1.2: Elements and Atoms
- Page ID
- 3742
\( \newcommand{\vecs}[1]{\overset { \scriptstyle \rightharpoonup} {\mathbf{#1}} } \)
\( \newcommand{\vecd}[1]{\overset{-\!-\!\rightharpoonup}{\vphantom{a}\smash {#1}}} \)
\( \newcommand{\dsum}{\displaystyle\sum\limits} \)
\( \newcommand{\dint}{\displaystyle\int\limits} \)
\( \newcommand{\dlim}{\displaystyle\lim\limits} \)
\( \newcommand{\id}{\mathrm{id}}\) \( \newcommand{\Span}{\mathrm{span}}\)
( \newcommand{\kernel}{\mathrm{null}\,}\) \( \newcommand{\range}{\mathrm{range}\,}\)
\( \newcommand{\RealPart}{\mathrm{Re}}\) \( \newcommand{\ImaginaryPart}{\mathrm{Im}}\)
\( \newcommand{\Argument}{\mathrm{Arg}}\) \( \newcommand{\norm}[1]{\| #1 \|}\)
\( \newcommand{\inner}[2]{\langle #1, #2 \rangle}\)
\( \newcommand{\Span}{\mathrm{span}}\)
\( \newcommand{\id}{\mathrm{id}}\)
\( \newcommand{\Span}{\mathrm{span}}\)
\( \newcommand{\kernel}{\mathrm{null}\,}\)
\( \newcommand{\range}{\mathrm{range}\,}\)
\( \newcommand{\RealPart}{\mathrm{Re}}\)
\( \newcommand{\ImaginaryPart}{\mathrm{Im}}\)
\( \newcommand{\Argument}{\mathrm{Arg}}\)
\( \newcommand{\norm}[1]{\| #1 \|}\)
\( \newcommand{\inner}[2]{\langle #1, #2 \rangle}\)
\( \newcommand{\Span}{\mathrm{span}}\) \( \newcommand{\AA}{\unicode[.8,0]{x212B}}\)
\( \newcommand{\vectorA}[1]{\vec{#1}} % arrow\)
\( \newcommand{\vectorAt}[1]{\vec{\text{#1}}} % arrow\)
\( \newcommand{\vectorB}[1]{\overset { \scriptstyle \rightharpoonup} {\mathbf{#1}} } \)
\( \newcommand{\vectorC}[1]{\textbf{#1}} \)
\( \newcommand{\vectorD}[1]{\overrightarrow{#1}} \)
\( \newcommand{\vectorDt}[1]{\overrightarrow{\text{#1}}} \)
\( \newcommand{\vectE}[1]{\overset{-\!-\!\rightharpoonup}{\vphantom{a}\smash{\mathbf {#1}}}} \)
\( \newcommand{\vecs}[1]{\overset { \scriptstyle \rightharpoonup} {\mathbf{#1}} } \)
\(\newcommand{\longvect}{\overrightarrow}\)
\( \newcommand{\vecd}[1]{\overset{-\!-\!\rightharpoonup}{\vphantom{a}\smash {#1}}} \)
\(\newcommand{\avec}{\mathbf a}\) \(\newcommand{\bvec}{\mathbf b}\) \(\newcommand{\cvec}{\mathbf c}\) \(\newcommand{\dvec}{\mathbf d}\) \(\newcommand{\dtil}{\widetilde{\mathbf d}}\) \(\newcommand{\evec}{\mathbf e}\) \(\newcommand{\fvec}{\mathbf f}\) \(\newcommand{\nvec}{\mathbf n}\) \(\newcommand{\pvec}{\mathbf p}\) \(\newcommand{\qvec}{\mathbf q}\) \(\newcommand{\svec}{\mathbf s}\) \(\newcommand{\tvec}{\mathbf t}\) \(\newcommand{\uvec}{\mathbf u}\) \(\newcommand{\vvec}{\mathbf v}\) \(\newcommand{\wvec}{\mathbf w}\) \(\newcommand{\xvec}{\mathbf x}\) \(\newcommand{\yvec}{\mathbf y}\) \(\newcommand{\zvec}{\mathbf