8.4 How Neurons Talk- Synaptic Communication
- Page ID
- 171410
\( \newcommand{\vecs}[1]{\overset { \scriptstyle \rightharpoonup} {\mathbf{#1}} } \)
\( \newcommand{\vecd}[1]{\overset{-\!-\!\rightharpoonup}{\vphantom{a}\smash {#1}}} \)
\( \newcommand{\dsum}{\displaystyle\sum\limits} \)
\( \newcommand{\dint}{\displaystyle\int\limits} \)
\( \newcommand{\dlim}{\displaystyle\lim\limits} \)
\( \newcommand{\id}{\mathrm{id}}\) \( \newcommand{\Span}{\mathrm{span}}\)
( \newcommand{\kernel}{\mathrm{null}\,}\) \( \newcommand{\range}{\mathrm{range}\,}\)
\( \newcommand{\RealPart}{\mathrm{Re}}\) \( \newcommand{\ImaginaryPart}{\mathrm{Im}}\)
\( \newcommand{\Argument}{\mathrm{Arg}}\) \( \newcommand{\norm}[1]{\| #1 \|}\)
\( \newcommand{\inner}[2]{\langle #1, #2 \rangle}\)
\( \newcommand{\Span}{\mathrm{span}}\)
\( \newcommand{\id}{\mathrm{id}}\)
\( \newcommand{\Span}{\mathrm{span}}\)
\( \newcommand{\kernel}{\mathrm{null}\,}\)
\( \newcommand{\range}{\mathrm{range}\,}\)
\( \newcommand{\RealPart}{\mathrm{Re}}\)
\( \newcommand{\ImaginaryPart}{\mathrm{Im}}\)
\( \newcommand{\Argument}{\mathrm{Arg}}\)
\( \newcommand{\norm}[1]{\| #1 \|}\)
\( \newcommand{\inner}[2]{\langle #1, #2 \rangle}\)
\( \newcommand{\Span}{\mathrm{span}}\) \( \newcommand{\AA}{\unicode[.8,0]{x212B}}\)
\( \newcommand{\vectorA}[1]{\vec{#1}} % arrow\)
\( \newcommand{\vectorAt}[1]{\vec{\text{#1}}} % arrow\)
\( \newcommand{\vectorB}[1]{\overset { \scriptstyle \rightharpoonup} {\mathbf{#1}} } \)
\( \newcommand{\vectorC}[1]{\textbf{#1}} \)
\( \newcommand{\vectorD}[1]{\overrightarrow{#1}} \)
\( \newcommand{\vectorDt}[1]{\overrightarrow{\text{#1}}} \)
\( \newcommand{\vectE}[1]{\overset{-\!-\!\rightharpoonup}{\vphantom{a}\smash{\mathbf {#1}}}} \)
\( \newcommand{\vecs}[1]{\overset { \scriptstyle \rightharpoonup} {\mathbf{#1}} } \)
\(\newcommand{\longvect}{\overrightarrow}\)
\( \newcommand{\vecd}[1]{\overset{-\!-\!\rightharpoonup}{\vphantom{a}\smash {#1}}} \)
\(\newcommand{\avec}{\mathbf a}\) \(\newcommand{\bvec}{\mathbf b}\) \(\newcommand{\cvec}{\mathbf c}\) \(\newcommand{\dvec}{\mathbf d}\) \(\newcommand{\dtil}{\widetilde{\mathbf d}}\) \(\newcommand{\evec}{\mathbf e}\) \(\newcommand{\fvec}{\mathbf f}\) \(\newcommand{\nvec}{\mathbf n}\) \(\newcommand{\pvec}{\mathbf p}\) \(\newcommand{\qvec}{\mathbf q}\) \(\newcommand{\svec}{\mathbf s}\) \(\newcommand{\tvec}{\mathbf t}\) \(\newcommand{\uvec}{\mathbf u}\) \(\newcommand{\vvec}{\mathbf v}\) \(\newcommand{\wvec}{\mathbf w}\) \(\newcommand{\xvec}{\mathbf x}\) \(\newcommand{\yvec}{\mathbf y}\) \(\newcommand{\zvec}{\mathbf z}\) \(\newcommand{\rvec}{\mathbf r}\) \(\newcommand{\mvec}{\mathbf m}\) \(\newcommand{\zerovec}{\mathbf 0}\) \(\newcommand{\onevec}{\mathbf 1}\) \(\newcommand{\real}{\mathbb R}\) \(\newcommand{\twovec}[2]{\left[\begin{array}{r}#1 \\ #2 \end{array}\right]}\) \(\newcommand{\ctwovec}[2]{\left[\begin{array}{c}#1 \\ #2 \end{array}\right]}\) \(\newcommand{\threevec}[3]{\left[\begin{array}{r}#1 \\ #2 \\ #3 \end{array}\right]}\) \(\newcommand{\cthreevec}[3]{\left[\begin{array}{c}#1 \\ #2 \\ #3 \end{array}\right]}\) \(\newcommand{\fourvec}[4]{\left[\begin{array}{r}#1 \\ #2 \\ #3 \\ #4 \end{array}\right]}\) \(\newcommand{\cfourvec}[4]{\left[\begin{array}{c}#1 \\ #2 \\ #3 \\ #4 \end{array}\right]}\) \(\newcommand{\fivevec}[5]{\left[\begin{array}{r}#1 \\ #2 \\ #3 \\ #4 \\ #5 \\ \end{array}\right]}\) \(\newcommand{\cfivevec}[5]{\left[\begin{array}{c}#1 \\ #2 \\ #3 \\ #4 \\ #5 \\ \end{array}\right]}\) \(\newcommand{\mattwo}[4]{\left[\begin{array}{rr}#1 \amp #2 \\ #3 \amp #4 \\ \end{array}\right]}\) \(\newcommand{\laspan}[1]{\text{Span}\{#1\}}\) \(\newcommand{\bcal}{\cal B}\) \(\newcommand{\ccal}{\cal C}\) \(\newcommand{\scal}{\cal S}\) \(\newcommand{\wcal}{\cal W}\) \(\newcommand{\ecal}{\cal E}\) \(\newcommand{\coords}[2]{\left\{#1\right\}_{#2}}\) \(\newcommand{\gray}[1]{\color{gray}{#1}}\) \(\newcommand{\lgray}[1]{\color{lightgray}{#1}}\) \(\newcommand{\rank}{\operatorname{rank}}\) \(\newcommand{\row}{\text{Row}}\) \(\newcommand{\col}{\text{Col}}\) \(\renewcommand{\row}{\text{Row}}\) \(\newcommand{\nul}{\text{Nul}}\) \(\newcommand{\var}{\text{Var}}\) \(\newcommand{\corr}{\text{corr}}\) \(\newcommand{\len}[1]{\left|#1\right|}\) \(\newcommand{\bbar}{\overline{\bvec}}\) \(\newcommand{\bhat}{\widehat{\bvec}}\) \(\newcommand{\bperp}{\bvec^\perp}\) \(\newcommand{\xhat}{\widehat{\xvec}}\) \(\newcommand{\vhat}{\widehat{\vvec}}\) \(\newcommand{\uhat}{\widehat{\uvec}}\) \(\newcommand{\what}{\widehat{\wvec}}\) \(\newcommand{\Sighat}{\widehat{\Sigma}}\) \(\newcommand{\lt}{<}\) \(\newcommand{\gt}{>}\) \(\newcommand{\amp}{&}\) \(\definecolor{fillinmathshade}{gray}{0.9}\)By the end of this section, you should be able to:
- Explain the structural and functional differences between electrical and chemical synapses.
