vectorA}[1]{\vec{#1}} % arrow\)
\( \newcommand{\vectorAt}[1]{\vec{\text{#1}}} % arrow\)
\( \newcommand{\vectorB}[1]{\overset { \scriptstyle \rightharpoonup} {\mathbf{#1}} } \)
\( \newcommand{\vectorC}[1]{\textbf{#1}} \)
\( \newcommand{\vectorD}[1]{\overrightarrow{#1}} \)
\( \newcommand{\vectorDt}[1]{\overrightarrow{\text{#1}}} \)
\( \newcommand{\vectE}[1]{\overset{-\!-\!\rightharpoonup}{\vphantom{a}\smash{\mathbf {#1}}}} \)
\( \newcommand{\vecs}[1]{\overset { \scriptstyle \rightharpoonup} {\mathbf{#1}} } \)
\(\newcommand{\longvect}{\overrightarrow}\)
\( \newcommand{\vecd}[1]{\overset{-\!-\!\rightharpoonup}{\vphantom{a}\smash {#1}}} \)
\(\newcommand{\avec}{\mathbf a}\) \(\newcommand{\bvec}{\mathbf b}\) \(\newcommand{\cvec}{\mathbf c}\) \(\newcommand{\dvec}{\mathbf d}\) \(\newcommand{\dtil}{\widetilde{\mathbf d}}\) \(\newcommand{\evec}{\mathbf e}\) \(\newcommand{\fvec}{\mathbf f}\) \(\newcommand{\nvec}{\mathbf n}\) \(\newcommand{\pvec}{\mathbf p}\) \(\newcommand{\qvec}{\mathbf q}\) \(\newcommand{\svec}{\mathbf s}\) \(\newcommand{\tvec}{\mathbf t}\) \(\newcommand{\uvec}{\mathbf u}\) \(\newcommand{\vvec}{\mathbf v}\) \(\newcommand{\wvec}{\mathbf w}\) \(\newcommand{\xvec}{\mathbf x}\) \(\newcommand{\yvec}{\mathbf y}\) \(\newcommand{\zvec}{\mathbf z}\) \(\newcommand{\rvec}{\mathbf r}\) \(\newcommand{\mvec}{\mathbf m}\) \(\newcommand{\zerovec}{\mathbf 0}\) \(\newcommand{\onevec}{\mathbf 1}\) \(\newcommand{\real}{\mathbb R}\) \(\newcommand{\twovec}[2]{\left[\begin{array}{r}#1 \\ #2 \end{array}\right]}\) \(\newcommand{\ctwovec}[2]{\left[\begin{array}{c}#1 \\ #2 \end{array}\right]}\) \(\newcommand{\threevec}[3]{\left[\begin{array}{r}#1 \\ #2 \\ #3 \end{array}\right]}\) \(\newcommand{\cthreevec}[3]{\left[\begin{array}{c}#1 \\ #2 \\ #3 \end{array}\right]}\) \(\newcommand{\fourvec}[4]{\left[\begin{array}{r}#1 \\ #2 \\ #3 \\ #4 \end{array}\right]}\) \(\newcommand{\cfourvec}[4]{\left[\begin{array}{c}#1 \\ #2 \\ #3 \\ #4 \end{array}\right]}\) \(\newcommand{\fivevec}[5]{\left[\begin{array}{r}#1 \\ #2 \\ #3 \\ #4 \\ #5 \\ \end{array}\right]}\) \(\newcommand{\cfivevec}[5]{\left[\begin{array}{c}#1 \\ #2 \\ #3 \\ #4 \\ #5 \\ \end{array}\right]}\) \(\newcommand{\mattwo}[4]{\left[\begin{array}{rr}#1 \amp #2 \\ #3 \amp #4 \\ \end{array}\right]}\) \(\newcommand{\laspan}[1]{\text{Span}\{#1\}}\) \(\newcommand{\bcal}{\cal B}\) \(\newcommand{\ccal}{\cal C}\) \(\newcommand{\scal}{\cal S}\) \(\newcommand{\wcal}{\cal W}\) \(\newcommand{\ecal}{\cal