z}\) \(\newcommand{\rvec}{\mathbf r}\) \(\newcommand{\mvec}{\mathbf m}\) \(\newcommand{\zerovec}{\mathbf 0}\) \(\newcommand{\onevec}{\mathbf 1}\) \(\newcommand{\real}{\mathbb R}\) \(\newcommand{\twovec}[2]{\left[\begin{array}{r}#1 \\ #2 \end{array}\right]}\) \(\newcommand{\ctwovec}[2]{\left[\begin{array}{c}#1 \\ #2 \end{array}\right]}\) \(\newcommand{\threevec}[3]{\left[\begin{array}{r}#1 \\ #2 \\ #3 \end{array}\right]}\) \(\newcommand{\cthreevec}[3]{\left[\begin{array}{c}#1 \\ #2 \\ #3 \end{array}\right]}\) \(\newcommand{\fourvec}[4]{\left[\begin{array}{r}#1 \\ #2 \\ #3 \\ #4 \end{array}\right]}\) \(\newcommand{\cfourvec}[4]{\left[\begin{array}{c}#1 \\ #2 \\ #3 \\ #4 \end{array}\right]}\) \(\newcommand{\fivevec}[5]{\left[\begin{array}{r}#1 \\ #2 \\ #3 \\ #4 \\ #5 \\ \end{array}\right]}\) \(\newcommand{\cfivevec}[5]{\left[\begin{array}{c}#1 \\ #2 \\ #3 \\ #4 \\ #5 \\ \end{array}\right]}\) \(\newcommand{\mattwo}[4]{\left[\begin{array}{rr}#1 \amp #2 \\ #3 \amp #4 \\ \end{array}\right]}\) \(\newcommand{\laspan}[1]{\text{Span}\{#1\}}\) \(\newcommand{\bcal}{\cal B}\) \(\newcommand{\ccal}{\cal C}\) \(\newcommand{\scal}{\cal S}\) \(\newcommand{\wcal}{\cal W}\) \(\newcommand{\ecal}{\cal E}\) \(\newcommand{\coords}[2]{\left\{#1\right\}_{#2}}\) \(\newcommand{\gray}[1]{\color{gray}{#1}}\) \(\newcommand{\lgray}[1]{\color{lightgray}{#1}}\) \(\newcommand{\rank}{\operatorname{rank}}\) \(\newcommand{\row}{\text{Row}}\) \(\newcommand{\col}{\text{Col}}\) \(\renewcommand{\row}{\text{Row}}\) \(\newcommand{\nul}{\text{Nul}}\) \(\newcommand{\var}{\text{Var}}\) \(\newcommand{\corr}{\text{corr}}\) \(\newcommand{\len}[1]{\left|#1\right|}\) \(\newcommand{\bbar}{\overline{\bvec}}\) \(\newcommand{\bhat}{\widehat{\bvec}}\) \(\newcommand{\bperp}{\bvec^\perp}\) \(\newcommand{\xhat}{\widehat{\xvec}}\) \(\newcommand{\vhat}{\widehat{\vvec}}\) \(\newcommand{\uhat}{\widehat{\uvec}}\) \(\newcommand{\what}{\widehat{\wvec}}\) \(\newcommand{\Sighat}{\widehat{\Sigma}}\) \(\newcommand{\lt}{<}\) \(\newcommand{\gt}{>}\) \(\newcommand{\amp}{&}\) \(\definecolor{fillinmathshade}{gray}{0.9}\)Elements
Elements consist of only one kind of atom and cannot be decomposed into simpler substances. Our planet is made up of some 90 elements. (Tiny amounts — sometimes only a few atoms — of additional elements have been made in nuclear physics laboratories, but they play no role in our story). Of these 90, only 25 or so are used to build living things. The table shows the 11 most prevalent elements in the lithosphere (the earth's crust) and in the human body.