- Describe the sequence of events at a chemical synapse, including neurotransmitter release, receptor binding, and signal termination.
- Distinguish between excitatory postsynaptic potentials (EPSPs) and inhibitory postsynaptic potentials (IPSPs).
- Explain the importance of synaptic integration, temporal summation, and spatial summation in neuronal communication.
- Identify major neurotransmitters and their general roles in the nervous system.
- Relate dysfunction in synaptic transmission to clinical conditions such as myasthenia gravis and depression.
Neuronal Communication and Synaptic Transmission
The nervous system depends on communication between billions of individual neurons. Each neuron acts like a messenger, but for information to spread through the network, signals must cross from one cell to another. This happens at specialized junctions called synapses, where the axon terminal of one neuron passes information to the next cell. Some synapses use direct electrical connections, while most rely on chemical messengers called neurotransmitters. Together, these mechanisms allow signals to move rapidly, precisely, and in a highly regulated manner.
A useful way to imagine this is to think of a relay race. One runner (the presynaptic neuron) carries the baton (the electrical signal) down the track. When the runner reaches the next teammate (the postsynaptic neuron), they must pass the baton across a short but crucial space. If the handoff is smooth, the race continues without delay. Similarly, at the synapse, information is handed off across a tiny gap, the synaptic cleft, ensuring that the signal continues its journey through the nervous system.
For neurons to work as a network, they must be able to pass information from one to the next. The point where this transfer happens is the synapse. A synapse is a specialized structure where the axon terminal of one cell (the presynaptic neuron) communicates with another cell (the postsynaptic neuron). Between them is a tiny gap called the synaptic cleft, which is only about 20–40 nanometers wide. Despite being microscopic, this space plays an enormous role in controlling how signals move through the nervous system.
The synapse is the junction where this communication occurs. Structurally, a synapse has three parts:
- the presynaptic neuron, which sends the signal;
- the synaptic cleft, a narrow extracellular gap; and
- the postsynaptic neuron, which receives the signal.

Figure 8.22: Synapsis. The synapse is a connection between a neuron and its target cell (which is not necessarily a neuron). The presynaptic element is the synaptic end bulb of the axon where Ca2+ enters the bulb to cause vesicle fusion and neurotransmitter release. The neurotransmitter diffuses across the synaptic cleft to bind to its receptor. The neurotransmitter is cleared from the synapse either by enzymatic degradation, neuronal reuptake, or glial reuptake.(OpenStax, 2013, licensed under CC-BY-SA, shared source content).
Synapses come in two main forms. Electrical synapses connect cells directly through gap junctions. These are like open doorways that allow ions to flow freely between cells. Because the signal does not need to convert from one form to another, communication is almost instantaneous. Electrical synapses are important in areas of the nervous system that require speed and coordination, such as rhythmic breathing or eye movement pathways.
Far more common, however, are chemical synapses. In these, the presynaptic neuron converts its electrical signal into a chemical one by releasing neurotransmitters from vesicles. These molecules travel across the synaptic cleft and bind to receptors on the postsynaptic cell, which then converts the chemical signal back into an electrical one. Although slower than electrical synapses, chemical synapses provide greater flexibility. Different neurotransmitters can excite, inhibit, or modulate the activity of the postsynaptic neuron, allowing for the complexity of thought, memory, and behavior.
Chemical Signals
Neurocrine Signaling: Neurotransmitters, Neuromodulators, and Neurohormones
The nervous system uses a wide variety of chemical messengers, and new ones are still being discovered. Collectively, these messengers are called neurocrine signals, but they do not all act in the same way. Some serve as neurotransmitters, others as neuromodulators, and still others as neurohormones.
Neurotransmitters and neuromodulators usually act locally, influencing nearby cells through paracrine signaling. The difference between them comes down to speed and the type of response. A chemical that is released into the synapse and triggers a rapid, short-lived effect on the postsynaptic cell is considered a neurotransmitter. In contrast, a neuromodulator produces slower, longer-lasting effects, often acting at sites both within and outside of the synapse. Importantly, many neurocrine molecules can fill both roles depending on the receptor they bind to.
Some neurocrine signals even act back on the very cell that released them, functioning as autocrine signals as well as paracrine ones. Neurohormones, in contrast, are secreted into the bloodstream and travel throughout the body, influencing distant targets. This framework prepares us to explore the specific groups of neurotransmitters and their receptors.
