9: Kirby-Bauer (Antibiotic Sensitivity)
- Page ID
- 3483
\( \newcommand{\vecs}[1]{\overset { \scriptstyle \rightharpoonup} {\mathbf{#1}} } \)
\( \newcommand{\vecd}[1]{\overset{-\!-\!\rightharpoonup}{\vphantom{a}\smash {#1}}} \)
\( \newcommand{\dsum}{\displaystyle\sum\limits} \)
\( \newcommand{\dint}{\displaystyle\int\limits} \)
\( \newcommand{\dlim}{\displaystyle\lim\limits} \)
\( \newcommand{\id}{\mathrm{id}}\) \( \newcommand{\Span}{\mathrm{span}}\)
( \newcommand{\kernel}{\mathrm{null}\,}\) \( \newcommand{\range}{\mathrm{range}\,}\)
\( \newcommand{\RealPart}{\mathrm{Re}}\) \( \newcommand{\ImaginaryPart}{\mathrm{Im}}\)
\( \newcommand{\Argument}{\mathrm{Arg}}\) \( \newcommand{\norm}[1]{\| #1 \|}\)
\( \newcommand{\inner}[2]{\langle #1, #2 \rangle}\)
\( \newcommand{\Span}{\mathrm{span}}\)
\( \newcommand{\id}{\mathrm{id}}\)
\( \newcommand{\Span}{\mathrm{span}}\)
\( \newcommand{\kernel}{\mathrm{null}\,}\)
\( \newcommand{\range}{\mathrm{range}\,}\)
\( \newcommand{\RealPart}{\mathrm{Re}}\)
\( \newcommand{\ImaginaryPart}{\mathrm{Im}}\)
\( \newcommand{\Argument}{\mathrm{Arg}}\)
\( \newcommand{\norm}[1]{\| #1 \|}\)
\( \newcommand{\inner}[2]{\langle #1, #2 \rangle}\)
\( \newcommand{\Span}{\mathrm{span}}\) \( \newcommand{\AA}{\unicode[.8,0]{x212B}}\)
\( \newcommand{\vectorA}[1]{\vec{#1}} % arrow\)
\( \newcommand{\vectorAt}[1]{\vec{\text{#1}}} % arrow\)
\( \newcommand{\vectorB}[1]{\overset { \scriptstyle \rightharpoonup} {\mathbf{#1}} } \)
\( \newcommand{\vectorC}[1]{\textbf{#1}} \)
\( \newcommand{\vectorD}[1]{\overrightarrow{#1}} \)
\( \newcommand{\vectorDt}[1]{\overrightarrow{\text{#1}}} \)
\( \newcommand{\vectE}[1]{\overset{-\!-\!\rightharpoonup}{\vphantom{a}\smash{\mathbf {#1}}}} \)
\( \newcommand{\vecs}[1]{\overset { \scriptstyle \rightharpoonup} {\mathbf{#1}} } \)
\(\newcommand{\longvect}{\overrightarrow}\)
\( \newcommand{\vecd}[1]{\overset{-\!-\!\rightharpoonup}{\vphantom{a}\smash {#1}}} \)
\(\newcommand{\avec}{\mathbf a}\) \(\newcommand{\bvec}{\mathbf b}\) \(\newcommand{\cvec}{\mathbf c}\) \(\newcommand{\dvec}{\mathbf d}\) \(\newcommand{\dtil}{\widetilde{\mathbf d}}\) \(\newcommand{\evec}{\mathbf e}\) \(\newcommand{\fvec}{\mathbf f}\) \(\newcommand{\nvec}{\mathbf n}\) \(\newcommand{\pvec}{\mathbf p}\) \(\newcommand{\qvec}{\mathbf q}\) \(\newcommand{\svec}{\mathbf s}\) \(\newcommand{\tvec}{\mathbf t}\) \(\newcommand{\uvec}{\mathbf u}\) \(\newcommand{\vvec}{\mathbf v}\) \(\newcommand{\wvec}{\mathbf w}\) \(\newcommand{\xvec}{\mathbf x}\) \(\newcommand{\yvec}{\mathbf y}\) \(\newcommand{\zvec}{\mathbf z}\) \(\newcommand{\rvec}{\mathbf r}\) \(\newcommand{\mvec}{\mathbf m}\) \(\newcommand{\zerovec}{\mathbf 0}\) \(\newcommand{\onevec}{\mathbf 1}\) \(\newcommand{\real}{\mathbb R}\) \(\newcommand{\twovec}[2]{\left[\begin{array}{r}#1 \\ #2 \end{array}\right]}\) \(\newcommand{\ctwovec}[2]{\left[\begin{array}{c}#1 \\ #2 \end{array}\right]}\) \(\newcommand{\threevec}[3]{\left[\begin{array}{r}#1 \\ #2 \\ #3 \end{array}\right]}\) \(\newcommand{\cthreevec}[3]{\left[\begin{array}{c}#1 \\ #2 \\ #3 \end{array}\right]}\) \(\newcommand{\fourvec}[4]{\left[\begin{array}{r}#1 \\ #2 \\ #3 \\ #4 \end{array}\right]}\) \(\newcommand{\cfourvec}[4]{\left[\begin{array}{c}#1 \\ #2 \\ #3 \\ #4 \end{array}\right]}\) \(\newcommand{\fivevec}[5]{\left[\begin{array}{r}#1 \\ #2 \\ #3 \\ #4 \\ #5 \\ \end{array}\right]}\) \(\newcommand{\cfivevec}[5]{\left[\begin{array}{c}#1 \\ #2 \\ #3 \\ #4 \\ #5 \\ \end{array}\right]}\) \(\newcommand{\mattwo}[4]{\left[\begin{array}{rr}#1 \amp #2 \\ #3 \amp #4 \\ \end{array}\right]}\) \(\newcommand{\laspan}[1]{\text{Span}\{#1\}}\) \(\newcommand{\bcal}{\cal B}\) \(\newcommand{\ccal}{\cal C}\) \(\newcommand{\scal}{\cal S}\) \(\newcommand{\wcal}{\cal W}\) \(\newcommand{\ecal}{\cal E}\) \(\newcommand{\coords}[2]{\left\{#1\right\}_{#2}}\) \(\newcommand{\gray}[1]{\color{gray}{#1}}\) \(\newcommand{\lgray}[1]{\color{lightgray}{#1}}\) \(\newcommand{\rank}{\operatorname{rank}}\) \(\newcommand{\row}{\text{Row}}\) \(\newcommand{\col}{\text{Col}}\) \(\renewcommand{\row}{\text{Row}}\) \(\newcommand{\nul}{\text{Nul}}\) \(\newcommand{\var}{\text{Var}}\) \(\newcommand{\corr}{\text{corr}}\) \(\newcommand{\len}[1]{\left|#1\right|}\) \(\newcommand{\bbar}{\overline{\bvec}}\) \(\newcommand{\bhat}{\widehat{\bvec}}\) \(\newcommand{\bperp}{\bvec^\perp}\) \(\newcommand{\xhat}{\widehat{\xvec}}\) \(\newcommand{\vhat}{\widehat{\vvec}}\) \(\newcommand{\uhat}{\widehat{\uvec}}\) \(\newcommand{\what}{\widehat{\wvec}}\) \(\newcommand{\Sighat}{\widehat{\Sigma}}\) \(\newcommand{\lt}{<}\) \(\newcommand{\gt}{>}\) \(\newcommand{\amp}{&}\) \(\definecolor{fillinmathshade}{gray}{0.9}\)Learning Objectives
- Determine the susceptibility of various bacterial species to various antibiotics and synthetic agents.
A true antibiotic is an antimicrobial chemical produced by microorganisms against other microorganisms. Mankind has made very good use of these antimicrobials in its fight against infectious disease. Many drugs are now completely synthetic or the natural drug is manipulated to change its structure somewhat, the latter called semisynthetics. Bacteria respond in different ways to antibiotics and chemosynthetic drugs, even within the same species. For example, Staphylococcus aureus is a common normal flora bacterium found in the body. If one isolated this bacterium from 5 different people, the 5 isolates would likely be different strains, that is, slight genetically different. It is also likely that if antibiotic sensitivity tests were run on these isolates, the results would vary against the different antibiotics used.