E}\) \(\newcommand{\coords}[2]{\left\{#1\right\}_{#2}}\) \(\newcommand{\gray}[1]{\color{gray}{#1}}\) \(\newcommand{\lgray}[1]{\color{lightgray}{#1}}\) \(\newcommand{\rank}{\operatorname{rank}}\) \(\newcommand{\row}{\text{Row}}\) \(\newcommand{\col}{\text{Col}}\) \(\renewcommand{\row}{\text{Row}}\) \(\newcommand{\nul}{\text{Nul}}\) \(\newcommand{\var}{\text{Var}}\) \(\newcommand{\corr}{\text{corr}}\) \(\newcommand{\len}[1]{\left|#1\right|}\) \(\newcommand{\bbar}{\overline{\bvec}}\) \(\newcommand{\bhat}{\widehat{\bvec}}\) \(\newcommand{\bperp}{\bvec^\perp}\) \(\newcommand{\xhat}{\widehat{\xvec}}\) \(\newcommand{\vhat}{\widehat{\vvec}}\) \(\newcommand{\uhat}{\widehat{\uvec}}\) \(\newcommand{\what}{\widehat{\wvec}}\) \(\newcommand{\Sighat}{\widehat{\Sigma}}\) \(\newcommand{\lt}{<}\) \(\newcommand{\gt}{>}\) \(\newcommand{\amp}{&}\) \(\definecolor{fillinmathshade}{gray}{0.9}\)Objectives
- Learn to perform the spore stain.
- Identify spores on a bacterial smear.
Endospore production is a very important characteristic of some bacteria, allowing them to resist adverse environmental conditions such as desiccation, chemical exposure, extreme heat, etc. They were identified in the 1800s (John Tyndall developed a process for destroying them with intermittent heat procedure), although the stain procedures to identify them did not develop until the early twentieth century. Bacterial endospores are the most resistant structures of all living organisms, and they can live in this dormant dehydrated state for hundreds and hundreds of years (even some documented at thousands of years). Endospores are not for reproduction: One spore forms inside of the vegetative cell. When the spore germinates, one vegetative cell will be produced. The stimulus for sporulation can vary—nutrient depletion, desiccation, chemicals, heat, etc.
As a spore forms inside of the vegetative cell, the spore wall chemically changes and thicken. This sporulation process changes the spore’s stainability, making it increasingly resistant to the staining dyes, and so a gimmick—steaming---enhances the primary dye’s penetration.
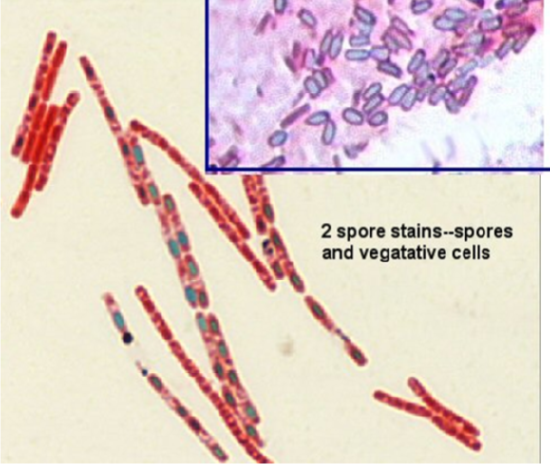
The primary dye malachite green is a relatively weakly binding dye to the cell wall and spore wall. In fact, if washed well with water, the dye comes right out of the cell wall, however not from the spore wall once the dye is locked in. That is why there does not need to be a decolorizer in this stain: it is based on the binding of the malachite green and the permeability of the spore vs. cell wall. . The steaming helps the malachite green to permeate the low-permeability spore wall. A variety of chemicals comprise the spore wall (keratin protein, calcium), but deeper in the wall is peptidoglycan. The keratin forming the outer portion of the endospore wall resists dye. The heating of the bacteria will make the spore wall more permeable to the malachite green, and it then attaches to the peptidoglycan. Once in, the malachite green will not come out because the overlying spore wall becomes less permeable when the smear cools.