Neurocrine Receptors
At chemical synapses, neurotransmitters and neuromodulators act by binding to neurocrine receptors, which fall into two main categories: receptor-channels and G protein-coupled receptors (GPCRs).
Receptor channels are ligand-gated ion channels that respond very quickly. When a neurotransmitter binds, the channel opens and ions flow across the membrane. Because of their rapid, direct effect, these are also called ionotropic receptors. Some are highly selective, opening only for a single ion type, while others are more flexible, such as channels that allow both sodium and potassium to pass.
GPCRs act more slowly because they rely on second messenger systems inside the cell. These receptors are also called metabotropic receptors. Instead of directly opening a channel, they trigger a signaling cascade that can eventually cause an ion channel to open or close, or influence other cellular processes. This slower mechanism allows for more complex and long-lasting effects compared to ionotropic receptors (see Chapter 5).
Except for nitric oxide, all neurotransmitters act by binding to specific receptor types, and most have multiple subtypes. These subtypes explain how the same neurotransmitter can produce very different effects in different tissues. For instance, serotonin (5-HT) has at least 20 identified receptor subtypes, including 5-HT₁ and 5-HT₂, each with distinct functions.
Research on neurotransmitters and their receptors has advanced rapidly thanks to modern molecular biology. Scientists have cloned the genes for many receptor subtypes, making it possible to create mutant receptors and study their behavior. In addition, new agonists (molecules that mimic neurotransmitter activity) and antagonists (molecules that block it) have been discovered or synthesized, providing powerful tools for both research and medicine. We will begin with acetylcholine, the best-known neurotransmitter, before moving into amines, amino acids, peptides, purines and gases, and lipids, each with their characteristic receptor types and functions.
Neurotransmitters
Acetylcholine (ACh)
Acetylcholine is one of the most studied neurotransmitters and serves as the primary chemical signal between motor neurons and skeletal muscle. It is synthesized from choline and acetyl coenzyme A (acetyl CoA) by the enzyme choline acetyltransferase. Once released into the synaptic cleft, acetylcholine binds to two major cholinergic receptor types:
-
Nicotinic receptors are ionotropic receptor-channels. They function as a ligand-gated receptor and an ion channel together. When ACh binds, the channel opens, allowing sodium (Na⁺) and potassium (K⁺) ions to move across the postsynaptic membrane. This produces rapid depolarization and is crucial for muscle contraction at the neuromuscular junction.
-
Muscarinic receptors are metabotropic GPCRs. Their effects are slower and more varied because they activate second messenger pathways. These receptors are especially important in the parasympathetic branch of the autonomic nervous system.
Acetylcholine’s activity is quickly terminated by the enzyme acetylcholinesterase (AChE), which breaks it down into acetate and choline. This rapid breakdown ensures that ACh’s effects are short-lived and tightly controlled.
Myasthenia Gravis (MG) is an autoimmune disorder in which the body produces antibodies that block or destroy nicotinic acetylcholine receptors (nAChRs) at the neuromuscular junction. Without enough functioning receptors, acetylcholine released from motor neurons cannot effectively trigger skeletal muscle contraction.
The hallmark symptom of MG is muscle weakness that worsens with activity and improves with rest. Commonly affected muscles include those that control eye and eyelid movement, facial expression, swallowing, and limb function. Severe cases can impair breathing if respiratory muscles are involved.
Diagnosis is often confirmed by blood tests that detect anti-AChR antibodies or by clinical tests that reveal muscle fatigue. Treatments focus on improving neuromuscular transmission and reducing the autoimmune response. Drugs such as acetylcholinesterase inhibitors (e.g., pyridostigmine) increase the amount of acetylcholine available at the synapse, while immunosuppressants reduce antibody production. In some cases, removing the thymus gland (thymectomy) can improve symptoms.
Myasthenia Gravis highlights the importance of acetylcholine and nicotinic receptors in normal muscle function, as well as the devastating effects that can occur when this system is disrupted.

Clinical conditions such as MG highlight what happens when acetylcholine signaling is disrupted at the neuromuscular junction. Yet the same receptors that can cause disease when targeted by the immune system are also influenced by everyday substances we encounter. From nicotine in tobacco to muscarine in certain mushrooms, naturally occurring compounds can strongly affect nicotinic and muscarinic receptors. Understanding these real-world examples not only deepens our appreciation of how sensitive the nervous system is to chemical signals but also connects textbook physiology to experiences and challenges people face outside the clinic.
The names of nicotinic and muscarinic receptors reflect how scientists first studied them, but they also explain why certain substances have strong effects on the nervous system.
Nicotine, found in tobacco products, binds to nicotinic receptors. Because these receptors are located at the neuromuscular junction and in autonomic ganglia, nicotine can cause a rapid increase in skeletal muscle activity and changes in heart rate and blood pressure. This is one reason nicotine is such a powerful stimulant and highly addictive.
Muscarine, on the other hand, is a toxin found in some mushrooms, especially Amanita muscaria. It binds strongly to muscarinic receptors, which are abundant in the parasympathetic nervous system. Muscarine poisoning can produce symptoms such as excessive salivation, sweating, slowed heart rate, and difficulty breathing — all exaggerated “rest and digest” effects.
By understanding how these natural substances interact with acetylcholine receptors, researchers gained insight into how the parasympathetic and somatic nervous systems operate, and physicians developed drugs that can either mimic or block these effects.
Amines
Another important family of neurotransmitters is the amines, which are derived from single amino acids such as tyrosine, tryptophan, or histidine. Although they share a common chemical origin, their functions in the nervous system are diverse, ranging from the control of movement to the regulation of mood, alertness, and even sleep-wake cycles. What unites them is that each amine interacts with specific receptor subtypes, many of which are GPCRs. This allows amine neurotransmitters to produce slower but longer-lasting effects compared to the rapid responses triggered by ionotropic receptors.
Norepinephrine and epinephrine (also known as noradrenaline and adrenaline) act primarily through adrenergic receptors, which are divided into α (alpha) and β (beta) types. These receptors mediate the classic “fight-or-flight” responses of the sympathetic nervous system, such as raising heart rate, dilating airways, and mobilizing energy stores from fat and glycogen.
