17.5: Phosphorylation Mechanisms for Generating ATP
- Page ID
- 3404
\( \newcommand{\vecs}[1]{\overset { \scriptstyle \rightharpoonup} {\mathbf{#1}} } \)
\( \newcommand{\vecd}[1]{\overset{-\!-\!\rightharpoonup}{\vphantom{a}\smash {#1}}} \)
\( \newcommand{\id}{\mathrm{id}}\) \( \newcommand{\Span}{\mathrm{span}}\)
( \newcommand{\kernel}{\mathrm{null}\,}\) \( \newcommand{\range}{\mathrm{range}\,}\)
\( \newcommand{\RealPart}{\mathrm{Re}}\) \( \newcommand{\ImaginaryPart}{\mathrm{Im}}\)
\( \newcommand{\Argument}{\mathrm{Arg}}\) \( \newcommand{\norm}[1]{\| #1 \|}\)
\( \newcommand{\inner}[2]{\langle #1, #2 \rangle}\)
\( \newcommand{\Span}{\mathrm{span}}\)
\( \newcommand{\id}{\mathrm{id}}\)
\( \newcommand{\Span}{\mathrm{span}}\)
\( \newcommand{\kernel}{\mathrm{null}\,}\)
\( \newcommand{\range}{\mathrm{range}\,}\)
\( \newcommand{\RealPart}{\mathrm{Re}}\)
\( \newcommand{\ImaginaryPart}{\mathrm{Im}}\)
\( \newcommand{\Argument}{\mathrm{Arg}}\)
\( \newcommand{\norm}[1]{\| #1 \|}\)
\( \newcommand{\inner}[2]{\langle #1, #2 \rangle}\)
\( \newcommand{\Span}{\mathrm{span}}\) \( \newcommand{\AA}{\unicode[.8,0]{x212B}}\)
\( \newcommand{\vectorA}[1]{\vec{#1}} % arrow\)
\( \newcommand{\vectorAt}[1]{\vec{\text{#1}}} % arrow\)
\( \newcommand{\vectorB}[1]{\overset { \scriptstyle \rightharpoonup} {\mathbf{#1}} } \)
\( \newcommand{\vectorC}[1]{\textbf{#1}} \)
\( \newcommand{\vectorD}[1]{\overrightarrow{#1}} \)
\( \newcommand{\vectorDt}[1]{\overrightarrow{\text{#1}}} \)
\( \newcommand{\vectE}[1]{\overset{-\!-\!\rightharpoonup}{\vphantom{a}\smash{\mathbf {#1}}}} \)
\( \newcommand{\vecs}[1]{\overset { \scriptstyle \rightharpoonup} {\mathbf{#1}} } \)
\( \newcommand{\vecd}[1]{\overset{-\!-\!\rightharpoonup}{\vphantom{a}\smash {#1}}} \)
Define photophosphorylation. Describe substrate-level phosphorylation and name to energy-generating pathways in which this occurs. Define oxidative phosphorylation. Name the two components of a hydrogen atom. Describe an oxidation-reduction reaction. Define dehydrogenation and hydrogenation. State the function of the following coenzymes and give their reduced form: - NAD+
- FAD
- NADP+
Briefly describe proton motive force and how it develops within a cell. Describe an electron transport chain and state its cellular function. Briefly describethe chemiosmotic theory of generation of ATP as a result of an electron transport chain. State the function of ATP synthases.
- NAD+
- FAD
- NADP+
Substrate-Level Phosphorylation
Substrate-level phosphorylation is the production of ATP from ADP by a direct transfer of a high-energy phosphate group from a phosphorylated intermediate metabolic compound in an exergonic catabolic pathway as shown in Figure \(\PageIndex{2}\). Such intermediate compounds are sometimes called high-energy transfer compounds (HETCs) and several HETCs are found as intermediates during glycolysis and aerobic respiration .
| Flash animation illustrating substrate-level phosphorylation. |
| html5 version of animation for iPad illustrating substrate-level phosphorylation. |
Oxidative Phosphorylation
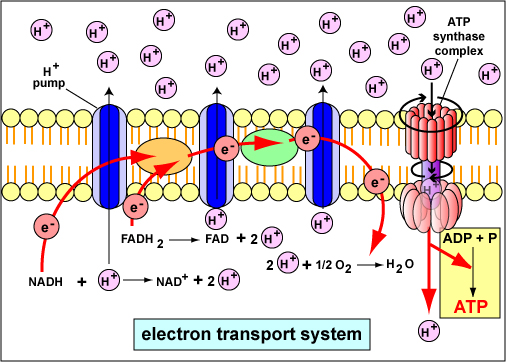
Oxidative phosphorylation is the production of ATP using energy derived from the transfer of electrons in an electron transport system and occurs by chemiosmosis.
