10.10: Antiviral Agents
- Page ID
- 3248
\( \newcommand{\vecs}[1]{\overset { \scriptstyle \rightharpoonup} {\mathbf{#1}} } \)
\( \newcommand{\vecd}[1]{\overset{-\!-\!\rightharpoonup}{\vphantom{a}\smash {#1}}} \)
\( \newcommand{\dsum}{\displaystyle\sum\limits} \)
\( \newcommand{\dint}{\displaystyle\int\limits} \)
\( \newcommand{\dlim}{\displaystyle\lim\limits} \)
\( \newcommand{\id}{\mathrm{id}}\) \( \newcommand{\Span}{\mathrm{span}}\)
( \newcommand{\kernel}{\mathrm{null}\,}\) \( \newcommand{\range}{\mathrm{range}\,}\)
\( \newcommand{\RealPart}{\mathrm{Re}}\) \( \newcommand{\ImaginaryPart}{\mathrm{Im}}\)
\( \newcommand{\Argument}{\mathrm{Arg}}\) \( \newcommand{\norm}[1]{\| #1 \|}\)
\( \newcommand{\inner}[2]{\langle #1, #2 \rangle}\)
\( \newcommand{\Span}{\mathrm{span}}\)
\( \newcommand{\id}{\mathrm{id}}\)
\( \newcommand{\Span}{\mathrm{span}}\)
\( \newcommand{\kernel}{\mathrm{null}\,}\)
\( \newcommand{\range}{\mathrm{range}\,}\)
\( \newcommand{\RealPart}{\mathrm{Re}}\)
\( \newcommand{\ImaginaryPart}{\mathrm{Im}}\)
\( \newcommand{\Argument}{\mathrm{Arg}}\)
\( \newcommand{\norm}[1]{\| #1 \|}\)
\( \newcommand{\inner}[2]{\langle #1, #2 \rangle}\)
\( \newcommand{\Span}{\mathrm{span}}\) \( \newcommand{\AA}{\unicode[.8,0]{x212B}}\)
\( \newcommand{\vectorA}[1]{\vec{#1}} % arrow\)
\( \newcommand{\vectorAt}[1]{\vec{\text{#1}}} % arrow\)
\( \newcommand{\vectorB}[1]{\overset { \scriptstyle \rightharpoonup} {\mathbf{#1}} } \)
\( \newcommand{\vectorC}[1]{\textbf{#1}} \)
\( \newcommand{\vectorD}[1]{\overrightarrow{#1}} \)
\( \newcommand{\vectorDt}[1]{\overrightarrow{\text{#1}}} \)
\( \newcommand{\vectE}[1]{\overset{-\!-\!\rightharpoonup}{\vphantom{a}\smash{\mathbf {#1}}}} \)
\( \newcommand{\vecs}[1]{\overset { \scriptstyle \rightharpoonup} {\mathbf{#1}} } \)
\(\newcommand{\longvect}{\overrightarrow}\)
\( \newcommand{\vecd}[1]{\overset{-\!-\!\rightharpoonup}{\vphantom{a}\smash {#1}}} \)
\(\newcommand{\avec}{\mathbf a}\) \(\newcommand{\bvec}{\mathbf b}\) \(\newcommand{\cvec}{\mathbf c}\) \(\newcommand{\dvec}{\mathbf d}\) \(\newcommand{\dtil}{\widetilde{\mathbf d}}\) \(\newcommand{\evec}{\mathbf e}\) \(\newcommand{\fvec}{\mathbf f}\) \(\newcommand{\nvec}{\mathbf n}\) \(\newcommand{\pvec}{\mathbf p}\) \(\newcommand{\qvec}{\mathbf q}\) \(\newcommand{\svec}{\mathbf s}\) \(\newcommand{\tvec}{\mathbf t}\) \(\newcommand{\uvec}{\mathbf u}\) \(\newcommand{\vvec}{\mathbf v}\) \(\newcommand{\wvec}{\mathbf w}\) \(\newcommand{\xvec}{\mathbf x}\) \(\newcommand{\yvec}{\mathbf y}\) \(\newcommand{\zvec}{\mathbf z}\) \(\newcommand{\rvec}{\mathbf r}\) \(\newcommand{\mvec}{\mathbf m}\) \(\newcommand{\zerovec}{\mathbf 0}\) \(\newcommand{\onevec}{\mathbf 1}\) \(\newcommand{\real}{\mathbb R}\) \(\newcommand{\twovec}[2]{\left[\begin{array}{r}#1 \\ #2 \end{array}\right]}\) \(\newcommand{\ctwovec}[2]{\left[\begin{array}{c}#1 \\ #2 \end{array}\right]}\) \(\newcommand{\threevec}[3]{\left[\begin{array}{r}#1 \\ #2 \\ #3 \end{array}\right]}\) \(\newcommand{\cthreevec}[3]{\left[\begin{array}{c}#1 \\ #2 \\ #3 \end{array}\right]}\) \(\newcommand{\fourvec}[4]{\left[\begin{array}{r}#1 \\ #2 \\ #3 \\ #4 \end{array}\right]}\) \(\newcommand{\cfourvec}[4]{\left[\begin{array}{c}#1 \\ #2 \\ #3 \\ #4 \end{array}\right]}\) \(\newcommand{\fivevec}[5]{\left[\begin{array}{r}#1 \\ #2 \\ #3 \\ #4 \\ #5 \\ \end{array}\right]}\) \(\newcommand{\cfivevec}[5]{\left[\begin{array}{c}#1 \\ #2 \\ #3 \\ #4 \\ #5 \\ \end{array}\right]}\) \(\newcommand{\mattwo}[4]{\left[\begin{array}{rr}#1 \amp #2 \\ #3 \amp #4 \\ \end{array}\right]}\) \(\newcommand{\laspan}[1]{\text{Span}\{#1\}}\) \(\newcommand{\bcal}{\cal B}\) \(\newcommand{\ccal}{\cal C}\) \(\newcommand{\scal}{\cal S}\) \(\newcommand{\wcal}{\cal W}\) \(\newcommand{\ecal}{\cal E}\) \(\newcommand{\coords}[2]{\left\{#1\right\}_{#2}}\) \(\newcommand{\gray}[1]{\color{gray}{#1}}\) \(\newcommand{\lgray}[1]{\color{lightgray}{#1}}\) \(\newcommand{\rank}{\operatorname{rank}}\) \(\newcommand{\row}{\text{Row}}\) \(\newcommand{\col}{\text{Col}}\) \(\renewcommand{\row}{\text{Row}}\) \(\newcommand{\nul}{\text{Nul}}\) \(\newcommand{\var}{\text{Var}}\) \(\newcommand{\corr}{\text{corr}}\) \(\newcommand{\len}[1]{\left|#1\right|}\) \(\newcommand{\bbar}{\overline{\bvec}}\) \(\newcommand{\bhat}{\widehat{\bvec}}\) \(\newcommand{\bperp}{\bvec^\perp}\) \(\newcommand{\xhat}{\widehat{\xvec}}\) \(\newcommand{\vhat}{\widehat{\vvec}}\) \(\newcommand{\uhat}{\widehat{\uvec}}\) \(\newcommand{\what}{\widehat{\wvec}}\) \(\newcommand{\Sighat}{\widehat{\Sigma}}\) \(\newcommand{\lt}{<}\) \(\newcommand{\gt}{>}\) \(\newcommand{\amp}{&}\) \(\definecolor{fillinmathshade}{gray}{0.9}\)- State why antibiotics are of no use against viruses and what we must rely on to control viruses.
- State the viruses the following antiviral agents are used against:
- amantadine, rimantidine, zanamivar, and oseltamivir
- acyclovir, famciclovir, penciclovir, and valacyclovir
- foscarnet, gancyclovir, cidofovir, valganciclovir, and fomivirsen
- AZT (ZDV), didanosine, zalcitabine, stavudine, lamivudine, emtricitabine, tenofovir, and abacavir
- nevirapine, delavirdine, and efavirenz
- saquinavir, ritonavir, idinavir, nelfinavir, amprenavir, atazanavir, fosamprenavir, ritonavir
- telaprevir, boceprevir, simeprevir, sofosbuvir
- Compare how the following drugs exhibit their antiviral action against HIV.
- nucleoside reverse transcriptase inhibitors
- protease inhibitors
- entry inhibitors
Since viruses lack the structures and metabolic processes that are altered by common antibiotics, antibiotics are virtually useless in treating viral infections. To date, relatively few antiviral chemotherapeutic agents are available and used to treat just a few limited viruses.