Dopamine, another amine derived from tyrosine, binds to a family of dopamine receptors (D₁–D₅). These GPCRs are concentrated in brain regions that control motivation, reward, and motor function. Disruption of dopamine signaling is linked to conditions such as Parkinson’s disease and schizophrenia.
Dopamine is often called the brain’s “reward chemical,” and one fun example of this is the way people crave chocolate. Eating chocolate activates taste receptors and stimulates the release of dopamine in brain regions involved in reward and pleasure, such as the nucleus accumbens. This dopamine surge creates feelings of enjoyment and reinforces the behavior — which is why many people reach for chocolate when they want a quick boost in mood.
Interestingly, chocolate also contains small amounts of compounds that can influence brain chemistry, including caffeine and theobromine, which may enhance its rewarding effects. While dopamine release from chocolate is not nearly as powerful as that caused by addictive drugs, the principle is the same: dopamine links a pleasurable experience with the motivation to repeat it.
This simple example shows how dopamine influences not just critical functions like movement, but also the small daily habits and cravings that shape our behavior.
Serotonin (5-HT), synthesized from the amino acid tryptophan, has one of the most diverse receptor families. It binds to both ionotropic receptors (such as 5-HT₃ channels) and multiple GPCR subtypes (including 5-HT₁ and 5-HT₂). Serotonin plays key roles in regulating mood, appetite, and sleep, and many antidepressant medications act by increasing serotonin availability in the synaptic cleft.
Finally, histamine, synthesized from histidine, binds to GPCRs (H₁–H₄). Beyond its well-known role in allergic reactions and inflammation, histamine also influences brain function by helping regulate wakefulness and arousal.
Summary of Amine Neurotransmitters and Receptors:
- Norepinephrine (NE) and epinephrine bind to adrenergic receptors (α and β types), which are all GPCRs. These receptors mediate sympathetic nervous system responses, such as increasing heart rate and mobilizing energy stores.
- Dopamine binds to dopamine receptors (D₁–D₅), all of which are GPCRs. Dopamine pathways regulate reward, motivation, and motor control.
- Serotonin (5-HT) binds to a large family of receptors, including ionotropic 5-HT₃ channels and numerous GPCR subtypes (e.g., 5-HT₁, 5-HT₂). Serotonin influences mood, appetite, and sleep.
- Histamine acts through GPCR subtypes (H₁–H₄). It is important in inflammation and wakefulness.
Parkinson’s disease is a progressive neurodegenerative disorder that highlights the importance of dopamine in the brain. In Parkinson’s, neurons in a midbrain region called the substantia nigra gradually die. These neurons normally release dopamine onto structures in the basal ganglia, which are critical for initiating and coordinating voluntary movements.
As dopamine levels fall, the balance of activity in motor pathways is disrupted. This leads to the hallmark symptoms of Parkinson’s disease: resting tremors, muscle rigidity, slowness of movement (bradykinesia), and difficulties with balance and coordination.
Treatment strategies often focus on boosting dopamine signaling. The most common therapy is L-DOPA (levodopa), a precursor molecule that can cross the blood–brain barrier and be converted into dopamine by surviving neurons. Other treatments target dopamine receptors directly with dopamine agonists, or reduce the breakdown of dopamine using MAO-B inhibitors. While these treatments can ease symptoms, they do not stop the underlying loss of dopaminergic neurons.
Parkinson’s disease illustrates how the loss of a single neurotransmitter system can disrupt entire neural circuits and profoundly affect daily life.
Amino Acids
While amino acids are best known as the building blocks of proteins, a few of them also serve directly as neurotransmitters, particularly in the central nervous system. These amino acid neurotransmitters form the foundation of excitatory and inhibitory signaling, providing the balance that keeps neural circuits stable.
The most important excitatory neurotransmitter in the brain is glutamate. When glutamate is released into a synapse, it binds to multiple receptor types. The two best-known ionotropic receptors are AMPA and NMDA receptors. AMPA receptors open quickly when glutamate binds, allowing sodium ions to enter and depolarize the postsynaptic cell. This rapid depolarization can then relieve the magnesium block that normally keeps NMDA receptors closed. Once unblocked, NMDA receptors open to allow calcium ions to enter. Calcium acts as a second messenger, triggering changes inside the cell that strengthen synaptic connections. This sequence — AMPA opening first, followed by NMDA activation — is the basis of synaptic plasticity, the ability of synapses to adapt with experience, which underlies learning and memory. Glutamate also binds to other ionotropic receptors (like kainate) and to metabotropic glutamate receptors (mGluRs) that fine-tune excitatory signaling more slowly.
Glutamate’s role as a neurotransmitter connects directly to everyday life through monosodium glutamate (MSG), a flavor enhancer commonly added to foods. MSG activates glutamate receptors on the tongue that contribute to the savory taste known as umami. This is the same basic chemical, glutamate, that excites neurons in the brain.
Although MSG has been controversial, scientific studies have not supported claims of widespread harmful effects in normal dietary amounts. Still, some individuals report sensitivity to large doses. The connection between MSG and glutamate neurotransmission is a reminder that the same molecules driving brain function also shape the sensory experiences of daily life.
On the inhibitory side, the primary neurotransmitter is GABA (gamma-aminobutyric acid). GABA has two major receptor types. GABA_A receptors are ionotropic chloride channels; when they open, chloride ions flow into the cell, making the inside more negative and less likely to fire an action potential. GABA_B receptors are GPCRs that activate slower inhibitory pathways. Together, GABA’s actions prevent runaway excitation and help maintain proper rhythm in neural circuits.
Because GABA is the brain’s main inhibitory neurotransmitter, anything that enhances its activity tends to calm neural circuits. Many anti-anxiety drugs, such as benzodiazepines (e.g., Valium®, Xanax®), work by increasing the effectiveness of GABA_A receptors. When these drugs bind, they make it easier for chloride channels to open in response to GABA, producing stronger inhibition. The result is a reduction in excessive neuronal firing, which helps relieve anxiety, promote relaxation, and even prevent seizures.
