4.3.3: Nutrient Cycles
- Page ID
- 32013
\( \newcommand{\vecs}[1]{\overset { \scriptstyle \rightharpoonup} {\mathbf{#1}} } \)
\( \newcommand{\vecd}[1]{\overset{-\!-\!\rightharpoonup}{\vphantom{a}\smash {#1}}} \)
\( \newcommand{\dsum}{\displaystyle\sum\limits} \)
\( \newcommand{\dint}{\displaystyle\int\limits} \)
\( \newcommand{\dlim}{\displaystyle\lim\limits} \)
\( \newcommand{\id}{\mathrm{id}}\) \( \newcommand{\Span}{\mathrm{span}}\)
( \newcommand{\kernel}{\mathrm{null}\,}\) \( \newcommand{\range}{\mathrm{range}\,}\)
\( \newcommand{\RealPart}{\mathrm{Re}}\) \( \newcommand{\ImaginaryPart}{\mathrm{Im}}\)
\( \newcommand{\Argument}{\mathrm{Arg}}\) \( \newcommand{\norm}[1]{\| #1 \|}\)
\( \newcommand{\inner}[2]{\langle #1, #2 \rangle}\)
\( \newcommand{\Span}{\mathrm{span}}\)
\( \newcommand{\id}{\mathrm{id}}\)
\( \newcommand{\Span}{\mathrm{span}}\)
\( \newcommand{\kernel}{\mathrm{null}\,}\)
\( \newcommand{\range}{\mathrm{range}\,}\)
\( \newcommand{\RealPart}{\mathrm{Re}}\)
\( \newcommand{\ImaginaryPart}{\mathrm{Im}}\)
\( \newcommand{\Argument}{\mathrm{Arg}}\)
\( \newcommand{\norm}[1]{\| #1 \|}\)
\( \newcommand{\inner}[2]{\langle #1, #2 \rangle}\)
\( \newcommand{\Span}{\mathrm{span}}\) \( \newcommand{\AA}{\unicode[.8,0]{x212B}}\)
\( \newcommand{\vectorA}[1]{\vec{#1}} % arrow\)
\( \newcommand{\vectorAt}[1]{\vec{\text{#1}}} % arrow\)
\( \newcommand{\vectorB}[1]{\overset { \scriptstyle \rightharpoonup} {\mathbf{#1}} } \)
\( \newcommand{\vectorC}[1]{\textbf{#1}} \)
\( \newcommand{\vectorD}[1]{\overrightarrow{#1}} \)
\( \newcommand{\vectorDt}[1]{\overrightarrow{\text{#1}}} \)
\( \newcommand{\vectE}[1]{\overset{-\!-\!\rightharpoonup}{\vphantom{a}\smash{\mathbf {#1}}}} \)
\( \newcommand{\vecs}[1]{\overset { \scriptstyle \rightharpoonup} {\mathbf{#1}} } \)
\(\newcommand{\longvect}{\overrightarrow}\)
\( \newcommand{\vecd}[1]{\overset{-\!-\!\rightharpoonup}{\vphantom{a}\smash {#1}}} \)
\(\newcommand{\avec}{\mathbf a}\) \(\newcommand{\bvec}{\mathbf b}\) \(\newcommand{\cvec}{\mathbf c}\) \(\newcommand{\dvec}{\mathbf d}\) \(\newcommand{\dtil}{\widetilde{\mathbf d}}\) \(\newcommand{\evec}{\mathbf e}\) \(\newcommand{\fvec}{\mathbf f}\) \(\newcommand{\nvec}{\mathbf n}\) \(\newcommand{\pvec}{\mathbf p}\) \(\newcommand{\qvec}{\mathbf q}\) \(\newcommand{\svec}{\mathbf s}\) \(\newcommand{\tvec}{\mathbf t}\) \(\newcommand{\uvec}{\mathbf u}\) \(\newcommand{\vvec}{\mathbf v}\) \(\newcommand{\wvec}{\mathbf w}\) \(\newcommand{\xvec}{\mathbf x}\) \(\newcommand{\yvec}{\mathbf y}\) \(\newcommand{\zvec}{\mathbf z}\) \(\newcommand{\rvec}{\mathbf r}\) \(\newcommand{\mvec}{\mathbf m}\) \(\newcommand{\zerovec}{\mathbf 