To understand oxidative phosphorylation, it is important to first review the hydrogen atom and the process of oxidation and reduction. An atom of hydrogen contains only one proton (H+) and one electron (e-). Therefore, the term proton and the term hydrogen ion (H+) are interchangeable. Also remember that electrons have stored energy, or potential energy, ready to do work and when an atom or molecule loses that electron (becomes oxidized) that energy is released and able to do cellular work.
Oxidation-reduction reactions are coupled chemical reactions in which one atom or molecule loses one or more electrons (oxidation ) while another atom or molecule gains those electrons (reduction ). The compound that loses electrons becomes oxidized; the compound that gains those electrons becomes reduced. In covalent compounds, however, it is usually easier to lose a whole hydrogen (H) atom - a proton and an electron - rather than just an electron. An oxidation reaction during which both a proton and an electron are lost is called dehydrogenation . A reduction reaction during which both a proton and an electron are gained is called hydrogenation .
Cells use specific molecules to carry the electrons that are removed during the oxidation of an energy source. These molecules are called electron carriers and they alternately become oxidized and reduced during electron and proton transfer. These include three freely diffusible coenzymes known as NAD+, FAD, and NADP+. The reduced forms of these coenzymes (NADH, FADH2, and NADPH) have reducing power because their bonds contain a form of usable energy.
- NAD+ , or nicotinamide adenine dinucleotide, is a coenzyme that often works in conjunction with an enzyme called a dehydrogenase. The enzyme removes two hydrogen atoms (2H+ and 2e-) from its substrate. Both electrons but only one proton are accepted by the NAD+ to produce its reduced form, NADH, plus H+. NADH is used to generate proton motive force (discussed below) that can drive the synthesis of ATP.
- FAD , or flavin adenine dinucleotide, is a coenzyme that also works in conjunction with an enzyme called a dehydrogenase. The enzyme removes two hydrogen atoms (2H+ and 2e-) from its substrate. Both electrons and both protons are accepted by the FAD to produce its reduced form, FADH2. FADH2 is used to generate proton motive force (discussed below) that can drive the synthesis of ATP.
- NADP+, or nicotinamide adenine dinucleotide phosphate, is a coenzyme that uses dehydrogenase to remove two hydrogen atoms (2H+ and 2e-) from its substrate. Both electrons but only one proton are accepted by the NADP+ to produce its reduced form, NADPH, plus H+. NADPH is not used for ATP synthesis but its electrons provide the energy for certain biosynthesis reactions such as ones involved in photosynthesis.
During the process of aerobic respiration, discussed in the next section, coupled oxidation-reduction reactions and electron carriers are often part of what is called an electron transport chain , a series of electron carriers that eventually transfers electrons from NADH and FADH2 to oxygen. The diffusible electron carriers NADH and FADH2 carry hydrogen atoms (protons and electrons) from substrates in exergonic catabolic pathways such as glycolysis and the citric acid cycle to other electron carriers that are embedded in membranes. These membrane-associated electron carriers include flavoproteins, iron-sulfur proteins, quinones, and cytochromes. The last electron carrier in the electron transport chain transfers the electrons to the terminal electron acceptor, oxygen.
The chemiosmotic theory explains the functioning of electron transport chains. According to this theory, the transfer of electrons down an electron transport system through a series of oxidation-reduction reactions releases energy (Figure \(\PageIndex{1}\)). This energy allows certain carriers in the chain to transport hydrogen ions (H+ or protons) across a membrane.
Depending on the type of cell, the electron transport chain may be found in the cytoplasmic membrane, the inner membrane of mitochondria, and the inner membrane of chloroplasts.
- In prokaryotic cells, the protons are transported from the cytoplasm of the bacterium across the cytoplasmic membrane to the periplasmic space located between the cytoplasmic membrane and the cell wall.
- In eukaryotic cells, protons are transported from the matrix of the mitochondria across the inner mitochondrial membrane to the intermembrane space located between the inner and outer mitochondrial membranes.
- In plant cells and the cells of algae, protons are transported from the stroma of the chloroplast across the thylakoid membrane into the interior space of the thylakoid.
As the hydrogen ions accumulate on one side of a membrane, the concentration of hydrogen ions creates an electrochemical gradient or potential difference (voltage) across the membrane. (The fluid on the side of the membrane where the protons accumulate acquires a positive charge; the fluid on the opposite side of the membrane is left with a negative charge.) The energized state of the membrane as a result of this charge separation is called proton motive force or PMF.
This proton motive force provides the energy necessary for enzymes called ATP synthases (Figure \(\PageIndex{5}\)), also located in the membranes mentioned above, to catalyze the synthesis of ATP from ADP and phosphate. This generation of ATP occurs as the protons cross the membrane through the ATP synthase complexes and re-enter either the bacterial cytoplasm (Figure \(\PageIndex{5}\)), the matrix of the mitochondria, or the stroma of the chloroplasts. As the protons move down the concentration gradient through the ATP synthase, the energy released causes the rotor and rod of the ATP synthase to rotate. The mechanical energy from this rotation is converted into chemical energy as phosphate is added to ADP to form ATP.