Alcohol also enhances GABA_A receptor activity, which is why drinking produces sedative and anxiolytic effects. However, prolonged alcohol exposure can alter GABA receptor sensitivity, contributing to tolerance and withdrawal symptoms. These clinical examples show how targeting GABA receptors can profoundly influence brain activity and behavior.
A third inhibitory neurotransmitter is glycine, which is especially important in the spinal cord and brainstem. Glycine receptors are ionotropic chloride channels, and like GABA_A receptors, they stabilize the resting membrane potential by making it harder for neurons to depolarize.
Without the balance between excitatory signals from glutamate and inhibitory signals from GABA and glycine, the nervous system would quickly become unstable, leading either to unresponsive silence or to uncontrolled seizures.
Summary of Amino Acid Neurotransmitters and Receptors
- Glutamate – Excitatory; binds ionotropic receptors (AMPA, NMDA, kainate) and metabotropic receptors (mGluRs); NMDA is critical for memory and plasticity.
- GABA (γ-aminobutyric acid) – Inhibitory; GABA_A receptors are ionotropic chloride channels, GABA_B receptors are GPCRs; both reduce excitability.
- Glycine – Inhibitory; binds ionotropic chloride channels in the spinal cord and brainstem; stabilizes resting potential.
Peptides
Neuropeptides include a wide variety of molecules such as substance P, endorphins, and vasopressin. These are often co-released with other neurotransmitters and bind almost exclusively to GPCRs. Peptide signaling tends to be slower but longer-lasting, often involved in regulating pain, stress, and emotional states.
Many neuropeptides influence how we experience pain and stress, and one of the most famous examples is the so-called runner’s high. During prolonged exercise, the brain releases endorphins, which are opioid-like peptides that bind to receptors in pain pathways. By reducing the perception of pain and creating a sense of euphoria, endorphins make it easier to keep going even when the body is under physical strain.
Other neuropeptides also shape emotional and social behavior. For example, vasopressin and oxytocin play important roles in bonding and trust, while substance P is critical for transmitting pain signals. These examples show how neuropeptides extend neurotransmission beyond quick electrical responses, influencing mood, motivation, and even social interactions in daily life.
The discovery of endorphins in the 1970s helped explain why the brain has natural opioid receptors. Endorphins normally reduce pain and produce feelings of well-being by binding to these receptors. However, many powerful drugs, both natural and synthetic, mimic their effects.
Opioid drugs such as morphine, oxycodone, and fentanyl bind to the same receptors as endorphins, providing strong pain relief. Unfortunately, because they also stimulate the brain’s reward pathways, these drugs can be highly addictive. Repeated use alters receptor sensitivity and neurotransmitter balance, creating dependence and making withdrawal difficult.
In recent years, the opioid crisis has underscored how a system designed to manage stress and pain can be hijacked by external substances. Treatments such as naloxone, which blocks opioid receptors, and methadone or buprenorphine, which act as safer substitutes, are used to manage overdoses and support recovery.
Neuropeptide systems like endorphins, which evolved to protect us, can also become targets for drugs that profoundly impact health and society.
Purines and Gases
Not all neurotransmitters fit the traditional mold of packaged vesicles and receptors. Some unconventional messengers, such as purines and gases, also play important roles in neural communication.
The purines ATP and adenosine can act as neurotransmitters. ATP often serves as a co-transmitter, released alongside more classical neurotransmitters, and it binds to P2X ionotropic receptors. Adenosine, on the other hand, acts through P1 GPCRs and usually produces inhibitory effects, slowing down neural activity. This system links neurotransmission directly to cellular metabolism, since ATP and adenosine are also central to energy use in cells.
The gases nitric oxide (NO) and carbon monoxide (CO) are even more unusual. Unlike most neurotransmitters, they are not stored in vesicles or released by exocytosis. Instead, they diffuse freely across membranes as soon as they are synthesized. Once inside a target cell, NO activates guanylyl cyclase, increasing cyclic GMP levels and producing effects such as smooth muscle relaxation and increased blood flow. These gaseous messengers are short-lived but powerful, often functioning in both the nervous and vascular systems.
The calming effects of adenosine become familiar to anyone who drinks coffee. As you stay awake longer, adenosine builds up in the brain, binding to its receptors and creating drowsiness. Caffeine works by blocking adenosine receptors, preventing this “sleep pressure” from being felt, and temporarily increasing alertness. This is why a morning cup of coffee feels like a mental boost — it interferes with purinergic signaling that normally promotes rest.
Nitric oxide (NO) is a vital regulator of blood vessel function. By activating guanylyl cyclase in smooth muscle cells, NO increases cyclic GMP levels, which leads to relaxation of the vessel walls and improved blood flow. This mechanism is essential for controlling blood pressure and ensuring that tissues receive enough oxygen and nutrients.
Clinically, drugs that enhance nitric oxide signaling are used to treat conditions such as angina pectoris (chest pain caused by reduced blood flow to the heart). Medications like nitroglycerin release NO, dilating the coronary blood vessels and relieving pain. Beyond heart disease, research has also linked nitric oxide pathways to immune defense, wound healing, and even learning and memory in the brain.
This broad impact shows how a gas that diffuses freely between cells can serve as a powerful signaling molecule, with implications far beyond traditional neurotransmission.
Nitric oxide (NO) is not only important in blood vessels but also plays a key role in the respiratory system. In the airways, NO helps relax smooth muscle, promoting bronchodilation and easier airflow. This signaling pathway is especially relevant in asthma, where inflammation and tightening of airway muscles make breathing difficult.
Clinically, nitric oxide levels in exhaled air are sometimes measured as a noninvasive biomarker of airway inflammation. Elevated exhaled NO can indicate worsening asthma, helping doctors adjust treatment. Medications that target smooth muscle relaxation, such as certain bronchodilators, may work in part by enhancing NO’s natural effects.
