18.5: Fermentation
- Page ID
- 3417
\( \newcommand{\vecs}[1]{\overset { \scriptstyle \rightharpoonup} {\mathbf{#1}} } \)
\( \newcommand{\vecd}[1]{\overset{-\!-\!\rightharpoonup}{\vphantom{a}\smash {#1}}} \)
\( \newcommand{\dsum}{\displaystyle\sum\limits} \)
\( \newcommand{\dint}{\displaystyle\int\limits} \)
\( \newcommand{\dlim}{\displaystyle\lim\limits} \)
\( \newcommand{\id}{\mathrm{id}}\) \( \newcommand{\Span}{\mathrm{span}}\)
( \newcommand{\kernel}{\mathrm{null}\,}\) \( \newcommand{\range}{\mathrm{range}\,}\)
\( \newcommand{\RealPart}{\mathrm{Re}}\) \( \newcommand{\ImaginaryPart}{\mathrm{Im}}\)
\( \newcommand{\Argument}{\mathrm{Arg}}\) \( \newcommand{\norm}[1]{\| #1 \|}\)
\( \newcommand{\inner}[2]{\langle #1, #2 \rangle}\)
\( \newcommand{\Span}{\mathrm{span}}\)
\( \newcommand{\id}{\mathrm{id}}\)
\( \newcommand{\Span}{\mathrm{span}}\)
\( \newcommand{\kernel}{\mathrm{null}\,}\)
\( \newcommand{\range}{\mathrm{range}\,}\)
\( \newcommand{\RealPart}{\mathrm{Re}}\)
\( \newcommand{\ImaginaryPart}{\mathrm{Im}}\)
\( \newcommand{\Argument}{\mathrm{Arg}}\)
\( \newcommand{\norm}[1]{\| #1 \|}\)
\( \newcommand{\inner}[2]{\langle #1, #2 \rangle}\)
\( \newcommand{\Span}{\mathrm{span}}\) \( \newcommand{\AA}{\unicode[.8,0]{x212B}}\)
\( \newcommand{\vectorA}[1]{\vec{#1}} % arrow\)
\( \newcommand{\vectorAt}[1]{\vec{\text{#1}}} % arrow\)
\( \newcommand{\vectorB}[1]{\overset { \scriptstyle \rightharpoonup} {\mathbf{#1}} } \)
\( \newcommand{\vectorC}[1]{\textbf{#1}} \)
\( \newcommand{\vectorD}[1]{\overrightarrow{#1}} \)
\( \newcommand{\vectorDt}[1]{\overrightarrow{\text{#1}}} \)
\( \newcommand{\vectE}[1]{\overset{-\!-\!\rightharpoonup}{\vphantom{a}\smash{\mathbf {#1}}}} \)
\( \newcommand{\vecs}[1]{\overset { \scriptstyle \rightharpoonup} {\mathbf{#1}} } \)
\(\newcommand{\longvect}{\overrightarrow}\)
\( \newcommand{\vecd}[1]{\overset{-\!-\!\rightharpoonup}{\vphantom{a}\smash {#1}}} \)
\(\newcommand{\avec}{\mathbf a}\) \(\newcommand{\bvec}{\mathbf b}\) \(\newcommand{\cvec}{\mathbf c}\) \(\newcommand{\dvec}{\mathbf d}\) \(\newcommand{\dtil}{\widetilde{\mathbf d}}\) \(\newcommand{\evec}{\mathbf e}\) \(\newcommand{\fvec}{\mathbf f}\) \(\newcommand{\nvec}{\mathbf n}\) \(\newcommand{\pvec}{\mathbf p}\) \(\newcommand{\qvec}{\mathbf q}\) \(\newcommand{\svec}{\mathbf s}\) \(\newcommand{\tvec}{\mathbf t}\) \(\newcommand{\uvec}{\mathbf u}\) \(\newcommand{\vvec}{\mathbf v}\) \(\newcommand{\wvec}{\mathbf w}\) \(\newcommand{\xvec}{\mathbf x}\) \(\newcommand{\yvec}{\mathbf y}\) \(\newcommand{\zvec}{\mathbf z}\) \(\newcommand{\rvec}{\mathbf r}\) \(\newcommand{\mvec}{\mathbf m}\) \(\newcommand{\zerovec}{\mathbf 0}\) \(\newcommand{\onevec}{\mathbf 