Most of the antiviral agents work by inhibiting viral DNA synthesis. These drugs chemically resemble normal DNA nucleosides, molecules containing deoxyribose and either adenine, guanine, cytosine, or thymine. Viral enzymes then add phosphate groups to these nucleoside analogs to form DNA nucleotide analogs. The DNA nucleotide analogs are then inserted into the growing viral DNA strand in place of a normal nucleotide. Once inserted, however, new nucleotides can't attach and DNA synthesis is stopped. They are selectively toxic because viral polymerases are more prone to incorporate nucleotide analogs into their nucleic acid than are host cell polymerases.
| Antiviral | Brand Name | Use |
|---|---|---|
| amantadine | Symmetrel | used prophylactically against influenza A ) in high-risk individuals. It prevents influenza A viruses from the uncoating step necessary for viral replication. |
| rimantidine | Flumadine | used for treatment and prophylaxis of influenza A. It prevents influenza A viruses from the uncoating step necessary for viral replication. |
| zanamivir: | Relenza | used to limit the duration of influenza A and B infections. It is an inhibitor of the influenza virus surface enzyme called neuraminidase that is needed for release of newly formed influenza viruses from the infected cell. |
| oseltamivir | Tamiflu | used limit the duration of influenza infections. It is an inhibitor of the influenza virus surface enzyme called neuraminidase that is needed for release of newly formed influenza viruses from the infected cell. |
| acyclovir | Zovirax | used against herpes simplex viruses (HSV) to treat genital herpes, mucocutaneous herpes in the immunosuppressed, HSV encephalitis, neonatal herpes, and to reduce the rate of recurrences of genital herpes. It is also used against varicella zoster viruses (VZV) ) to treat shingles. It chemically resembles a normal DNA nucleoside. Once inserted into the growing DNA chain it inhibits further viral DNA replication. |
| trifluridine | Viroptic | used to treat eye infection (keratitis and conjunctivitis) caused by HSV. It chemically resembles a normal DNA nucleoside. Once inserted into the growing DNA chain it inhibits further viral DNA replication. |
| famciclovir | Famvir | used to treat HSV and VZV infections. It chemically resembles a normal DNA nucleoside. Once inserted into the growing DNA chain it inhibits further viral DNA replication. |
| valacyclovir |
Valtrex | used to treat HSV and VZV infections. It chemically resembles a normal DNA nucleoside. Once inserted into the growing DNA chain it inhibits further viral DNA replication. |
| penciclovir | Denavir | used in treating HSV infections. It chemically resembles a normal DNA nucleoside. Once inserted into the growing DNA chain it inhibits further viral DNA replication. |
| gancyclovir | Cytovene; Vitrasert | used in treating severe cytomegalovirus (CMV) infections such as retinitis. It chemically resembles a normal DNA nucleoside. Once inserted into the growing DNA chain it inhibits further viral DNA replication. |
| valganciclovir | Valcyte | used in treating severe CMV infections such as retinitis). It chemically resembles a normal DNA nucleoside. Once inserted into the growing DNA chain it inhibits further viral DNA replication. |
| foscarnet | Foscavir | used in treating severe CMV infections such as retinitis. It chemically resembles a normal DNA nucleoside. Once inserted into the growing DNA chain it inhibits further viral DNA replication. |
| cidofovir | Vistide | used in treating CMV retinitis. It chemically resembles a normal DNA nucleoside. Once inserted into the growing DNA chain it inhibits further viral DNA replication. |
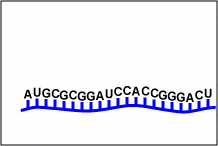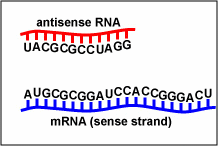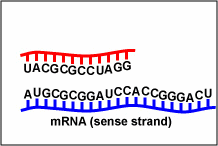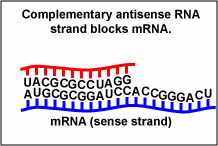