The identification of spores is also very important for the clinical microbiologist who is analyzing a patient's body fluid or tissue since there are not that many spore forming genera.
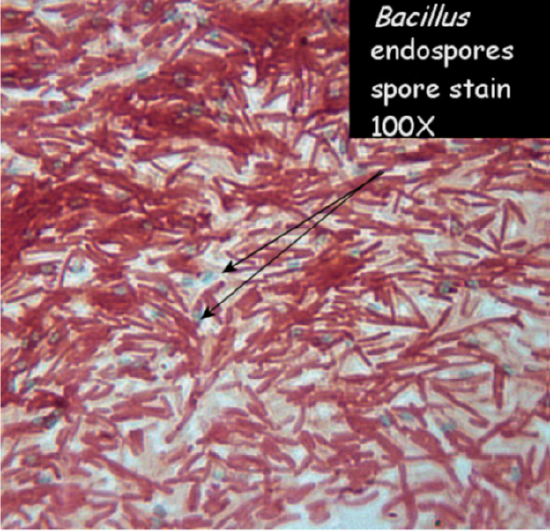
In fact, there are two major pathogenic spore-forming genera, Bacillus and Clostridium, together causing a number of lethal
diseases---botulism, gangrene, tetanus, and anthrax, to name a few. Some bacteria have to be put into unfavorable situations (high cell density and starvation are two key triggers) to go into sporulation; others will make spores easily without much provocation (Bacillus subtilus). Vegetative cells have not yet made spores, may be in the process of making the spore, or will not make them at all. The vegetative cell is metabolically active, whereas the spore is not. Another useful feature is the identification of where the endospore is located within the vegetative cell---terminal, subterminal, or central. A particular species of the genus will form spores in a specific area, producing another useful taxonomic identification tool.
MATERIALS NEEDED
- Reagents: Malachite green, safrinin, acid alcohol
- stain rack
- hot plate
- paper towel (cut the size of the slide)
- Bacillus culture on an slant or plate agar (an old culture to enhance sporulation)
THE PROCEDURE
- Prepare a smear of the Bacillus species---air-dry and heat-fix.
- Put a beaker of water on the hot plate and boil until steam is coming up from the water. Then turn the hot plate down so that the water is barely boiling.
- Place the wire stain rack over the beaker which now has steam coming up from the boiled water.
- Cut a small piece of paper towel and place it on top of the smear on the slide. The towel will keep the dye from evaporating too quickly, thereby giving more contact time between the dye and the bacterial walls.
- Flood the smear with the primary dye, malachite green, and leave for 5 minutes. Keep the paper towel moist with the malachite green. DO NOT let the dye dry on the towel.
- Remove and discard the small paper towel piece.
- Wash really WELL with water and move the slide and wire rack from the boiling water to the regular stain tray to finish up the last step in the procedure.
- Place the smear in the stain jar or flood the smear with the counterstain dye, safrinin, and leave for 1 minute.
- Wash WELL with water. Blot dry with bibulous paper.
- BE SURE TO CLEAN UP THE SINK, REPLACE DYE BOTTLES, PUT AWAY DYE TUBE. CLEAN DIRTY SLIDES AND REPLACE IN THE CLEAN SLIDE BOX FOR YOUR LAB.
Note: Alternative cold method spore stain
Without heat you have to really rough up the spore wall to get in the dye. Heat fix the smear by running the slide through the flame about 20 times, and leave malachite green on for 20 minutes during the stain process.
- Interpret the results using the protocol below.
Interpretation
- Spores will be a light green
- Vegetative cell walls will pick up the counterstain safranin
Notice where the spore is in the cell, if there is one---terminal, central, subterminal. This may be helpful information in the identification of the unknown.
QUESTIONS
- What is the purpose of the steam in this stain?
- Why does there not have to be a decolorizer in this stain?
Contributors and Attributions
Jackie Reynolds, Professor of Biology (Richland College)