This example highlights how an unconventional neurotransmitter — a simple gas — contributes directly to respiratory health and provides useful diagnostic information in managing a common chronic disease.
Lipids
Certain lipid-derived molecules, such as endocannabinoids, also function as neurotransmitters. Endocannabinoids bind to cannabinoid receptors (CB₁ and CB₂), both GPCRs. These unusual messengers are often released from the postsynaptic cell and travel backward to the presynaptic terminal, providing retrograde signaling that can reduce neurotransmitter release.
The body’s endocannabinoid system is now known to regulate processes such as pain, appetite, memory, and stress. Plant-derived cannabinoids, such as tetrahydrocannabinol (THC) in cannabis, mimic these natural messengers by binding to CB₁ and CB₂ receptors. CB₁ receptors are concentrated in the brain and influence mood, coordination, and memory, while CB₂ receptors are found more in immune tissues and help regulate inflammation.
Medicinal cannabis and synthetic cannabinoids are being studied for conditions ranging from chronic pain to epilepsy. However, because THC can disrupt short-term memory, impair coordination, and alter motivation, recreational use carries risks, especially for adolescents whose brains are still developing. On the other hand, non-psychoactive cannabinoids such as CBD (cannabidiol) do not strongly bind to CB₁ receptors and are being investigated for their potential therapeutic effects without producing a “high.”
Together, these clinical insights reveal how lipid-derived neurotransmitters open the door to both natural mechanisms of resilience and complex medical debates.
Table 8.2 Neurotransmitters
| Chemical | Receptor | Type | Receptor Location | Key Agonists, Antagonists, and Potentiators |
|---|---|---|---|---|
| Acetylcholine (ACh) | Cholinergic | |||
| – Nicotinic (nAChR) | ICR (Na⁺, K⁺) | Skeletal muscles, autonomic neurons, CNS | Agonist: nicotine; Antagonists: curare, α-bungarotoxin | |
| – Muscarinic (M) | GPCR | Smooth and cardiac muscle, endocrine and exocrine glands, CNS | Agonist: muscarine; Antagonist: atropine | |
| Amines | ||||
| Norepinephrine (NE), Epinephrine (E) | Adrenergic (α, β) | GPCR | Smooth and cardiac muscle, glands, CNS | Antagonists: α-receptors: ergotamine, phentolamine; β-receptors: propranolol |
| Dopamine (DA) | Dopamine (D) | GPCR | CNS | Agonist: bromocriptine; Antagonists: antipsychotic drugs |
| Serotonin (5-HT) | Serotonergic (5-HT) | ICR (Na⁺, K⁺), GPCR | CNS | Agonist: sumatriptan; Antagonist: LSD |
| Histamine (H) | Histamine (H) | GPCR | CNS | Antagonists: ranitidine (Zantac®), cimetidine (Tagamet®) |
| Amino Acids | ||||
| Glutamate | Glutamatergic ionotropic (iGluR) | CNS | ||
| – AMPA | ICR (Na⁺, K⁺) | CNS | Agonist: quisqualate | |
| – NMDA | ICR (Na⁺, K⁺) | CNS | Potentiator: serine | |
| – Metabotropic (mGluR) | GPCR | CNS | Potentiator: glycine | |
| GABA (γ-aminobutyric acid) | GABA | ICR (Cl⁻), GPCR | CNS | Antagonist: picrotoxin; Potentiators: alcohol, barbiturates |
| Glycine | Glycine (GlyR) | ICR (Cl⁻) | CNS | Antagonist: strychnine |
| Purines | ||||
| Adenosine | Purine (P) | GPCR | CNS | |
| Gases | ||||
| Nitric oxide (NO) | None | N/A | N/A | N/A |
Neurotransmitters Synthesis and Vesicle Storage
When viewed under an electron microscope, the axon terminal of a presynaptic neuron reveals a busy landscape packed with synaptic vesicles. These vesicles store neurotransmitters and release them into the synaptic cleft when a signal arrives. Some vesicles are already docked at specialized regions of the presynaptic membrane, known as active zones, ready to fuse and release their contents instantly. Others form a reserve pool, clustering nearby until they are needed. Surrounding these vesicles are mitochondria, which supply the ATP required for vesicle loading, transport, and the energy demands of neurotransmission.
In this section, we will explore how neurotransmitters are synthesized, stored, released, and eventually terminated. The process begins with synthesis, which depends on the type of neurotransmitter.
Peptide neurotransmitters must be made in the neuron’s cell body, since axon terminals lack the organelles for protein synthesis. As with hormones, peptide neurotransmitters are first produced as large propeptides that are packaged into vesicles along with modifying enzymes. These vesicles travel down the axon by fast axonal transport. Once inside the vesicle, the propeptide is cleaved into one or more active peptides. For example, a single propeptide may yield several active messengers that are later released together, much like the way endocrine cells process pre-prohormones into multiple active hormones.
In contrast, small-molecule neurotransmitters such as acetylcholine, amines, and purines are synthesized and packaged locally in the axon terminal. The enzymes required for their synthesis are first made in the cell body, transported to the terminal by slow axonal transport (Figure 8.8), and then used in the cytosol to assemble neurotransmitters directly at the site of release. This arrangement ensures that neurons can maintain a ready supply of both peptide and small-molecule neurotransmitters, tailored to their specific signaling needs.
Neurotransmitter Release
As seen in Figure 8.23a), when an action potential reaches the axon terminal, the process of neurotransmitter release begins. (1) The arrival of the action potential depolarizes the terminal membrane. (2) This change in voltage opens calcium-VGCs, allowing Ca²⁺ ions to rush into the cell. (3) The sudden rise in calcium triggers exocytosis, where synaptic vesicles fuse with the presynaptic membrane and release their stored neurotransmitters. (4) The neurotransmitters diffuse across the synaptic cleft and bind to ligand-receptors on the postsynaptic membrane. (5) This binding event initiates a response in the postsynaptic cell, such as depolarization or hyperpolarization, depending on the receptor type.