0}\) \(\newcommand{\onevec}{\mathbf 1}\) \(\newcommand{\real}{\mathbb R}\) \(\newcommand{\twovec}[2]{\left[\begin{array}{r}#1 \\ #2 \end{array}\right]}\) \(\newcommand{\ctwovec}[2]{\left[\begin{array}{c}#1 \\ #2 \end{array}\right]}\) \(\newcommand{\threevec}[3]{\left[\begin{array}{r}#1 \\ #2 \\ #3 \end{array}\right]}\) \(\newcommand{\cthreevec}[3]{\left[\begin{array}{c}#1 \\ #2 \\ #3 \end{array}\right]}\) \(\newcommand{\fourvec}[4]{\left[\begin{array}{r}#1 \\ #2 \\ #3 \\ #4 \end{array}\right]}\) \(\newcommand{\cfourvec}[4]{\left[\begin{array}{c}#1 \\ #2 \\ #3 \\ #4 \end{array}\right]}\) \(\newcommand{\fivevec}[5]{\left[\begin{array}{r}#1 \\ #2 \\ #3 \\ #4 \\ #5 \\ \end{array}\right]}\) \(\newcommand{\cfivevec}[5]{\left[\begin{array}{c}#1 \\ #2 \\ #3 \\ #4 \\ #5 \\ \end{array}\right]}\) \(\newcommand{\mattwo}[4]{\left[\begin{array}{rr}#1 \amp #2 \\ #3 \amp #4 \\ \end{array}\right]}\) \(\newcommand{\laspan}[1]{\text{Span}\{#1\}}\) \(\newcommand{\bcal}{\cal B}\) \(\newcommand{\ccal}{\cal C}\) \(\newcommand{\scal}{\cal S}\) \(\newcommand{\wcal}{\cal W}\) \(\newcommand{\ecal}{\cal E}\) \(\newcommand{\coords}[2]{\left\{#1\right\}_{#2}}\) \(\newcommand{\gray}[1]{\color{gray}{#1}}\) \(\newcommand{\lgray}[1]{\color{lightgray}{#1}}\) \(\newcommand{\rank}{\operatorname{rank}}\) \(\newcommand{\row}{\text{Row}}\) \(\newcommand{\col}{\text{Col}}\) \(\renewcommand{\row}{\text{Row}}\) \(\newcommand{\nul}{\text{Nul}}\) \(\newcommand{\var}{\text{Var}}\) \(\newcommand{\corr}{\text{corr}}\) \(\newcommand{\len}[1]{\left|#1\right|}\) \(\newcommand{\bbar}{\overline{\bvec}}\) \(\newcommand{\bhat}{\widehat{\bvec}}\) \(\newcommand{\bperp}{\bvec^\perp}\) \(\newcommand{\xhat}{\widehat{\xvec}}\) \(\newcommand{\vhat}{\widehat{\vvec}}\) \(\newcommand{\uhat}{\widehat{\uvec}}\) \(\newcommand{\what}{\widehat{\wvec}}\) \(\newcommand{\Sighat}{\widehat{\Sigma}}\) \(\newcommand{\lt}{<}\) \(\newcommand{\gt}{>}\) \(\newcommand{\amp}{&}\) \(\definecolor{fillinmathshade}{gray}{0.9}\)- Explain the processes of carbon, nitrogen, and phosphorus cycling.
- Explain the process by which rhizobia infect legumes and form root nodules.
Nutrient cycles, also known as biogeochemical cycles, describe the movement of chemical elements through different media, such as the atmosphere, soil, rocks, bodies of water, and organisms. Nutrient cycles keep essential elements available to plants and other organisms. The cycling of three macronutrients are discussed below. The first is the carbon cycle. Plants acquire carbon through photosynthesis. The nitrogen and phosphorus cycles are also discussed, and plant acquire nitrogen and phosphorus as mineral nutrients from the soil.
