8.2: Isolating Genomic DNA
- Page ID
- 4108
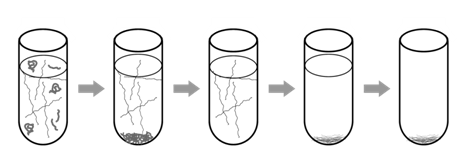
DNA purification strategies rely on the chemical properties of DNA that distinguish it from other molecules in the cell, namely that it is a very long, negatively charged molecule. To extract purified DNA from a tissue sample, cells are broken open by grinding or lysing in a solution that contains chemicals that protect the DNA while disrupting other components of the cell (Figure \(\PageIndex{2}\)). These chemicals may include detergents, which dissolve lipid membranes and denature proteins. A cation such as Na+ helps to stabilize the negatively charged DNA and separate it from proteins such as histones. A chelating agent, such as EDTA, is added to protect DNA by sequestering Mg2+ ions, which can otherwise serve as a necessary co-factor for nucleases (enzymes that digest DNA). As a result, free, double-stranded DNA molecules are released from the chromatin into the extraction buffer, which also contains proteins and all other cellular components. (The basics of this procedure can be done with household chemicals and are presented on YouTube.)
The free DNA molecules are subsequently isolated by one of several methods. Commonly, proteins are removed by adjusting the salt concentration so they precipitate. The supernatant, which contains DNA and other, smaller metabolites, is then mixed with ethanol, which causes the DNA to precipitate. A small pellet of DNA can be collected by centrifugation, and after removal of the ethanol, the DNA pellet can be dissolved in water (usually with a small amount of EDTA and a pH buffer) for the use in other reactions. Note that this process has purified all of the DNA from a tissue sample; if we want to further isolate a specific gene or DNA fragment, we must use additional techniques, such as PCR.