| fomivirsen | Vitravene | used in treating CMV retinitis. Fomivirsen inhibits cytomegalovirus (CMV) replication through an antisense RNA (microRNA or miRNA mechanism. The nucleotide sequence of fomivirsen is complementary to a sequence in mRNA transcripts (Figure \(\PageIndex{1}\)) that encodes several proteins responsible for regulation of viral gene expression that are essential for production of infectious CMV. Binding of fomivirsen to the target mRNA results in inhibition of protein synthesis, subsequently inhibiting virus replication. |
| ribavirin | Copegus; Rebetol; Virazole | used in treating severe acute respiratory syndrome (SARS). In combination with other drugs it is used to treat hepatitis C virus (HCV). It chemically resembles a normal RNA nucleoside. Once inserted into the growing RNA chain it inhibits further viral RNA replication. |
| telaprevir | Incivek | for the treatment of chronic hepatitis C (hepatitis C virus or HCV genotype 1). It is a protease inhibitor that binds to the active site of an HCV-encoded protease and prevent it from cleaving the long polyprotein from polycistronic HCV genes into proteins essential to the structure and function of HCV. |
boceprevir |
Victrelis | for the treatment of chronic hepatitis C (hepatitis C virus or HCV genotype 1) infection. It is used in combination with peginterferon alfa and ribavirin. Boceprevir is a protease inhibitor that binds to the active site of an HCV-encoded protease and prevent it from cleaving the long polyprotein from polycistronic HCV genes into proteins essential to the structure and function of HCV. |
| simeprevir | Olysio | use for the treatment of chronic hepatitis C (hepatitis C virus or HCV genotype 1) infection. Used in combination with peginterferon alfa and ribavirin. Simeprevir is a protease inhibitor that binds to the active site of an HCV-encoded protease and prevent it from cleaving the long polyprotein from polycistronic HCV genes into proteins essential to the structure and function of HCV. |
| sofosbuvir | Sovaldi | Use for the treatment of chronic hepatitis C infection. Used in combination with ribavirin for hepatitis C virus or HCV genotypes 2 and 4; used in combination with peginterferon alfa and ribavirin for HCV genotypes 1 and 4. The second indication is the first approval of an interferon-free regimen for the treatment of chronic HCV infection. Sofosbuvir is a nucleotide polymerase inhibitor that binds to the active site of an HCV-encoded RNA polymerase preventing the synthesis of the viral RNA genome. |
| lamivudine | Epivir-HBV | used in treating chronic hepatitis B. It chemically resembles a normal DNA nucleoside. Once inserted into the growing DNA chain it inhibits further viral DNA replication. |
| adefovir dipivoxil | Hepsera | used in treating hepatitis B. |
Current anti-HIV drugs include the following (classified by their action):
HIV nucleoside-analog reverse transcriptase inhibitors
To replicate, HIV uses the enzyme reverse transcriptase to make a DNA copy of its RNA genome. A complementary copy of this DNA is then made to produce a double-stranded DNA intermediate which is able to insert into host cell chromosomes to form a provirus. Most reverse transcriptase inhibitors are nucleoside analogs. A nucleoside is part of the building block of DNA, consisting of a nitrogenous base bound to the sugar deoxyribose but no phosphate group. A nucleoside analog chemically resembles a normal nucleoside (Figure \(\PageIndex{2}\)).
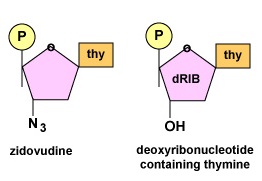
Once phosphate groups are added by either viral or host cell enzymes, the drugs now chemically resemble normal DNA nucleotides, the building block molecules for DNA synthesis. The nucleotide analog binds to the active site of the reverse transcriptase which, in turn, inserts it into the growing DNA strand in place of a normal nucleotide. Once inserted, however, new DNA nucleotides are unable to attach to the drug and DNA synthesis is stopped. This results in an incomplete provirus. For example, zidovudine (AZT, ZDV, Retrovir), as shown in Figure \(\PageIndex{1}\), resembles the deoxyribonucleotide containing the base thymine. Once zidovudine is inserted into the growing DNA strand being transcribed from the viral RNA by reverse transcriptase, no further nucleotides can be attached (Figure \(\PageIndex{3}\)).