1}\) \(\newcommand{\real}{\mathbb R}\) \(\newcommand{\twovec}[2]{\left[\begin{array}{r}#1 \\ #2 \end{array}\right]}\) \(\newcommand{\ctwovec}[2]{\left[\begin{array}{c}#1 \\ #2 \end{array}\right]}\) \(\newcommand{\threevec}[3]{\left[\begin{array}{r}#1 \\ #2 \\ #3 \end{array}\right]}\) \(\newcommand{\cthreevec}[3]{\left[\begin{array}{c}#1 \\ #2 \\ #3 \end{array}\right]}\) \(\newcommand{\fourvec}[4]{\left[\begin{array}{r}#1 \\ #2 \\ #3 \\ #4 \end{array}\right]}\) \(\newcommand{\cfourvec}[4]{\left[\begin{array}{c}#1 \\ #2 \\ #3 \\ #4 \end{array}\right]}\) \(\newcommand{\fivevec}[5]{\left[\begin{array}{r}#1 \\ #2 \\ #3 \\ #4 \\ #5 \\ \end{array}\right]}\) \(\newcommand{\cfivevec}[5]{\left[\begin{array}{c}#1 \\ #2 \\ #3 \\ #4 \\ #5 \\ \end{array}\right]}\) \(\newcommand{\mattwo}[4]{\left[\begin{array}{rr}#1 \amp #2 \\ #3 \amp #4 \\ \end{array}\right]}\) \(\newcommand{\laspan}[1]{\text{Span}\{#1\}}\) \(\newcommand{\bcal}{\cal B}\) \(\newcommand{\ccal}{\cal C}\) \(\newcommand{\scal}{\cal S}\) \(\newcommand{\wcal}{\cal W}\) \(\newcommand{\ecal}{\cal E}\) \(\newcommand{\coords}[2]{\left\{#1\right\}_{#2}}\) \(\newcommand{\gray}[1]{\color{gray}{#1}}\) \(\newcommand{\lgray}[1]{\color{lightgray}{#1}}\) \(\newcommand{\rank}{\operatorname{rank}}\) \(\newcommand{\row}{\text{Row}}\) \(\newcommand{\col}{\text{Col}}\) \(\renewcommand{\row}{\text{Row}}\) \(\newcommand{\nul}{\text{Nul}}\) \(\newcommand{\var}{\text{Var}}\) \(\newcommand{\corr}{\text{corr}}\) \(\newcommand{\len}[1]{\left|#1\right|}\) \(\newcommand{\bbar}{\overline{\bvec}}\) \(\newcommand{\bhat}{\widehat{\bvec}}\) \(\newcommand{\bperp}{\bvec^\perp}\) \(\newcommand{\xhat}{\widehat{\xvec}}\) \(\newcommand{\vhat}{\widehat{\vvec}}\) \(\newcommand{\uhat}{\widehat{\uvec}}\) \(\newcommand{\what}{\widehat{\wvec}}\) \(\newcommand{\Sighat}{\widehat{\Sigma}}\) \(\newcommand{\lt}{<}\) \(\newcommand{\gt}{>}\) \(\newcommand{\amp}{&}\) \(\definecolor{fillinmathshade}{gray}{0.9}\)- Define fermentation.
- State the mechanism for ATP generation during fermentation.
- Briefly describethe function of glycolysis during fermentation and indicate the reactants and products.
- Compare the maximum yield of ATP from one molecule of glucose for aerobic respiration and for fermentation.
Fermentation is an anaerobic breakdown of carbohydrates in which an organic molecule is the final electron acceptor. It does not involve an electron transport system. Furthermore,:
- Fermentation is a partial breakdown of glucose producing only 2 net ATP's per glucose by way of substrate-level phosphorylation ;
- Fermentation involves only glycolysis; and
- Fermentation is found in bacteria that are obligate anaerobes and facultative anaerobes.
