3: Dilution Techniques and Pipetting
- Page ID
- 3253
\( \newcommand{\vecs}[1]{\overset { \scriptstyle \rightharpoonup} {\mathbf{#1}} } \)
\( \newcommand{\vecd}[1]{\overset{-\!-\!\rightharpoonup}{\vphantom{a}\smash {#1}}} \)
\( \newcommand{\dsum}{\displaystyle\sum\limits} \)
\( \newcommand{\dint}{\displaystyle\int\limits} \)
\( \newcommand{\dlim}{\displaystyle\lim\limits} \)
\( \newcommand{\id}{\mathrm{id}}\) \( \newcommand{\Span}{\mathrm{span}}\)
( \newcommand{\kernel}{\mathrm{null}\,}\) \( \newcommand{\range}{\mathrm{range}\,}\)
\( \newcommand{\RealPart}{\mathrm{Re}}\) \( \newcommand{\ImaginaryPart}{\mathrm{Im}}\)
\( \newcommand{\Argument}{\mathrm{Arg}}\) \( \newcommand{\norm}[1]{\| #1 \|}\)
\( \newcommand{\inner}[2]{\langle #1, #2 \rangle}\)
\( \newcommand{\Span}{\mathrm{span}}\)
\( \newcommand{\id}{\mathrm{id}}\)
\( \newcommand{\Span}{\mathrm{span}}\)
\( \newcommand{\kernel}{\mathrm{null}\,}\)
\( \newcommand{\range}{\mathrm{range}\,}\)
\( \newcommand{\RealPart}{\mathrm{Re}}\)
\( \newcommand{\ImaginaryPart}{\mathrm{Im}}\)
\( \newcommand{\Argument}{\mathrm{Arg}}\)
\( \newcommand{\norm}[1]{\| #1 \|}\)
\( \newcommand{\inner}[2]{\langle #1, #2 \rangle}\)
\( \newcommand{\Span}{\mathrm{span}}\) \( \newcommand{\AA}{\unicode[.8,0]{x212B}}\)
\( \newcommand{\vectorA}[1]{\vec{#1}} % arrow\)
\( \newcommand{\vectorAt}[1]{\vec{\text{#1}}} % arrow\)
\( \newcommand{\vectorB}[1]{\overset { \scriptstyle \rightharpoonup} {\mathbf{#1}} } \)
\( \newcommand{\vectorC}[1]{\textbf{#1}} \)
\( \newcommand{\vectorD}[1]{\overrightarrow{#1}} \)
\( \newcommand{\vectorDt}[1]{\overrightarrow{\text{#1}}} \)
\( \newcommand{\vectE}[1]{\overset{-\!-\!\rightharpoonup}{\vphantom{a}\smash{\mathbf {#1}}}} \)
\( \newcommand{\vecs}[1]{\overset { \scriptstyle \rightharpoonup} {\mathbf{#1}} } \)
\(\newcommand{\longvect}{\overrightarrow}\)
\( \newcommand{\vecd}[1]{\overset{-\!-\!\rightharpoonup}{\vphantom{a}\smash {#1}}} \)
\(\newcommand{\avec}{\mathbf a}\) \(\newcommand{\bvec}{\mathbf b}\) \(\newcommand{\cvec}{\mathbf c}\) \(\newcommand{\dvec}{\mathbf d}\) \(\newcommand{\dtil}{\widetilde{\mathbf d}}\) \(\newcommand{\evec}{\mathbf e}\) \(\newcommand{\fvec}{\mathbf f}\) \(\newcommand{\nvec}{\mathbf n}\) \(\newcommand{\pvec}{\mathbf p}\) \(\newcommand{\qvec}{\mathbf q}\) \(\newcommand{\svec}{\mathbf s}\) \(\newcommand{\tvec}{\mathbf t}\) \(\newcommand{\uvec}{\mathbf u}\) \(\newcommand{\vvec}{\mathbf v}\) \(\newcommand{\wvec}{\mathbf w}\) \(\newcommand{\xvec}{\mathbf x}\) \(\newcommand{\yvec}{\mathbf y}\) \(\newcommand{\zvec}{\mathbf z}\) \(\newcommand{\rvec}{\mathbf r}\) \(\newcommand{\mvec}{\mathbf m}\) \(\newcommand{\zerovec}{\mathbf 0}\) \(\newcommand{\onevec}{\mathbf 1}\) \(\newcommand{\real}{\mathbb R}\) \(\newcommand{\twovec}[2]{\left[\begin{array}{r}#1 \\ #2 \end{array}\right]}\) \(\newcommand{\ctwovec}[2]{\left[\begin{array}{c}#1 \\ #2 \end{array}\right]}\) \(\newcommand{\threevec}[3]{\left[\begin{array}{r}#1 \\ #2 \\ #3 \end{array}\right]}\) \(\newcommand{\cthreevec}[3]{\left[\begin{array}{c}#1 \\ #2 \\ #3 \end{array}\right]}\) \(\newcommand{\fourvec}[4]{\left[\begin{array}{r}#1 \\ #2 \\ #3 \\ #4 \end{array}\right]}\) \(\newcommand{\cfourvec}[4]{\left[\begin{array}{c}#1 \\ #2 \\ #3 \\ #4 \end{array}\right]}\) \(\newcommand{\fivevec}[5]{\left[\begin{array}{r}#1 \\ #2 \\ #3 \\ #4 \\ #5 \\ \end{array}\right]}\) \(\newcommand{\cfivevec}[5]{\left[\begin{array}{c}#1 \\ #2 \\ #3 \\ #4 \\ #5 \\ \end{array}\right]}\) \(\newcommand{\mattwo}[4]{\left[\begin{array}{rr}#1 \amp #2 \\ #3 \amp #4 \\ \end{array}\right]}\) \(\newcommand{\laspan}[1]{\text{Span}\{#1\}}\) \(\newcommand{\bcal}{\cal B}\) \(\newcommand{\ccal}{\cal C}\) \(\newcommand{\scal}{\cal S}\) \(\newcommand{\wcal}{\cal W}\) \(\newcommand{\ecal}{\cal E}\) \(\newcommand{\coords}[2]{\left\{#1\right\}_{#2}}\) \(\newcommand{\gray}[1]{\color{gray}{#1}}\) \(\newcommand{\lgray}[1]{\color{lightgray}{#1}}\) \(\newcommand{\rank}{\operatorname{rank}}\) \(\newcommand{\row}{\text{Row}}\) \(\newcommand{\col}{\text{Col}}\) \(\renewcommand{\row}{\text{Row}}\) \(\newcommand{\nul}{\text{Nul}}\) \(\newcommand{\var}{\text{Var}}\) \(\newcommand{\corr}{\text{corr}}\) \(\newcommand{\len}[1]{\left|#1\right|}\) \(\newcommand{\bbar}{\overline{\bvec}}\) \(\newcommand{\bhat}{\widehat{\bvec}}\) \(\newcommand{\bperp}{\bvec^\perp}\) \(\newcommand{\xhat}{\widehat{\xvec}}\) \(\newcommand{\vhat}{\widehat{\vvec}}\) \(\newcommand{\uhat}{\widehat{\uvec}}\) \(\newcommand{\what}{\widehat{\wvec}}\) \(\newcommand{\Sighat}{\widehat{\Sigma}}\) \(\newcommand{\lt}{<}\) \(\newcommand{\gt}{>}\) \(\newcommand{\amp}{&}\) \(\definecolor{fillinmathshade}{gray}{0.9}\)Learning Objectives
- Become proficient with the use of the pipette.
- Identify different ways to dilute.
- Learn how to solve a dilution problem.
Dilutions are used many times during the semester in the microbiology lab, for a variety of purposes. Therefore, it is important that each person understand how to use the pipette, how to read the pipette accurately, and how to determine what dilution was produced. It is a common practice to determine bacterial counts for both liquid and solid specimens--suspensions of E. coli in nutrient broth all the way to soil samples and hamburger meat. The following protocol is a step-by-step procedure to working dilution problems, and includes some practice problems at the end. The purpose can be determination of bacterial, fungal, or viral counts (commonly called colony - forming units, CFUs, for bacterial or fungal counts, or plaque - forming units, PFUs, for viral counts).
