Investigation: Gel Electrophoresis
- Page ID
- 21433
\( \newcommand{\vecs}[1]{\overset { \scriptstyle \rightharpoonup} {\mathbf{#1}} } \)
\( \newcommand{\vecd}[1]{\overset{-\!-\!\rightharpoonup}{\vphantom{a}\smash {#1}}} \)
\( \newcommand{\dsum}{\displaystyle\sum\limits} \)
\( \newcommand{\dint}{\displaystyle\int\limits} \)
\( \newcommand{\dlim}{\displaystyle\lim\limits} \)
\( \newcommand{\id}{\mathrm{id}}\) \( \newcommand{\Span}{\mathrm{span}}\)
( \newcommand{\kernel}{\mathrm{null}\,}\) \( \newcommand{\range}{\mathrm{range}\,}\)
\( \newcommand{\RealPart}{\mathrm{Re}}\) \( \newcommand{\ImaginaryPart}{\mathrm{Im}}\)
\( \newcommand{\Argument}{\mathrm{Arg}}\) \( \newcommand{\norm}[1]{\| #1 \|}\)
\( \newcommand{\inner}[2]{\langle #1, #2 \rangle}\)
\( \newcommand{\Span}{\mathrm{span}}\)
\( \newcommand{\id}{\mathrm{id}}\)
\( \newcommand{\Span}{\mathrm{span}}\)
\( \newcommand{\kernel}{\mathrm{null}\,}\)
\( \newcommand{\range}{\mathrm{range}\,}\)
\( \newcommand{\RealPart}{\mathrm{Re}}\)
\( \newcommand{\ImaginaryPart}{\mathrm{Im}}\)
\( \newcommand{\Argument}{\mathrm{Arg}}\)
\( \newcommand{\norm}[1]{\| #1 \|}\)
\( \newcommand{\inner}[2]{\langle #1, #2 \rangle}\)
\( \newcommand{\Span}{\mathrm{span}}\) \( \newcommand{\AA}{\unicode[.8,0]{x212B}}\)
\( \newcommand{\vectorA}[1]{\vec{#1}} % arrow\)
\( \newcommand{\vectorAt}[1]{\vec{\text{#1}}} % arrow\)
\( \newcommand{\vectorB}[1]{\overset { \scriptstyle \rightharpoonup} {\mathbf{#1}} } \)
\( \newcommand{\vectorC}[1]{\textbf{#1}} \)
\( \newcommand{\vectorD}[1]{\overrightarrow{#1}} \)
\( \newcommand{\vectorDt}[1]{\overrightarrow{\text{#1}}} \)
\( \newcommand{\vectE}[1]{\overset{-\!-\!\rightharpoonup}{\vphantom{a}\smash{\mathbf {#1}}}} \)
\( \newcommand{\vecs}[1]{\overset { \scriptstyle \rightharpoonup} {\mathbf{#1}} } \)
\(\newcommand{\longvect}{\overrightarrow}\)
\( \newcommand{\vecd}[1]{\overset{-\!-\!\rightharpoonup}{\vphantom{a}\smash {#1}}} \)
\(\newcommand{\avec}{\mathbf a}\) \(\newcommand{\bvec}{\mathbf b}\) \(\newcommand{\cvec}{\mathbf c}\) \(\newcommand{\dvec}{\mathbf d}\) \(\newcommand{\dtil}{\widetilde{\mathbf d}}\) \(\newcommand{\evec}{\mathbf e}\) \(\newcommand{\fvec}{\mathbf f}\) \(\newcommand{\nvec}{\mathbf n}\) \(\newcommand{\pvec}{\mathbf p}\) \(\newcommand{\qvec}{\mathbf q}\) \(\newcommand{\svec}{\mathbf s}\) \(\newcommand{\tvec}{\mathbf t}\) \(\newcommand{\uvec}{\mathbf u}\) \(\newcommand{\vvec}{\mathbf v}\) \(\newcommand{\wvec}{\mathbf w}\) \(\newcommand{\xvec}{\mathbf x}\) \(\newcommand{\yvec}{\mathbf y}\) \(\newcommand{\zvec}{\mathbf z}\) \(\newcommand{\rvec}{\mathbf r}\) \(\newcommand{\mvec}{\mathbf m}\) \(\newcommand{\zerovec}{\mathbf 0}\) \(\newcommand{\onevec}{\mathbf 1}\) \(\newcommand{\real}{\mathbb R}\) \(\newcommand{\twovec}[2]{\left[\begin{array}{r}#1 \\ #2 \end{array}\right]}\) \(\newcommand{\ctwovec}[2]{\left[\begin{array}{c}#1 \\ #2 \end{array}\right]}\) \(\newcommand{\threevec}[3]{\left[\begin{array}{r}#1 \\ #2 \\ #3 \end{array}\right]}\) \(\newcommand{\cthreevec}[3]{\left[\begin{array}{c}#1 \\ #2 \\ #3 \end{array}\right]}\) \(\newcommand{\fourvec}[4]{\left[\begin{array}{r}#1 \\ #2 \\ #3 \\ #4 \end{array}\right]}\) \(\newcommand{\cfourvec}[4]{\left[\begin{array}{c}#1 \\ #2 \\ #3 \\ #4 \end{array}\right]}\) \(\newcommand{\fivevec}[5]{\left[\begin{array}{r}#1 \\ #2 \\ #3 \\ #4 \\ #5 \\ \end{array}\right]}\) \(\newcommand{\cfivevec}[5]{\left[\begin{array}{c}#1 \\ #2 \\ #3 \\ #4 \\ #5 \\ \end{array}\right]}\) \(\newcommand{\mattwo}[4]{\left[\begin{array}{rr}#1 \amp #2 \\ #3 \amp #4 \\ \end{array}\right]}\) \(\newcommand{\laspan}[1]{\text{Span}\{#1\}}\) \(\newcommand{\bcal}{\cal B}\) \(\newcommand{\ccal}{\cal C}\) \(\newcommand{\scal}{\cal S}\) \(\newcommand{\wcal}{\cal W}\) \(\newcommand{\ecal}{\cal E}\) \(\newcommand{\coords}[2]{\left\{#1\right\}_{#2}}\) \(\newcommand{\gray}[1]{\color{gray}{#1}}\) \(\newcommand{\lgray}[1]{\color{lightgray}{#1}}\) \(\newcommand{\rank}{\operatorname{rank}}\) \(\newcommand{\row}{\text{Row}}\) \(\newcommand{\col}{\text{Col}}\) \(\renewcommand{\row}{\text{Row}}\) \(\newcommand{\nul}{\text{Nul}}\) \(\newcommand{\var}{\text{Var}}\) \(\newcommand{\corr}{\text{corr}}\) \(\newcommand{\len}[1]{\left|#1\right|}\) \(\newcommand{\bbar}{\overline{\bvec}}\) \(\newcommand{\bhat}{\widehat{\bvec}}\) \(\newcommand{\bperp}{\bvec^\perp}\) \(\newcommand{\xhat}{\widehat{\xvec}}\) \(\newcommand{\vhat}{\widehat{\vvec}}\) \(\newcommand{\uhat}{\widehat{\uvec}}\) \(\newcommand{\what}{\widehat{\wvec}}\) \(\newcommand{\Sighat}{\widehat{\Sigma}}\) \(\newcommand{\lt}{<}\) \(\newcommand{\gt}{>}\) \(\newcommand{\amp}{&}\) \(\definecolor{fillinmathshade}{gray}{0.9}\)Detecting the Duchenne Muscular Dystrophy (DMD) Mutation
*adapted from Washington University's Modern Genetics curriculum
Introduction
Mary and John Smith have three children: Daniel, age 5; Alice, age 4; and Michael, age 1. Mary is two months pregnant. Recently Mary and John noticed that Daniel was having trouble climbing the stairs. He also complained several times that he was really tired after playing tag with his sister. Daniel’s doctor examined the child and suggested he be tested for Duchenne Muscular Dystrophy (DMD), as he was displaying many of the classic symptoms of the disease. Mary was terrified, she remembered that her uncle died of the disease when he was very young. Could this run in her family?
The doctor suggested that perhaps all of their family should be tested, even the fetus that Mary was carrying. He referred them to a genetic counselor who explained how the disease is inherited. They learned that DMD is a sex-linked genetic disease, which means that it results from damage to a gene on the X chromosome. That is why almost everyone with DMD is male. A girl may inherit an X chromosome with a defective copy of the DMD gene from her mother and like her mother; she will be a carrier for DMD. But the girl most likely will be protected from developing DMD by the normal X chromosome she gets from her father. In contrast, a boy does not get an X chromosome from his father, and if the X chromosome he gets from his mother carries a defective copy of the DMD gene, he will develop DMD.
Usually boys with DMD are healthy until the age of 4 or 5, at which time their muscles start to weaken. The doctor told the Smiths that Daniel would probably need a wheelchair in a few years, and that he would probably die before the age of 21. Although scientists are working to find a cure for this disease, there is no effective treatment for DMD now.