The Carbon Cycle
The carbon cycle is actually comprised of several interconnected cycles: one dealing with rapid carbon exchange among living organisms and the other dealing with the long-term cycling of carbon through geologic processes (Figure \(\PageIndex{1}\)). The overall effect is that carbon is constantly recycled in the dynamic processes taking place in the atmosphere, at the surface and in the crust of the earth. The vast majority of carbon resides as inorganic minerals in crustal rocks. Other reservoirs of carbon, places where carbon accumulates, include the oceans and atmosphere. Some of the carbon atoms in your body today may long ago have resided in a dinosaur's body, or perhaps were once buried deep in the Earth's crust as carbonate rock minerals.
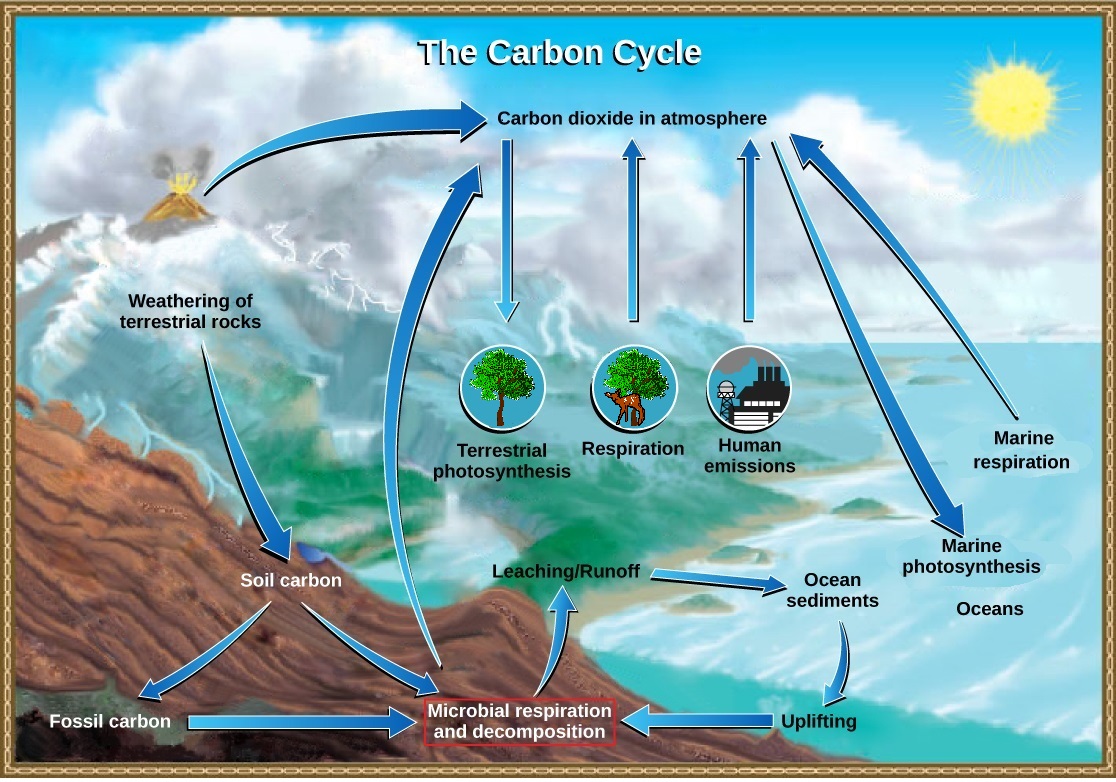
Carbon Cycles Slowly between Land and the Ocean
On land, carbon is stored in soil as organic carbon in the form of decomposing organisms or terrestrial rocks. Decomposed plants and algae are sometimes buried and compressed between layers of sediments. After millions of years fossil fuels such as coal, oil, and natural gas are formed. The weathering of terrestrial rock and minerals release carbon into the soil.