USE OF THE PIPETTE
Fig. 1 Fig. 2 Fig. 3
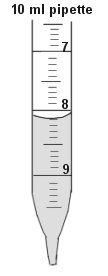
- Be SURE that you know how to read the lines on the various sizes of pipettes. Remember that the reading is taken at the bottom of the meniscus. (the reading on the pipet in picture is at 8.1ml. Refer to Fig. 1)
- Use the smallest volume pipet for the volume you are pipetting
- Place the end of the pipette straight into the opening on the pi-pump (green pi-pump for 5 and 10ml, blue pi-pump for 1m. Refer to Fig. 2 and Fig. 3l).
- Place the pipette tip into the solution; rotate the pi-pump wheel so that the fluid ascends.
- Slowly move the wheel so that you correctly deliver the required amount.
Note
In this little exercise, no aseptic technique is required, but accuracy is all important. You will be starting out with a methylene blue-colored water and diluting it in various ways. If done correctly, the last tube of each of the 3 dilution sets will be the EXACT shade of blue. If not, you have inaccurately diluted the samples---either pipetting inaccurately or adding water to the tubes inaccurately.
MATERIALS NEEDED
- flask of blue water
- nonsterile pipettes - 1, 5, and 10ml
- 6 nonsterile test tubes
- blue and green pi-pumps
THE PROCEDURE
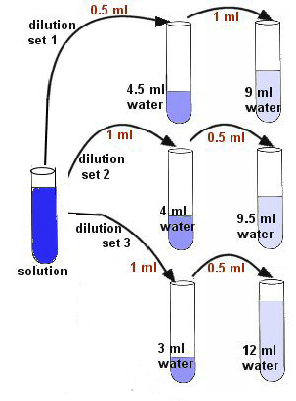
Fig. 4
- You will need a bottle of blue dye.
- Prepare the following dilution blanks with tap water using a 10ml pipette and the green pi-pump---4.5ml, 9ml, 9.5ml, 4ml, 3ml, 12ml.
- Make 3 sets of dilution tubes as seen in the Fig. 4. Mix the tube contents.
- Dilution set 1: Transfer 0.5ml of blue water into the 4.5ml of water, then 1ml of tube 1 into the next tube of 9ml water.
- Dilution set 2: Transfer 1ml of blue water into the 4ml of water, then 0.5ml of tube 1 into the next tube of 9.5ml.
- Dilution set 3: Transfer 1ml of blue water into the 3ml of water, then 0.5ml of tube 1 into the next tube of 12ml.
4. Interpretation of your dilution tubes--
- Record the dilution values of your tubes in the below.
- Check the last tubes of each set against each other to make sure that they are the same shade of blue.
Definition
dilution factor = amount of sample/( amount of sample + amount in tube)
total dilution factor = previous dilution of tube X dilution of next container
Example: dilution set 1
0.5 ml added to 4.5ml = 0.5/5 = 5/50 = 1/10 for 1st tube
1ml added to 9ml = 1/10 (2nd tube) X previous dilution of 1/10(1st tube) = total dilution of 1/100
DILUTION SET 1: The total dilution of tube 1 is 1/10, and for tube 2 it is 1/100.
DILUTION SET 2: The total dilution of tube 1 is ________, and for tube 2 it is __________.
DILUTION SET 3: The total dilution of tube 1 is ________, and for tube 2 it is __________.
The last tube of each of the 3 sets should come out to be the SAME total dilution.
QUESTIONS
 What is the meniscus?
What is the meniscus?- If you transfer 0.1ml of a sample into a 99.9ml saline blank, what is the dilution?
- How much fluid is IN the pipette at right (Fig. 5)?
- For the dilution tubes with colored water, fill in the following values
SET 1 TOTAL DILUTION
tube 1 1/10
tube 2 1/100
SET 2 TOTAL DILUTION
tube 1 _______
tube 2 _______
SET 3 TOTAL DILUTION
tube 1 _______ Fig. 5
tube 2 _______
Contributors
Jackie Reynolds, Professor of Biology (Richland College)