The genetics specialist took blood samples from each member of the family, including the fetus. The Smith's blood samples would be separated and the DNA isolated from white blood cells. But the DMD gene that the Smith family wished you to analyze was only one of many thousand genes in each of the resulting DNA samples. So, to get enough of this particular gene to study, analysts use a procedure called PCR (short for polymerase chain reaction) to amplify (make many copies of) the tiny section of DNA that contained the DMD gene.
Then the samples are analyzed to determine which ones contain only copies of the wild-type copies of DMD allele (normal phenotype), which ones contain only copies of the mutant DMD allele, and which ones contain copies of both DMD alleles.
Mutations that cause this disease involve a deletion of part of the DNA from the DMD gene, so that the mutant alleles will be shorter than the wild-type alleles. As illustrated in the diagram, this implies that Mary, who is likely a carrier for DMD, has one normal and one short (mutant) allele at the DMD locus. Since John, being a male, has only one X chromosome and does not have DMD, his DMD allele must be of normal length. If Daniel has the DMD disorder, he must have received the X chromosome with the defective DMD gene from his mother.
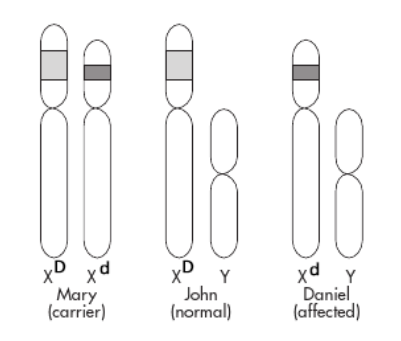
Now the question is, Which DMD alleles do the family members have? That’s what you will try to determine.
DNA molecules that differ in length can be separated and analyzed by a process known as gel electrophoresis. This method uses an electric current to push DNA molecules through a gel-like substance called agarose. Small DNA molecules move through the gel faster than large ones, and will move farther in the gel than larger segments.
You will use gel electrophoresis to simulate the procedure that the genetic counseling lab would use to determine the genotypes of family members with respect to a gene of interest – the DMD gene. Your job will be to determine which allele(s) each member of the Smith family possesses, by inspecting the gel at the end of the electrophoresis.
PRE LAB QUESTIONS
- What are the symptoms of Duchenne Muscular Dystrophy? Why is it significant to the doctor that Mary’s uncle had this disease?
- Illustrate using a Punnett square the method of inheritance showing Mary and John, and the four genotypes possible in their offspring, assuming that Mary is a carrier for the disease.
- Can a boy be a carrier for DMD without having the disease? Why or why not?
- What is the purpose of the PCR procedure?
- Describe how the wild-type allele differs from the mutant DMD allele, with respect to the chromosome.
- What is the relationship between the size of the DNA fragments and their appearance during gel electrophoresis?
- What is the relationship between genes and chromosomes with respect to Duchenne Muscular Dystrophy.
Day 1 – Setting the Gel
MATERIALS For each group of students (group size to be determined by equipment availability):
- Prepared 0.8% agarose solution (1.6 g of agarose to 200 ml of water, heat in 500 ml flask, caution when boiling).
- 1 gel-casting tray plus masking tape, 1 or 2 gel-casting combs and ~50 ml of 0.8% agarose in water (prepared in step #1)
- A gel electrophoresis chamber and power supply
- 1 10-20 µl micropipettor | 6 dye samples and 6 pipette tips
PROCEDURE - Creating the Gel Casts
- Seal the ends of the gel-casting tray with masking tape. Insert the comb near one end of the casting tray. Place the tray on a level surface where it will not be disturbed while the gel solidifies.
- Obtain a flask with melted agarose solution. It should be warm and liquified but not too hot to handle.
- Carefully pour about 50 ml. The gel should cover only about one-third of the height of the comb teeth.
- Do not move or jar the casting tray while the agarose solidifies. As it becomes solid (10-15 minutes), the agarose will change from clear to cloudy. When the agarose is solid, cover it with plastic wrap, leaving the comb and the tape intact. The casting tray will be refrigerated and will be used the next day.
*Read the directions for day 2*
Day 2 - Electrophoresis
-
Take the gel mold to the area where the gel electrophoresis chamber has been set up next to the power supply. Place the gel in the electrophoresis chamber so that the end containing the comb is toward the black electrode.
-
Carefully fill the chamber with tap water to a level that just covers the entire surface of the gel, do not disturb the gel as you fill the chamber.
-
Your instructor will demonstrate how to use the micropipette to load the samples into the gel. Be sure to keep track of which sample has been loaded into each well.