The Kirby-Bauer test for antibiotic susceptibility (also called the disc diffusion test) is a standard that has been used for years. First developed in the 1950s, it was refined and by W. Kirby and A. Bauer, then standardized by the World Health Organization in 1961. It has been superseded in clinical labs by automated tests. However, the K-B is still used in some labs, or used with certain bacteria that automation does not work well with. This test is used to determine the resistance or sensitivity of aerobes or facultative anaerobes to specific chemicals, which can then be used by the clinician for treatment of patients with bacterial infections. The presence or absence of an inhibitory area around the disc identifies the bacterial sensitivity to the drug (Figure \(\PageIndex{1}\)).
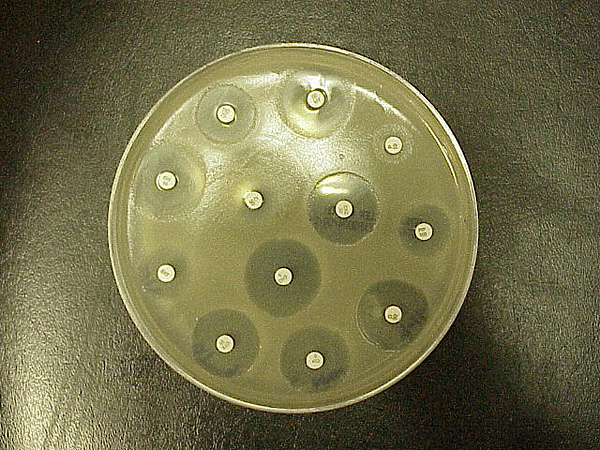
The basics are easy: The bacterium is swabbed on the agar and the antibiotic discs are placed on top. The antibiotic diffuses from the disc into the agar in decreasing amounts the further it is away from the disc. If the organism is killed or inhibited by the concentration of the antibiotic, there will be NO growth in the immediate area around the disc: This is called the zone of inhibition (Figure 9.1). The zone sizes are looked up on a standardized chart to give a result of sensitive, resistant, or intermediate. Many charts have a corresponding column that also gives the MIC (minimal inhibitory concentration) for that drug. The MIC is currently the standard test run for antibiotic sensitivity testing because it produces more pertinent information on minimal dosages.
The Mueller-Hinton medium being used for the Kirby-Bauer test is very high in protein.
MATERIALS NEEDED: per table
- 2 Mueller-Hinton agar plates
- 24 hr old cultures (likely to be Staph, E. coli, Bacillus subtilus, Enterococcus fecalis) sterile swabs
- antibiotics
- ethanol
- forceps
- Pseudomonas aeruginosa Kirby-Bauer plate for demo
- guidelines chart for interpretation of antibiotic susceptibility
THE PROCEDURES
2nd swab coverage ►
- Swab a Mueller-Hinton plate with ONLY 2 of the bacteria (tables will run different combinations of the 4 bacteria). Dip a sterile swab into the broth and express any excess moisture by pressing the swab against the side of the tube.
- Swab the surface of the agar completely (you do not want to leave any unswabbed agar areas at all). In the pictures below, you can see what happens when the plate is not swabbed correctly with even coverage of the bacterium over the entire agar.
- After completely swabbing the plate, turn it 90 degrees and repeat the swabbing process. (It is not necessary to re-moisten the swab.) Run the swab around the circumference of the plate before discarding it in the discard bag.
- Allow the surface to dry for about 5 minutes before placing antibiotic disks on the agar.
- THE ANTIBIOTIC DISKS:
- You are using individual antibiotic dispensers.
 You will probably have to use a pair of forceps to remove an antibiotic disc from the dispenser: the forceps have to be sterile. Place the forceps in alcohol, flame the forceps until they catch on fire, let the flame go out----sterile forceps.