Carbon-containing compounds in the soil can be washed into bodies of water through leaching. This water eventually enters the ocean. Atmospheric carbon dioxide also dissolves in the ocean, reacting with water molecules to form carbonate ions (CO32-). Some of these ions combine with calcium ions in the seawater to form calcium carbonate (CaCO3), a major component of the shells of marine organisms. These organisms eventually die and their shells form sediments on the ocean floor. Over geologic time, the calcium carbonate forms limestone, which comprises the largest carbon reservoir on Earth.
Carbonate also precipitates in sediments, forming carbonate rocks, such as limestone. Carbon sediments from the ocean floor are taken deep within Earth by the process of subduction: the movement of one tectonic plate beneath another. The ocean sediments are subducted by the actions of plate tectonics, melted and then returned to the surface during volcanic activity. Plate tectonics can also cause uplifting, returning ocean sediments to land.
Carbon Cycles Quickly between Organisms and the Atmosphere
Carbon dioxide is converted into glucose, an energy-rich organic molecule through photosynthesis by plants, algae, and some bacteria. They can then produce other organic molecules like complex carbohydrates (such as starch), proteins and lipids, which animals can eat. Most terrestrial autotrophs obtain their carbon dioxide directly from the atmosphere, while marine autotrophs acquire it in the dissolved form (bicarbonate, HCO3–).
Plants, animals, and other organisms break down these organic molecules during the process of aerobic cellular respiration, which consumes oxygen and releases energy, water and carbon dioxide. Carbon dioxide is returned to the atmosphere during gaseous exchange. Another process by which organic material is recycled is the decomposition of dead organisms. During this process, bacteria and fungi break down the complex organic compounds. Decomposers may do respiration, releasing carbon dioxide, or other processes that release methane (CH4). Photosynthesis and respiration are actually reciprocal to one another with regard to the cycling of carbon: photosynthesis removes carbon dioxide from the atmosphere and respiration returns it. A significant disruption of one process can therefore affect the amount of carbon dioxide in the atmosphere.
Cellular respiration is only one process that releases carbon dioxide. Physical processes, such as the eruption of volcanoes and release from hydrothermal vents (openings in the ocean floor) add carbon dioxide to the atmosphere. Additionally, the combustion of wood and fossil fuels releases carbon dioxide. The level of carbon dioxide in the atmosphere is greatly influenced by the reservoir of carbon in the oceans. The exchange of carbon between the atmosphere and water reservoirs influences how much carbon is found in each.
Nitrogen Cycle
Getting nitrogen into living organisms is difficult. Plants and algae are not equipped to incorporate nitrogen from the atmosphere (where it exists as tightly bonded, triple covalent N2) although this molecule comprises approximately 78 percent of the atmosphere. Because most of the nitrogen is stored in the atmosphere, the atmosphere is considered a reservoir of nitrogen.
Nitrogen Fixation
The nitrogen molecule (N2) is quite inert. To break it apart so that its atoms can combine with other atoms requires the input of substantial amounts of energy. Nitrogen fixation is the process of converting nitrogen gas into ammonia (NH3), which spontaneously becomes ammonium (NH4+). Ammonium is found in bodies of water and in the soil (Figure \(\PageIndex{2}\)).