Proton motive force is also used to transport substances across membranes during active transport and to rotate bacterial flagella.
At the end of the electron transport chain involved in aerobic respiration, the last electron carrier in the membrane transfers 2 electrons to half an oxygen molecule (an oxygen atom) that simultaneously combines with 2 protons from the surrounding medium to produce water as an end product (Figure \(\PageIndex{3}\)). The electron transport chains involved in photosynthesis ultimately transfer 2 electrons to NADP+ that simultaneously combines with 2 protons from the surrounding medium to produce NADPH.
Summary
- Photophosphorylation uses the radiant energy of the sun to drive the synthesis of ATP.
- This is a process seen only in cells capable of photosynthesis.
- Substrate-level phosphorylation is the production of ATP from ADP by a direct transfer of a high-energy phosphate group from a phosphorylated intermediate metabolic compound in an exergonic catabolic pathway.
- Oxidative phosphorylation is the production of ATP using energy derived from the transfer of electrons in an electron transport system and occurs by chemiosmosis.
- An atom of hydrogen contains only one proton (H+) and one electron.
- Electrons have stored energy, or potential energy, ready to do work. When an atom or molecule loses that electron (becomes oxidized) that energy is released and able to do cellular work.
- Oxidation-reduction reactions are coupled chemical reactions in which one atom or molecule loses one or more electrons (oxidation) while another atom or molecule gains those electrons (reduction).
- An oxidation reaction during which both a proton and an electron are lost is called dehydrogenation.
- A reduction reaction during which both a proton and an electron are gained is called hydrogenation.
- Cells use specific molecules such as NAD+, FAD, and NADP+ to carry the electrons that are removed during the oxidation of an energy source. These molecules are called electron carriers and they alternately become oxidized and reduced during electron and proton transfer.
- Coupled oxidation-reduction reactions and electron carriers are often part of what is called an electron transport chain.
- The chemiosmotic theory explains the functioning of electron transport chains. According to this theory, the transfer of electrons down an electron transport system through a series of oxidation-reduction reactions releases energy. This energy allows certain carriers in the chain to transport hydrogen ions (H+ or protons) across a membrane.
- In prokaryotic cells, the protons are transported from the cytoplasm of the bacterium across the cytoplasmic membrane to the periplasmic space located between the cytoplasmic membrane and the cell wall; in eukaryotic cells, protons are transported from the matrix of the mitochondria across the inner mitochondrial membrane to the intermembrane space located between the inner and outer mitochondrial membranes; in plant cells and the cells of algae, protons are transported from the stroma of the chloroplast across the thylakoid membrane into the interior space of the thylakoid.
- As the hydrogen ions accumulate on one side of a membrane, the concentration of hydrogen ions creates an electrochemical gradient or potential difference (voltage) across the membrane called proton motive force (PMF).
- This proton motive force provides the energy necessary for enzymes called ATP synthases to catalyze the synthesis of ATP from ADP and phosphate.
Questions
Study the material in this section and then write out the answers to these question. Do not just click on the answers and write them out. This will not test your understanding of this tutorial.
- Define photophosphorylation. (ans)
- Briefly describe the process of substrate-level phosphorylation. (ans)
- Briefly describe the process of oxidative phosphorylation. (ans)
- Another name for a hydrogen ion (H+) is: (ans)
- An atom or molecule gains an electron. This best describes:
- oxidation (ans)
- reduction (ans)
- dehydrogenation (ans)
- hydrogenation (ans)
- When a molecule gains electrons or both protons and electrons, we say it becomes:
- oxidized (ans)
- reduced (ans)
- Cells use specific molecules to carry the electrons that are removed during the oxidation of an energy source. These molecules are called electron carriers and they alternately become oxidized and reduced during electron and proton transfer. Name three freely diffusible coenzymes and give both their oxidized and reduced state. (ans)
- A coenzyme that often works in conjunction with an enzyme called a dehydrogenase. The enzyme removes two hydrogen atoms (2H+ and 2e-) from its substrate. Both electrons but only one proton are accepted to produce its reduced form that is used to generate proton motive force for driving the synthesis of ATP. This best describes:
- NAD+(ans)
- FAD (ans)
- NADP+ (ans)
- NADH + H+ is the ________________ form of NAD+. (ans)
- Describe an electron transport chain. (ans)
- Based on the chemiosmotic theory, briefly describe proton motive force and how it develops within a cell. (ans)
- Based on the chemiosmotic theory, briefly describe how proton motive force leads to the generation of ATP. (ans)