-
Turn the power supply on to run electricity through your chamber. This will cause your samples to move.
CAUTION: Electric shock hazard! Do not put fingers or other objects into the box while power supply is on.
Run the electrophoresis for approximately 15 minutes then turn off the power supply and disconnect the leads. You can remove the gel molds to more easily view the bands created by the samples moving.
Draw your mold showing how the bands appear. Be sure to label each well with the family member’s name.
Test Results and Diagnosis
1. Indicate the genotypes and phenotypes for each of the family members
Mother Mary XDXd Father John XDY
Daniel __________________________________ Alice __________________________________
Michael _________________________________ Fetus __________________________________
2. Summarize the results of the test, which family members are affected by the DMD allele. Write this as if your were delivering your report to the family.
3. How is this relationship between genes and chromosomes applied to the technology of gel electrophoresis? In your answer, consider why the test you performed works to determine individuals with the gene. Be thorough in your explanation.
ADVANCE PREPARATION OF CHEMICALS
Materials Can be Obtained from:
Electrophoresis Power Supply
Gel Electrophoresis Chamber
Extra Comb-
Extra Gel Casting Tray
Agarose (powder)
Pipette Tips
Microcentrifuge tubes
Sigma Chemical Company
Bromphenol Blue- catalog # B0126
Xylene Cyanole- catalog # X4126
You can also order "GEL LOADING Solution" and Bromphenol Blue from Flinn Scientific
1. Prepare 0.8% agarose solution, 50 ml per group. The following recipe is for 200 ml(4 groups):
- To 200 ml water in a 500 ml flask add 1.6 grams agarose.
- Cap flask with foil and heat carefully on a hot plate, or cap with plastic wrap and heat carefully in a microwave oven for 3-5 minutes.
- Safety Note: Agarose solution can superheat and either boil over during heating or erupt violently when the flask is touched. Handle hot agarose very carefully, wearing safety goggles and heavy gloves.
- Swirl flask and make sure that all agarose has dissolved.
- If gels are to be poured right away, cool flask to about 60°C before pouring.
- If gels are to be poured later the same day, hold flask in a 60°C water bath until use.
- If gels are to be poured another day, store solution covered and refrigerated. (Keeps forseveral weeks.) Then, well in advance of scheduled use, reheat agarose carefully on a hot plate or in a microwave oven until it is completely melted. Then bring it to about 60°C before pouring gels. (cool enough to touch to inside of wrist) If you will be pouring the gels yourself, follow the instructions given on the student pages for this exercise. When the gels have solidified, wrap each one in plastic wrap to prevent it from drying out.
2. Prepare stock solutions of bromphenol blue and xylene cyanole. In this exercise we will simulate the DNA samples of the various Smith family members with a pair of dyes, bromphenol blue and xylene cyanole. Bromphenol blue (BB) travels faster in an agarose gel, so it will be used to represent the "defective allele" that is a partially deleted version of the dystrophin (DMD) gene. Xylene cyanole (XC) travels more slowly in the gel, and so it will be used to represent the "normal" dystrophin allele.
Prepare stock solutions as follows:
- Label two small beakers or flasks BB and XC. Add 10 ml of deionized water and 1 ml of glycerol to each. Swirl to mix. Safety Note: Certain dyes can be dangerous to your health when they are in the dry state but become harmless once they have been dissolved. So weigh out all dyes in a fume hood, and wear a dust mask, goggles, and gloves until they are in solution.
- Weigh out 0.025 grams (25 mg) of bromphenol blue and add it to the BB container.
- Weigh out 0.025 grams (25 mg) of xylene cyanole and add it to the XC container.
- Carefully stir or swirl both containers until the dyes are thoroughly dissolved.
3. Prepare simulated Smith family DNA samples from the stock dye solutions. Set up six microcentrifuge tubes, lettered A–F, for each group. Add dyes as shown in the table below. Then cap the tubes and place each set of six in a sandwich baggie or other small container to hand out to each lab group. As you begin the experiment, inform the students which family member is represented by which letter, but do not provide the other information in the table.
Tube Family member Genetic condition Dye sample used
A Mother Carrier 25 μl BB & 25 μl XC
B Father Normal 50 μl BB
C Daniel Affected 50 μl XC
D Alice Carrier 25 μl BB & 25 μl XC
E Michael Normal 50 μl BB
F Fetus Carrier 25 μl BB & 25 μl XC
Adapted from: Modern Genetics for All Students
TIP: Many colleges will loan you materials, such as electrophoresis chambers, power supplies, and micropipettes