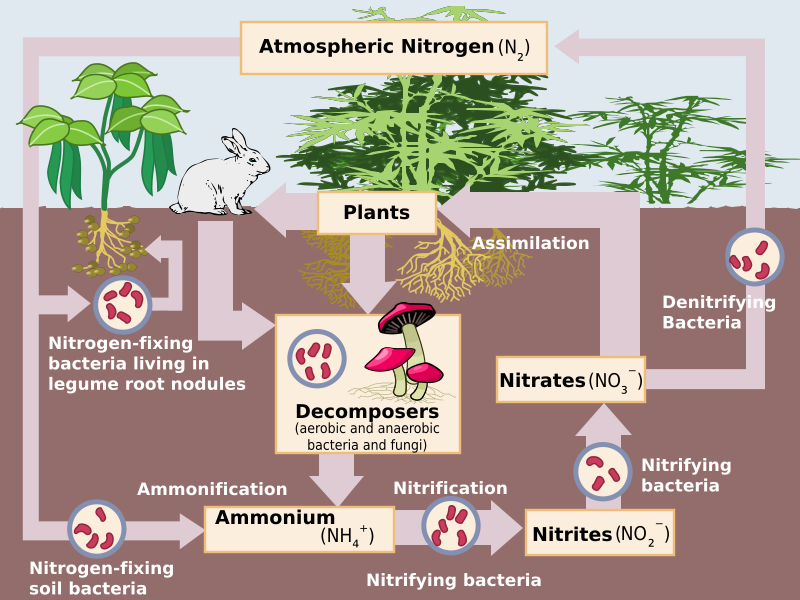
Three processes are responsible for most of the nitrogen fixation in the biosphere. The first is atmospheric fixation by lightning. The enormous energy of lightning breaks nitrogen molecules and enables their atoms to combine with oxygen in the air forming nitrogen oxides. These dissolve in rain, forming nitrates, that are carried to the earth. Atmospheric nitrogen fixation probably contributes some 5-8% of the total nitrogen fixed. The second process is industrial fixation. Under great pressure, at a temperature of 600°C, and with the use of a catalyst, atmospheric nitrogen and hydrogen (usually derived from natural gas or petroleum) can be combined to form ammonia (NH3). Ammonia can be used directly as fertilizer, but most of it is further processed to urea and ammonium nitrate (NH4NO3).
The third process is biological fixation. Some nitrogen-fixing bacteria, like Azotobacter, are free-living. Others (Rhizobium and Bradyrhizobium) form a symbiotic relationship with plants in Fabaceae (bean or legume family), which includes beans, peas, soybeans, alfalfa, clovers, and many other species (Figure \(\PageIndex{3}\)). Bacteria that form root nodules in legumes are informally called rhizobia. Frankia forms root nodules in alders, which are non-legume trees. Nitrogen-fixing cyanobacteria that are symbiotic with the water fern Azolla are essential to maintaining the fertility of semi-aquatic environments like rice paddies. Lichens that contain cyanobacteria can also fix nitrogen. Biological nitrogen fixation requires a complex set of enzymes and a huge expenditure of ATP. Although the first stable product of the process is ammonia, this is quickly incorporated into protein and other organic nitrogen compounds.

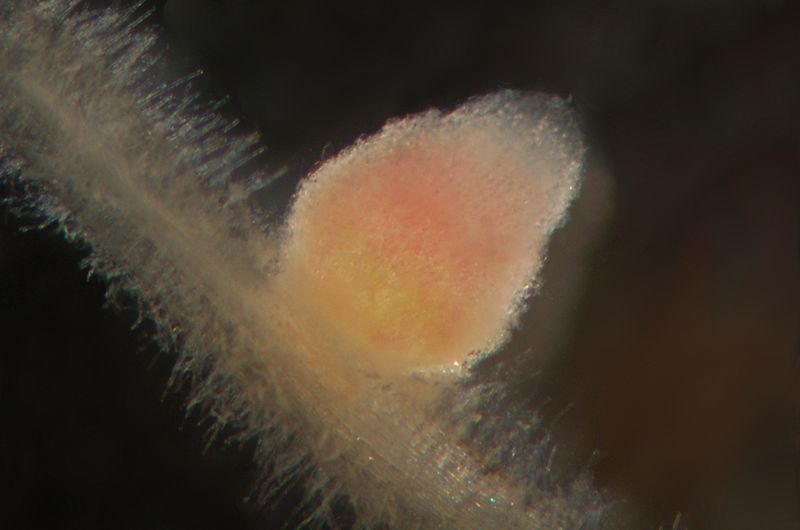
Rhizobia live freely in the soil, but they cannot fix atmospheric nitrogen until they have infected the roots of a legume. Legumes provide nitrogen-fixing bacteria with carbohydrates for energy and limit oxygen in the root nodule. Nitrogenase, the enzyme that fixes nitrogen, is inhibited by oxygen, but the bacteroids still need some oxygen to conduct cellular respiration to produce ATP. Legumes produce leghemoglobin, which binds oxygen like the hemoglobin of vertebrates. This reduces oxygen availability in the root nodule. Leghemoglobin contains iron and appears red when bound to oxygen; in fact, a freshly-cut nodule is red or pink (Figure \(\PageIndex{4}\)).