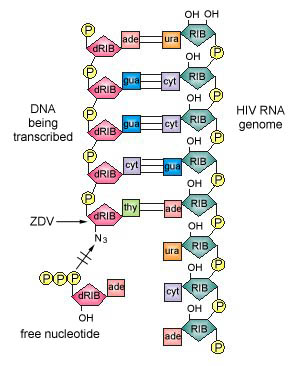
Examples of nucleoside reverse transcriptase inhibitors include:
- zidovudine (AZT; ZDV; Retrovir)
- didanosine (ddI; dideoxyinosine; Videx)
- stavudine (d4T; Zerit)
- lamivudine (3TC; Epivir)
- abacavir (ABC; Ziagen)
- emtricitabine (FTC; Emtriva, Coviracil)
Nucleotide Reverse Transcriptase Inhibitors (NtRTIs)
A NtRTI inhibitor is a a nucleotide analog. A nucleotide is the building block of DNA, consisting of a nitrogenous base bound to the sugar deoxyribose, and a phosphate group. A nucleotide analog chemically resembles a normal nucleotide. The nucleotide analog binds to the active site of the reverse transcriptase which, in turn, inserts it into the growing DNA strand in place of a normal nucleotide. Once inserted, however, new DNA nucleotides are unable to attach to the drug and DNA synthesis is stopped. This results in an incomplete provirus. An example of nucleoside reverse transcriptase inhibitor is tenofovir (TDF;Viread).
3. HIV Non-Nucleoside Reverse Transcriptase Inhibitors (NNRTIs)
These drugs do not resemble regular DNA building blocks. They bind to an allosteric site that regulates reverse transcriptase activity rather than to the enzyme's active site itself as do the above nucleoside analogues (see Figure \(\PageIndex{4}\)). This also prevents HIV provirus formation.
- nevirapine (NVP; Viramune)
- delavirdine (DLV;Rescriptor)
- efavirenz (EFV; Sustiva)
- rilpivirine (Edurant)
- etravirine (ETR, TMC125; Intelence)
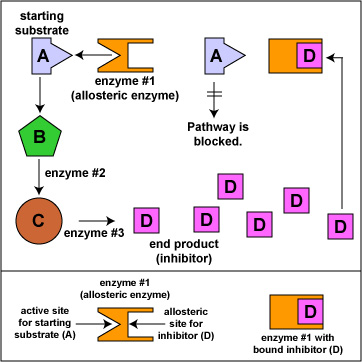
Figure \(\PageIndex{4}\): Noncompetitive Inhibition with Allosteric Enzymes. When the end product (inhibitor) of a pathway combines with the allosteric site of the enzyme, this alters the active site of the enzyme so it can no longer bind to the starting substrate of the pathway. This blocks production of the end product.
HIV Protease Inhibitors (PIs)
In order for maturation of HIV to occur, a HIV enzyme termed a protease has to cleave a long HIV-encoded gag-pol polyprotein to produce reverse transcriptase and integrase (coded by the HIV pol gene) and gag polyprotein (coded by the HIV gag gene). The HIV protease then cleaves the gag polyprotein into capsid protein p17, matrix protein p24, and nucleocapsid protein p7, as well as proteins p6, p2, and p1 whose functions are not yet fully understood (see Figs. 4A, 4B, and 4C). Proteases also cleave the env-polyprotein (coded by the HIV env gene) into the envelope glycoproteins gp120 and gp41 (see Figure \(\PageIndex{5}\)). Protease inhibitors are drugs that bind to the active site of this HIV-encoded protease and prevent it from cleaving the long gag-pol polyprotein and the gag polyprotein into essential proteins essential to the structure of HIV and to RNA packaging within its nucleocapsid (see 4C). As a result, viral maturation does not occur and noninfectious viral particles are produced.
Protease inhibitors include:
- saquinavir (SQV; Inverase)
- ritonavir (RTV; Norvir)
- idinavir (IDV; Crixivan)
- nelfinavir (NFV; Viracept)
- amprenavir (APV; Agenerase)
- atazanavir (ATV; Reyataz)
- fosamprenavir (FPV; Lexiva)
- ritonavir (RTV; Norvir)
- darunavir (DRV; TMC114; Prezista)
- tipranavir (TPV; Aptivus)
Entry Inhibitors (EIs)
EIs are agents interfering with the entry of HIV-1 into cells. During the adsorption and penetration stages of the life cycle of HIV, a portion or domain of the HIV surface glycoprotein gp120 binds to a CD4 molecule on the host cell. This induces a change in shape that brings the chemokine receptor binding domains of the gp120 into proximity with the host cell chemokine receptor. This brings about another conformational change that exposes a previously buried portion of the transmembrane glycoprotein gp41 that enables the viral envelope to fuse with the host cell membrane. EIs interfere with various stages of this process.
a. Agents that block the binding of gp120 to host chemokine receptor 5 (CCR5).