Exocytosis is a universal cellular mechanism, but in neurons it occurs with remarkable speed to keep up with rapid signaling demands. Certain neurotoxins, such as tetanus toxin and botulinum toxin, disrupt this process by targeting the proteins that control vesicle fusion, effectively blocking neurotransmitter release
Neurotransmitter Termination
For signaling to remain precise, neurotransmitter action must be brief. The process of termination, shown in Figure 8.23b), ensures that the cleft is cleared quickly so that new signals can be distinguished. (1) Some neurotransmitters are taken back into the presynaptic terminal for reuse or transported into nearby glial cells. (2) Others are broken down by enzymes (acetylcholinesterase) in the synaptic cleft, which inactivates them. (3) Still others simply diffuse away from the cleft, reducing their local concentration until the signal fades. These steps guarantee that communication between neurons remains fast, controlled, and ready for the next signal.
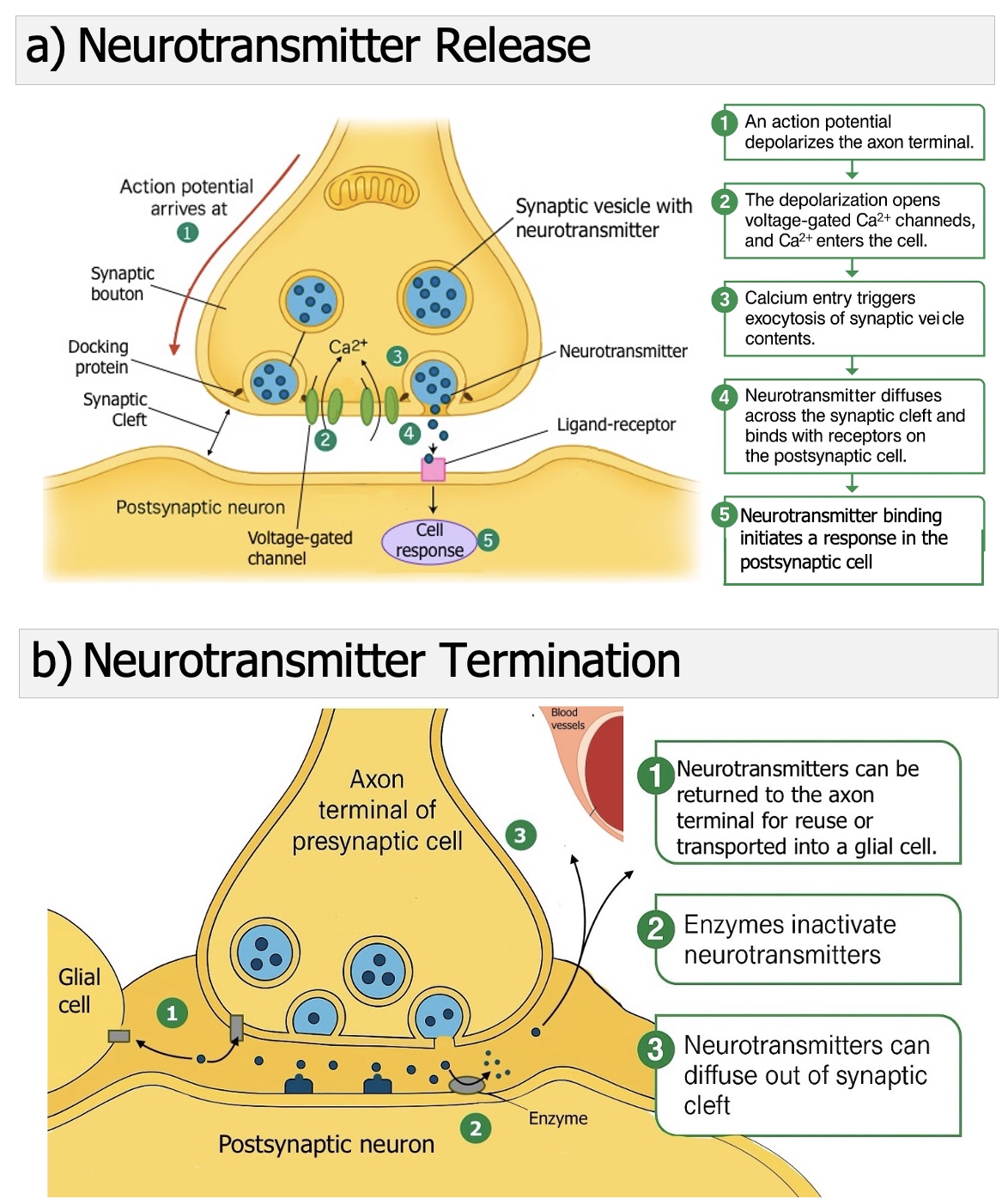
Figure 8.23: Neurotransmitter Release and Termination. When an action potential reaches the axon terminal, voltage-gated Ca²⁺ channels open and Ca²⁺ enters the cell. Ca²⁺ triggers synaptic vesicles to fuse with the presynaptic membrane and release neurotransmitter into the synaptic cleft. Neurotransmitter binds receptors on the postsynaptic membrane to change ion flow and cell activity. The signal ends when neurotransmitter is cleared by reuptake into the presynaptic cell or glia, enzymatic breakdown in the cleft, or diffusion away (Mac Leod, 2025, licensed CC-BY)
A good example of neurotransmitter termination is acetylcholine (ACh), shown in Figure 8.24. After being released into the synaptic cleft, ACh is rapidly broken down by the enzyme acetylcholinesterase (AChE), which is located in the extracellular matrix and in the postsynaptic membrane. This breakdown produces choline and acetyl CoA. The choline is then transported back into the presynaptic terminal by a sodium-dependent cotransporter. Once inside, choline is reused to make new acetylcholine, allowing the neuron to recycle its resources and prepare for the next signal.
Other neurotransmitters are cleared in a similar way. For example, the signal from norepinephrine is stopped when the intact neurotransmitter is taken back up into the presynaptic terminal using a cotransporter. Once inside, norepinephrine can either be returned to synaptic vesicles for future release or broken down by enzymes such as monoamine oxidase (MAO), which is found in mitochondria. This recycling system ensures that neurotransmitters or their components can be reused efficiently to refill empty vesicles and maintain continuous communication.