Glycolysis during Fermentation
Function: As during aerobic respiration, glycolysis is a partial breakdown of a six-carbon glucose molecule into two, three-carbon molecules of pyruvate, 2NADH +2H+, and 2 net ATP as a result of substrate-level phosphorylation, as shown in (see Figure \(\PageIndex{1}\) and Figure \(\PageIndex{2}\)).
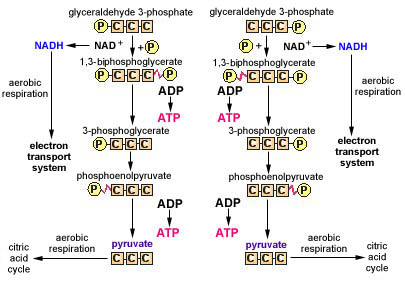
Glycolysis occurs in the cytoplasm of the cell. As mentioned above, the overall reaction is:
glucose (6C) + 2 NAD+ +2 ADP +2 inorganic phosphates (Pi)
yields 2 pyruvate (3C) + 2 NADH + 2 H+ + 2 net ATP
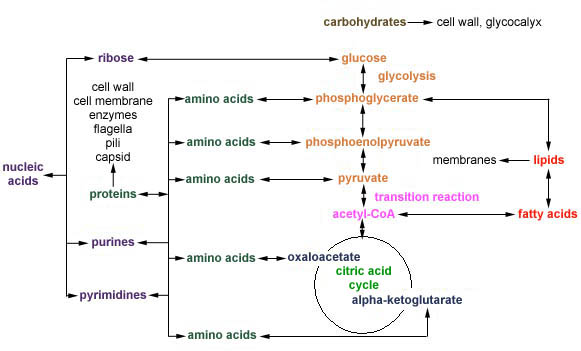
Glycolysis also produces a number of key precursor metabolites, as shown in Figure \(\PageIndex{3}\).
Since there is no electron transport system, the protons and electrons donated by certain intermediate precursor molecules during glycolysis generate no additional molecules of ATP. Instead, they combine with the coenzyme NAD+, the organic molecule which serves as the final electron and proton acceptor, reducing it to NADH + H+ (see Figure \(\PageIndex{1}\) and Figure \(\PageIndex{2}\)).

Glycolysis is a partial breakdown of a six-carbon glucose molecule into two, three-carbon molecules of pyruvate, 2NADH +2H+, and 2 net ATP as a result of substrate-level phosphorylation. Glycolysis occurs in the cytoplasm of the cell.
The 2 pyruvic acids are then converted into one of many different fermentation end products in several non-energy-producing steps.
Fermentation end products
Some fermentation end products produced by microorganisms are very beneficial to humans and are the basis of a number of industries (brewing industry, dairy industry, etc.). Fermentation is used in the production of many food products including bread, alcohol, yogurt, sour cream, cheeses, vinegar, sauerkraut, pickles, olives, soy sauce, poi, and kimchi. Examples of microbial fermentation end products include:
- Saccharomyces: ethyl alcohol and CO2
- Streptococcus and Lactobacillus: lactic acid
- Propionibacterium: proprionic acid, acetic acid, and CO2
- Escherichia coli: acetic acid, lactic acid, succinic acid, ethyl alcohol, CO2, and H2
- Enterobacter: formic acid, ethyl alcohol, 2,3-butanediol, lactic acid, CO2, and H2
- Clostridium: butyric acid, butyl alcohol, acetone, isopropyl alcohol, CO2, and H2
Summary
- Fermentation is an anaerobic breakdown of carbohydrates in which an organic molecules the final electron acceptor and does not involve an electron transport system.
- Fermentation is a partial breakdown of glucose producing only 2 net ATP's per glucose by way of substrate-level phosphorylation, involves only glycolysis, and is found in anaerobic and facultative anaerobic bacteria.
- The overall reaction is glucose (6C) + 2 NAD+ +2 ADP +2 inorganic phosphates (Pi) yields 2 pyruvate (3C) + 2 NADH + 2 H+ + 2 net ATP.
- Glycolysis also produces a number of key precursor metabolites.
- Some fermentation end products produced by microorganisms are very beneficial to humans and are the basis of a number of industries (brewing industry, dairy industry, etc.).