After the gp120 on the envelope of HIV binds to a CD4 molecule on the host cell, it must then also bind to a co-receptor - a chemokine receptor. CCR5-tropic strains of HIV bind to the chemokine receptor CCR5 (see Figure \(\PageIndex{6}\)). (An estimated 50%-60% of people having previously received HIV medication have circulating CCR5-tropic HIV.)
maraviroc (MVC; Selzentry; Celsentri) is a chemokine receptor binding blocker that binds to CCR5 and blocks gp120 from binding to the co-receptor thus blocking adsorption of HIV to the host cell.
b. Agents that block the fusion of the viral envelope with the cytoplasmic membrane of the host cell.
enfuvirtide (ENF; T-20; Fuzeon) binds a gp41 subunit of the viral envelope glycoprotein and prevents the conformational changes required for the fusion of the viral envelope with the cellular cytoplasmic membrane.
5. Integrase Inhibitors
Integrase inhibitors disable HIV integrase, the enzyme that inserts the HIV double-stranded DNA intermediate into host cell DNA. It prevents production of a provirus.
raltegravir (Isentress)
6. Fixed-dose combinations
Tablets containing two or more anti-HIV medications:
- abacivir + lamivudine (Epzicom)
- abacivir + lamivudine + zidovudine (Trizivir)
- efavirenz + emtricitabine + tenofovir DF (Atripla)
- emtricitabine + tenofovir DF (Truvada)
- lamivudine + zidovudine (Combivir)
Certain antiviral cytokines called type-1 interferons have been produced by recombinant DNA technology and several are used to treat certain severe viral infections. These include:
- recombinant interferon alfa-2a (Roferon-A): a cytokine used to treat Kaposi's sarcoma, chronic myelogenous leukemia, and hairy cell leukemia.
- peginterferon alfa-2a (Pegasys) : used to treat hepatitis C (HCV).
- recombinant interferon-alpha 2b (Intron A): a cytokine produced by recombinant DNA technology and used to treat Hepatitis B; malignant melanoma, Kaposi's sarcoma, follicular lymphoma, hairy cell leukemia, warts, and Hepatitis C.
- peginterferon alfa-2b (PEG-Intron; PEG-Intron Redipen): used to treat hepatitis C (HCV).
- recombinant Interferon alfa-2b plus the antiviral drug ribavirin (Rebetron): used to treat hepatitis C (HCV).
- recombinant interferon-alpha n3 (Alferon N): used to treat warts.
- recombinant iInterferon alfacon-1 (Infergen) : used to treat hepatitis C (HCV).
Most of the current antiviral agents don't kill and eliminate the viruses, but rather inhibit their replication and decrease the severity of the disease. As with other microbes, resistant virus strains can emerge with treatment.
Since there are no antiviral drugs for the vast majority of viral infections and most drugs that are available are only partially effective against limited types of viruses, to control viruses, we must rely on the body's immune responses. As will be seen in detail in Units 5 and 6, the immune responses include innate immunity as well as adaptive immunity (antibody production and cell-mediated immunity). Adaptive immunity can be either naturally acquired or, in some cases, artificially acquired.
For a more detailed description of any specific antimicrobial agent, see the website of RxList - The Internet Drug Index.
Summary
- Relatively few antiviral chemotherapeutic agents are currently available and they are only somewhat effective against just a few limited viruses.
- Many antiviral agents resemble normal DNA nucleosides molecules and work by inhibiting viral DNA synthesis.
- Some antiviral agents are protease inhibitors that bind to a viral protease and prevent it from cleaving the long polyprotein from polycistronic genes into proteins essential to viral structure and function.
- Some antiviral agents are entry inhibitors that prevent the virus from either binding to or entering the host cell.
- Antiviral agents are available for only a few viruses, including certain influenza viruses, herpes viruses, cytomegaloviruses, hepatitis C viruses, and HIV.
- Certain interferon cytokines have been produced by recombinant DNA technology and several are used for certain severe viral infections.