.jpeg?revision=1&size=bestfit&width=575)
Figure 8.24: Synthesis and Recycling of Acetylcholine. Acetylcholine is broken down by acetylcholinesterase, and choline is recycled into the presynaptic terminal. Norepinephrine is taken back up, where it is either repackaged into vesicles or broken down by monoamine oxidase (MAO) (Mac Leod, 2025, licensed CC-BY)
Stronger Stimuli Release More Neurotransmitter
When an action potential reaches the axon terminal, it always releases about the same amount of neurotransmitter. Neurons, however, have another way to signal the strength and duration of a stimulus: they vary the frequency of action potentials. A longer-lasting stimulus produces a longer train of action potentials, while a stronger stimulus increases the number of action potentials fired per second. This higher firing rate delivers more signals to the terminal, which can result in greater neurotransmitter release, as shown in Figure 8.25.
For example, think about how a sensory neuron communicates the intensity of a touch or sound to the CNS. If a graded potential that reaches the trigger zone just passes threshold, it does not generate a single action potential but rather a small burst. As the graded potential becomes stronger, it drives action potentials at a faster pace, sending a clearer “strong signal” to the CNS.
Patterns of activity in the brain are even more diverse. Some neurons are tonically active, firing steady trains of action potentials like a metronome. Others show bursting patterns, producing short bursts of action potentials separated by quiet intervals, almost like rhythmic drumming. These patterns arise because different neurons use slightly different ion channels, which vary in how quickly they open or close, what voltage activates them, and how they respond to neuromodulators. This diversity makes signaling in the brain far more complex and flexible than the relatively simple firing pattern of a somatic motor neuron.

Figure 8.25: Stimulus Strength and Neurotransmitter Release. Stronger stimuli produce action potentials at a higher frequency. More action potentials arriving at the axon terminal per unit time allow more Ca²⁺ to enter, which increases synaptic vesicle fusion and releases more neurotransmitter. This creates a larger postsynaptic response (stronger signaling) without changing the size of each individual action potential (Mac Leod, 2025, licensed CC-BY)
Check Your Understanding
- What role does the sodium–potassium pump play in maintaining the resting membrane potential of a neuron?
- How does a graded potential differ from an action potential in terms of size, distance traveled, and strength?
- Explain what happens at the threshold potential that makes the action potential an all-or-none event.
- Why does the refractory period prevent action potentials from traveling backward along the axon?
- Describe how neurotransmitters are released from the presynaptic terminal and how their actions are terminated in the synaptic cleft.
- How does the nervous system use the frequency of action potentials to code the strength of a stimulus?
Glossary
- Action potential (AK-shun poh-TEN-shul): A rapid, all-or-none electrical signal that travels along the axon of a neuron.
- After-hyperpolarization (AF-ter HAI-per-poh-luh-rih-ZAY-shun): The brief dip below resting membrane potential that follows repolarization.
- AMPA receptor (AM-puh ree-SEP-ter): A glutamate receptor that opens quickly to allow sodium ions to enter, producing fast excitatory signals.
- Axon terminal (AK-son TER-muh-nuhl): The enlarged end of an axon where neurotransmitters are stored in vesicles and released.
- Depolarization (DEE-poh-luh-rih-ZAY-shun): A decrease in the difference in charge across the membrane, making the inside less negative.
- Excitatory postsynaptic potential (ik-SAI-tuh-tor-ee pohst-sin-AP-tik poh-TEN-shul, EPSP): A graded depolarization in the postsynaptic membrane that brings the neuron closer to threshold.
- GABA (GAM-uh): The main inhibitory neurotransmitter in the brain.
- GABA_A receptor (GAM-uh-AY ree-SEP-ter): An ionotropic receptor that opens chloride channels to inhibit neural activity.
- GABA_B receptor (GAM-uh-BEE ree-SEP-ter): A metabotropic G-protein-coupled receptor that produces slower inhibitory effects.
- Glycine (GLY-seen): An inhibitory neurotransmitter especially important in the spinal cord and brainstem.
- Hyperpolarization (HAI-per-poh-luh-rih-ZAY-shun): An increase in the difference in charge across the membrane, making the inside more negative.
- Inhibitory postsynaptic potential (in-HIB-ih-tor-ee pohst-sin-AP-tik poh-TEN-shul, IPSP): A graded hyperpolarization that moves the neuron farther from threshold.
- Muscarinic receptor (MUS-kuh-RIN-ik ree-SEP-ter): A type of acetylcholine receptor that is G-protein-coupled, slower-acting, and found in target organs.
- Myelin (MY-uh-lin): Insulating material around axons that speeds action potential conduction.
- Neurotransmitter (NUR-oh-trans-mit-er): A chemical messenger released by neurons to communicate with other cells.
- Nicotine receptor (NIK-oh-teen ree-SEP-ter) [Nicotinic receptor]: A fast, ionotropic acetylcholine receptor activated by nicotine.
- NMDA receptor (EN-em-DEE-AY ree-SEP-ter): A glutamate receptor that allows calcium entry, essential for learning and memory.
- Refractory period (ree-FRAK-tor-ee PEER-ee-uhd): The time after an action potential during which a neuron cannot fire another, or requires a stronger-than-normal stimulus.
- Resting membrane potential (RES-ting MEM-brayn poh-TEN-shul): The steady electrical difference across a neuron’s membrane, usually about –70 mV.
- Saltatory conduction (SAL-tuh-tor-ee kon-DUK-shun): Rapid conduction of action potentials in myelinated axons, where the signal “jumps” between nodes of Ranvier.
- Summation (suh-MAY-shun): The combining of graded potentials at the axon hillock, which can bring the membrane to threshold.
- Synapse (SIN-aps): The junction where a neuron communicates with another cell.
- Threshold potential (THRESH-hold poh-TEN-shul): The membrane voltage (around –55 mV) that must be reached to trigger an action potential.