Root hairs release chemicals called flavonoids, which cause bacteria to synthesize Nod factors. These are chemical signals that bind to root epidermal cell receptors and induce the legume to produce nodules. Nod factors mediate interactions between specific strains of rhizobia and the associated legume species. Different strains of rhizobia produce different Nod factors, and different legumes produce receptors of different specificity. Because of the specificity of the interaction between the Nod factor and the receptor on the legume, some strains of rhizobia will infect only peas, some only clover, some only alfalfa, etc. The treating of legume seeds with the proper strain of rhizobia is a routine agricultural practice.
If the combination is correct, the bacteria enter an epidermal cell of the root and then migrate into the cortex. Their path runs within an intracellular channel that grows through one cortex cell after another. This infection thread is constructed by root hair, not the bacteria, and is formed only in response to the infection. When the infection thread reaches a cell deep in the cortex, it bursts, and the rhizobia are engulfed by endocytosis into membrane-enclosed symbiosomes within the cytoplasm. At this time the cortical (cortex) cell goes through several rounds of mitosis—without cytokinesis —so the cell becomes polyploid. The cortical cells then begin to divide rapidly forming a nodule. This response is driven by the translocation of cytokinins from epidermal cells to the cells of the cortex. The rhizobia also go through a period of rapid multiplication within the nodule cells. Then they begin to change shape and lose their motility. The bacteroids, as they are now called, may almost fill the cell. Only now does nitrogen fixation begin.
Nitrification
Ammonium is converted by bacteria and archaea into nitrites (NO2−) and then nitrates (NO3−) through the process of nitrification. Like ammonium, nitrites and nitrates are found in water and the soil.
Denitrification
Some nitrates are converted back into nitrogen gas, which is released into the atmosphere. The process, called denitrification, is conducted by bacteria.
Assimilation
Ammonium and nitrates can be used directly by plants and other producers to make organic molecules such as DNA and proteins through the process of assimilation. This nitrogen is now available to consumers. Organic nitrogen is especially important to the study of ecosystem dynamics because many processes, such as primary production, are limited by the available supply of nitrogen.
Ammonification
Consumers excrete organic nitrogen compounds that return to the environment. Additionally dead organisms at each trophic level contain organic nitrogen. Microorganisms, such as bacteria and fungi, decompose these wastes and dead tissues, ultimately producing ammonium through the process of ammonification.