Living matter
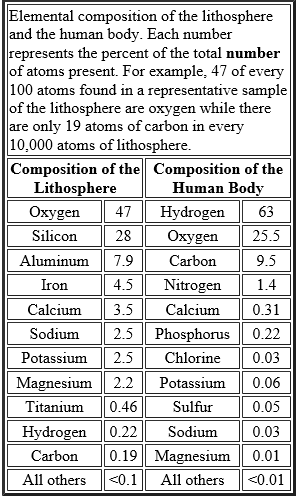 uses only a fraction of the elements available to it
uses only a fraction of the elements available to it- but, as the table shows, the relative proportions of those it does acquire from its surroundings are quite different from the proportions in the environment
So,
- the composition of living things is not simply a reflection of the elements available to them
- For example, hydrogen, carbon, and nitrogen together represent less than 1% of the atoms found in the earth's crust but some 74% of the atoms in living matter.
- one of the properties of life is to take up certain elements that are scarce in the nonliving world and concentrate them within living cells.
Some sea animals accumulate elements like vanadium and iodine within their cells to concentrations a thousand or more times as great as in the surrounding sea water. It has even been proposed that uranium be "mined" from the sea by extracting it from certain algae that can take up uranium from sea water and concentrate it within their cells.
There is still some uncertainty about the exact number of elements required by living things. Some elements, e.g., aluminum, are found in tiny amounts in living tissue, but whether they are playing an essential role or are simply an accidental acquisition (aluminum probably is) is sometimes difficult to determine.
Atoms
Each element is made up of one kind of atom. We can define an atom as the smallest part of an element that can enter into combination with other elements.
Structure of the atom
Each atom consists of a small, dense, positively-charged nucleus surrounded by much lighter, negatively-charged electrons. The nucleus of the simplest atom, the hydrogen atom (H), consists of a single positively-charged proton. Because of its single proton, the atom of hydrogen is assigned an atomic number of 1 and a single electron. The charge of the electron is the same magnitude as that of the proton, so the atom as a whole is electrically neutral. Its proton accounts for almost all the weight of the atom.
The nucleus of the atom of the element helium (He) has two protons (hence helium has an atomic number of 2) and two neutrons. Neutrons have the same weight as protons but no electrical charge. The helium atom has two electrons so that, once again, the atom as a whole is neutral.
The structure of each of the other kinds of atoms follows the same plan. From Lithium (At. No. = 3) to uranium (At. No. = 92), the atoms of each element can be listed in order of increasing atomic number. There are no gaps in the list. Each element has a unique atomic number and its atoms have one more proton and one more electron than the atoms of the element that precedes it in the list.
Electrons
| Atomic Number | Element | Energy Levels or "shells" | ||||
|---|---|---|---|---|---|---|
| K | L | M | N | O | ||
| 1 | Hydrogen (H) | 1 | ||||
| 2 | Helium (He) | 2 | ||||
| 3 | Lithium (Li) | 2 | 1 | |||
| 4 | Beryllium (Be) | 2 | 2 | |||
| 5 | Boron (B) | 2 | 3 | |||
| 6 | Carbon (C) | 2 | 4 | |||
| 7 | Nitrogen (N) | 2 | 5 | |||
| 8 | Oxygen (O) | 2 | 6 | |||
| 9 | Fluorine (F) | 2 | 7 | |||
| 10 | Neon (Ne) | 2 | 8 | |||
| 11 | Sodium (Na) | 2 | 8 | 1 | ||
| 12 | Magnesium (Mg) | 2 | 8 | 2 | ||
| 13 | Aluminum (Al) | 2 | 8 | 3 | ||
| 14 | Silicon (Si) | 2 | 8 | 4 | ||
| 15 | Phosphorus (P) | 2 | 8 | 5 | ||
| 16 | Sulfur (S) | 2 | 8 | 6 | ||
| 17 | Chlorine (Cl) | 2 | 8 | 7 | ||
| 18 | Argon (Ar) | 2 | 8 | 8 | ||
| 19 | Potassium (K) | 2 | 8 | 8 | 1 | |