You will probably have to use a pair of forceps to remove an antibiotic disc from the dispenser: the forceps have to be sterile. Place the forceps in alcohol, flame the forceps until they catch on fire, let the flame go out----sterile forceps.- Lightly touch each disc with your sterile inoculating loop to make sure that it is in good contact with the agar surface. Incubate upside down and incubate at 37o C.
INTERPRETATION
 Place the metric ruler across the zone of inhibition, at the widest diameter, and measure from one edge of the zone to the other edge. HOLDING THE PLATE UP TO THE LIGHT MIGHT HELP.
Place the metric ruler across the zone of inhibition, at the widest diameter, and measure from one edge of the zone to the other edge. HOLDING THE PLATE UP TO THE LIGHT MIGHT HELP.- Use millimeter measurements. The disc diameter will actually be part of that number.
- If there is NO zone at all, report it as 0---even though the disc itself is around 7 mm.
- Zone diameter is reported in millimeters, looked up on the chart, and result reported as sensitive, resistant, or intermediate.
- Record the results for your table, as well as other tables, in the table.
|
Antibiotic (Antimicrobial Agent) |
DISC CODE |
Resistant (< or = mm) |
Intermediate (mm) |
Susceptible (= or > mm) |
|---|---|---|---|---|
| Amoxicillin (other) | AMC | <13 | 14-17 | >>18 |
| Amoxicillin (Staph) | AMC | 19 | >20 | |
| Ampicillin (other) | AM | 11 | 12-13 | >14 |
| Ampicillin (Staph) | AM | 28 | >29 | |
| Carbenicillin (other) | CB | 17 | 18-22 | >23 |
| Carbenicillin (Pseudomonas) | CB | 13 | 14-16 | >17 |
| Cefoxitin | FOX | 14 | 15-17 | >18 |
| Cephalothin | CF | 14 | 15-17 | >18 |
| Chloramphenicol | C | 12 | 13-17 | >18 |
| Ciprofloxacin | CIP-5 | 15 | 16-20 | >21 |
| Clindamycin | CC-2 | 14 | 15-20 | >21 |
| Enoxacin (Fluoroquinolone, 2nd gen.) | ENX-10 | 14 | 15-17 | >18 |
| Erthromycin | E | 13 | 14-22 | >23 |
| Gentamycin | GM | 12 | 13-14 | >15 |
| Kanamycin | K-30 | 13 | 14-17 | >18 |
| Methicillin (Staph) | M(orDP) | 9 | 10-13 | >14 |
| Oxacillin (Staph) | OX | 10 | 11-12 | >13 |
| Penicillin G (Enterococcus) | P | 14 | >15 | |
| Penicillin G (Staph) | P | 28 | >29 | |
| Streptomycin | S-10 | 14 | 15-20 | >21 |
| Sulfamethoxazole-trimethoprim | SXT | 10 | 11-15 | >16 |
| Tetracycline | Te-30 | 14 | 15-18 | >19 |
| Tobramycin | NN-10 | 12 | 13-14 | >15 |
| Vancomycin | Va-30 | 9 | 10-11 | >12 |
TABLE FOR RESULTS
Record the results for the 5 bacteria with all of the antibiotics.
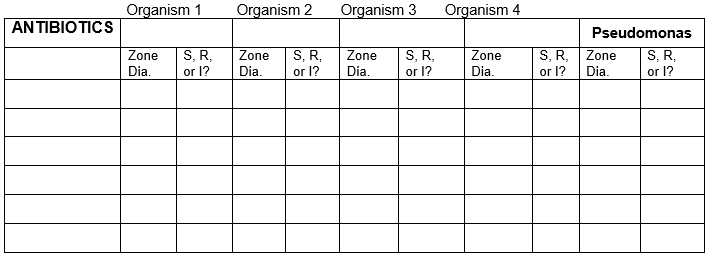
zone diameter in millimeter units. Use chart for sensitive, resistant, or intermediate
QUESTIONS
- The larger the zone size, the more ____________ the bacterium is to that antibiotic.
- What measurement units are used to measure the zone sizes?
- How does the sensitivity of the Staph compare with the sensitivity of the Pseudomonas?