Phosphorus Cycle
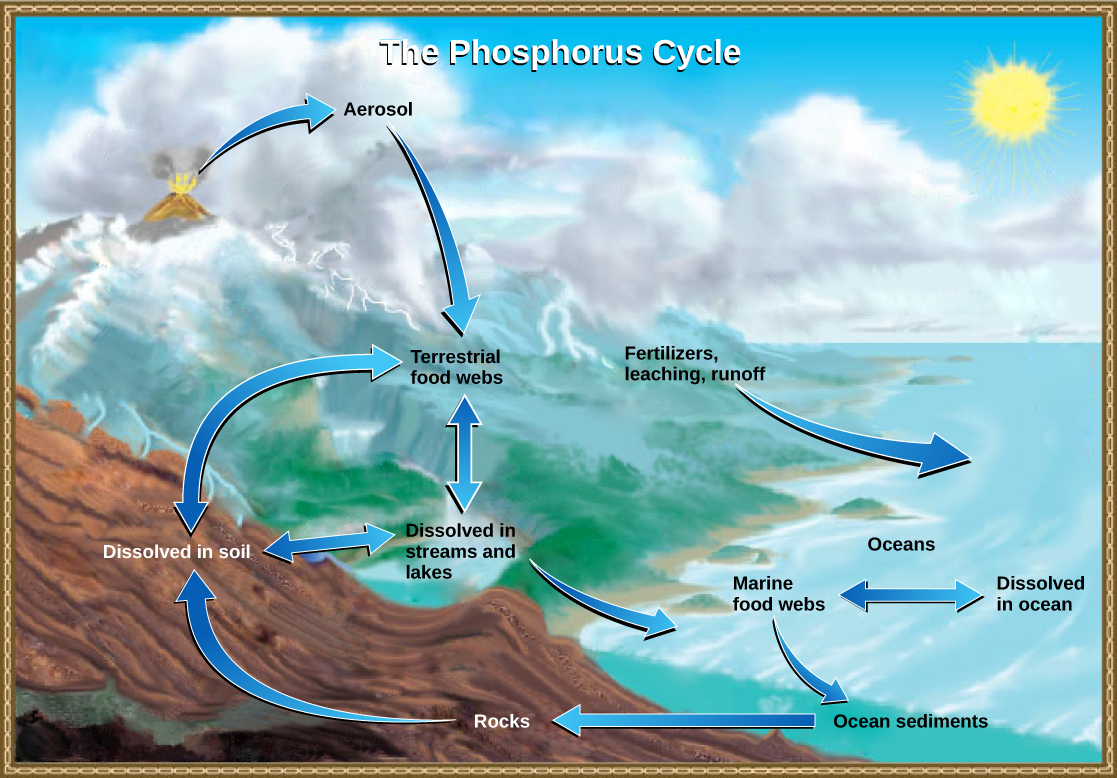
Several forms of nitrogen (N2, NH4+, NO3−, etc.) were involved in the nitrogen cycle, but phosphorus remains primarily in the form of the phosphate ion (PO43-). Also in contrast to the nitrogen cycle, there is no form of phosphorus in the atmosphere.
Rocks are a reservoir for phosphorus, and these rocks have their origins in the ocean. Phosphate-containing ocean sediments form primarily from the bodies of marine organisms and from their excretions. However, volcanic ash, aerosols, and mineral dust may also be significant phosphate sources. This sediment is then moved to land over geologic time by the uplifting of Earth’s surface (Figure \(\PageIndex{5}\)). The movement of phosphate from the ocean to the land and through the soil is extremely slow, with the average phosphate ion having an oceanic residence time between 20,000 and 100,000 years.
Weathering of rocks releases phosphates into the soil and bodies of water. Plants can assimilate phosphates in the soil and incorporate it into organic molecules, making phosphorus available to consumers in terrestrial food webs. Waste and dead organisms are decomposed by fungi and bacteria, releasing phosphates back into the soil. Some phosphate is leached from the soil, entering into rivers, lakes, and the ocean. Primary producers in aquatic food webs, such as algae and photosynthetic bacteria, assimilate phosphate, and organic phosphate is thus available to consumers in aquatic food webs. Similar to terrestrial food webs, phosphorus is reciprocally exchanged between phosphate dissolved in the ocean and organic phosphorus in marine organisms.
Attributions
Curated and authored by Melissa Ha using the following sources:
- 3.2 Biogeochemical Cycles, 2.2 Energy, and 2.4 Energy Enters Ecosystems Through Photosynthesis from Environmental Biology by Matthew R. Fisher (licensed under CC-BY)
- Carbon Cycle and Nitrogen Cycle from Biology by John W. Kimball (licensed under CC-BY)
- 6.2 Biogeochemical Cycles and the Flow of Energy in the Earth System from Sustainability: A Comprehensive Foundation by Tom Theis and Jonathan Tomkin, Editors (licensed under CC-BY). Download for free at CNX.
- 2 Cycling of Matter from AP Environmental Science by University of California College Prep, University of California (licensed under CC-BY). Download for free at CNX.