| 20 | Calcium (Ca) | 2 | 8 | 8 | 2 | |
| 21 | Scandium (Sc) | 2 | 8 | 9 | 2 | |
| 22 | Titanium (Ti) | 2 | 8 | 10 | 2 | |
| 23 | Vanadium (V) | 2 | 8 | 11 | 2 | |
| 24 | Chromium (Cr) | 2 | 8 | 13 | 1 | |
| 25 | Manganese (Mn) | 2 | 8 | 13 | 2 | |
| 26 | Iron (Fe) | 2 | 8 | 14 | 2 | |
| 27 | Cobalt (Co) | 2 | 8 | 15 | 2 | |
| 28 | Nickel (Ni) | 2 | 8 | 16 | 2 | |
| 29 | Copper (Cu) | 2 | 8 | 18 | 1 | |
| 30 | Zinc (Zn) | 2 | 8 | 18 | 2 | |
| 31 | Gallium (Ga) | 2 | 8 | 18 | 3 | |
| 32 | Germanium (Ge) | 2 | 8 | 18 | 4 | |
| 33 | Arsenic (As) | 2 | 8 | 18 | 5 | |
| 34 | Selenium (Se) | 2 | 8 | 18 | 6 | |
| 35 | Bromine (Br) | 2 | 8 | 18 | 7 | |
| 36 | Krypton (Kr) | 2 | 8 | 18 | 8 | |
| 42 | Molybdenum (Mo) | 2 | 8 | 18 | 13 | 1 |
| 48 | Cadmium (Cd) | 2 | 8 | 18 | 18 | 2 |
| 50 | Tin (Sn) | 2 | 8 | 18 | 18 | 4 |
| 53 | Iodine (I) | 2 | 8 | 18 | 18 | 7 |
Electrons are confined to relatively discrete regions around the nucleus. The two electrons of helium, for example, are confined to a spherical zone surrounding the nucleus called the K shell or K energy level.
Lithium (At. No. = 3) has three electrons, two in the K shell and one located farther from the nucleus in the L shell. Being farther away from the opposite (+) charges of the nucleus, this third electron is held less tightly.
Each of the following elements, in order of increasing atomic number, adds one more electron to the L shell until we reach neon (At. No. = 10) which has eight electrons in the L shell.
Sodium places its eleventh electron in a still higher energy level, the M shell.
From sodium to argon, this shell is gradually filled with electrons until, once again, a maximum of eight is reached.
Note that after the K shell with its maximum of two electrons, the maximum number of electrons in any other outermost shell is eight.
As we shall see, the chemical properties of each element are strongly influenced by the number of electrons in its outermost energy level (shell).
This table shows the electronic structure of the atoms of elements 1 – 36 with those that have been demonstrated to be used by living things shown in red. Four elements of still higher atomic numbers that have been shown to be used by living things are also included.
The electronic structure of an atom plays the major role in its chemistry.
The pattern of electrons in an atom — especially those in the outermost shell — determines
- the valence of the atom; that is, the ratios with which it interacts with other atoms, and to a large degree,
- the electronegativity of the atom; that is, the strength with which it attracts other electrons.
Elements with the same number of electrons in their outermost shell show similar chemical properties.
Example 1: Fluorine, chlorine, bromine, and iodine each have 7 electrons in their outermost shell. These so-called halogens are also quite similar in their chemical behavior. When dissolved in water, for example, they all produce germicidal solutions.
Example 2: Those elements with 1, 2, or 3 electrons in their outermost shell are the metals.
Example 3: Those elements with 4, 5, 6, or 7 in their outermost shell are the nonmetals.
Example 4: Helium (with its 2), neon, argon, and krypton (each with 8) have "filled" their outermost shells. They are the so-called inert or "noble" gases. They have no chemistry at all. Under normal conditions they do not interact with other atoms. So, it is the number and arrangement of the electrons in the atoms of an element that establish the chemical behavior of that element.
This is how it works:
The atoms of an element interact with other atoms in such ways and ratios that they can "fill" their outermost shell with 8 electrons (2 for hydrogen). They may do this by
- acquiring more electrons from another atom
- losing electrons to another atom
- sharing electrons with another atom
The number of electrons that an atom must acquire, or lose, or share to reach a stable configuration of 8 (2 for hydrogen) is called its valence.
Hydrogen, lithium, sodium, and potassium atoms all have a single electron in their outermost shell. Fluorine, chlorine, bromine, and iodine atoms all have 7. Any atom of the first group will interact with a single atom of any of the second group forming, HCl, NaCl, KI, etc. The result of all of these interactions is a pair of atoms each with an outermost shell like that of one of the inert gases: 2 for hydrogen, 8 for the others.
The elements with 2 electrons in their outermost shell interact with chlorine and the other halogens to form, e.g., BeCl2, MgCl2, CaCl2. Again, the result is a pair of atoms each with a stable octet of electrons in its outermost shell.
The elements with 3 electrons in their outermost shell will interact with chlorine in a ratio of 1:3, forming BCl3, AlCl3.
Carbon atoms, with their 4 electrons in the L shell interact with chlorine to form CCl4.
Nitrogen, with its 5 outermost electrons, interacts with hydrogen atoms in a ratio of 1:3, forming ammonia (NH3).
Oxygen and sulfur, with their 6 outermost electrons react with hydrogen to form water (H2O) and hydrogen sulfide (H2S).
What determines whether a pair of atoms swap or share electrons?
The answer is their relative electronegativities. If two atoms differ greatly in their affinity for electrons; that is, in their electronegativity, then the strongly electronegative atom will take the electron away from the weakly electronegative one.
Example: Na (weakly electronegative) gives up its single electron to an atom of chlorine (strongly electronegative) to form NaCl. The sodium atom now has only 10 electrons but still 11 protons so there is a net positive charge of one on the atom. Similarly, chlorine now has one more electron than proton so its now has a net negative charge of 1. Electrically charged atoms are called ions. The mutual attraction of opposite electrical charges holds the ions together by ionic bonds.
Example: Carbon and hydrogen are both only weakly electronegative so neither can remove electrons from the other. Instead they achieve a stable configuration by sharing their outermost electrons forming covalent bonds of CH4.
Isotopes
The number of protons in the nucleus of its atoms, which is its atomic number, defines each element. However, the nuclei of a given element may have varying numbers of neutrons. Because neutrons have weight (about the same as that of protons), such atoms differ in the atomic weight.
Atoms of the same element that differ in their atomic weight are called isotopes.
Atomic weights are expressed in terms of a standard atom: the isotope of carbon that has 6 protons and 6 neutrons in its nucleus. This atom is designated carbon-12 or 12C. It is arbitrarily assigned an atomic weight of 12 daltons (named after John Dalton, the pioneer in the study of atomic weights). Thus a dalton is 1/12 the weight of an atom of 12C. Both protons and neutrons have weights very close to 1 dalton each. Carbon-12 is the most common isotope of carbon. Carbon-13 (13C) with 6 protons and 7 neutrons, and carbon-14 (14C) with 6 protons and 8 neutrons are found in much smaller quantities.
Isotopes as "tracers"
One can prepare, for example, a carbon compound used by living things that has many of its normal 12C atoms replaced by 14C atoms. Carbon-14 happens to be radioactive. By tracing the fate of radioactivity within the organism, one can learn the normal pathway of this carbon compound in that organism. Thus 14C serves as an isotopic "label" or "tracer".
The basis of this technique is that the weight of the nucleus of an atom has little or no effect on the chemical properties of that atom. The chemistry of an element and the atoms of which it is made — whatever their atomic weight — is a function of the atomic number of that element. As long as the atom had 6 protons, it is an atom of carbon irrespective of the number of neutrons. Thus while 6 protons and 8 neutrons produce an isotope of carbon, 14C, 7 protons and 7 neutrons produce a totally-different element, nitrogen-14.
Contributors and Attributions
John W. Kimball. This content is distributed under a Creative Commons Attribution 3.0 Unported (CC BY 3.0) license and made possible by funding from The Saylor Foundation.


