3.5: Procedure
- Page ID
- 123032
\( \newcommand{\vecs}[1]{\overset { \scriptstyle \rightharpoonup} {\mathbf{#1}} } \)
\( \newcommand{\vecd}[1]{\overset{-\!-\!\rightharpoonup}{\vphantom{a}\smash {#1}}} \)
\( \newcommand{\dsum}{\displaystyle\sum\limits} \)
\( \newcommand{\dint}{\displaystyle\int\limits} \)
\( \newcommand{\dlim}{\displaystyle\lim\limits} \)
\( \newcommand{\id}{\mathrm{id}}\) \( \newcommand{\Span}{\mathrm{span}}\)
( \newcommand{\kernel}{\mathrm{null}\,}\) \( \newcommand{\range}{\mathrm{range}\,}\)
\( \newcommand{\RealPart}{\mathrm{Re}}\) \( \newcommand{\ImaginaryPart}{\mathrm{Im}}\)
\( \newcommand{\Argument}{\mathrm{Arg}}\) \( \newcommand{\norm}[1]{\| #1 \|}\)
\( \newcommand{\inner}[2]{\langle #1, #2 \rangle}\)
\( \newcommand{\Span}{\mathrm{span}}\)
\( \newcommand{\id}{\mathrm{id}}\)
\( \newcommand{\Span}{\mathrm{span}}\)
\( \newcommand{\kernel}{\mathrm{null}\,}\)
\( \newcommand{\range}{\mathrm{range}\,}\)
\( \newcommand{\RealPart}{\mathrm{Re}}\)
\( \newcommand{\ImaginaryPart}{\mathrm{Im}}\)
\( \newcommand{\Argument}{\mathrm{Arg}}\)
\( \newcommand{\norm}[1]{\| #1 \|}\)
\( \newcommand{\inner}[2]{\langle #1, #2 \rangle}\)
\( \newcommand{\Span}{\mathrm{span}}\) \( \newcommand{\AA}{\unicode[.8,0]{x212B}}\)
\( \newcommand{\vectorA}[1]{\vec{#1}} % arrow\)
\( \newcommand{\vectorAt}[1]{\vec{\text{#1}}} % arrow\)
\( \newcommand{\vectorB}[1]{\overset { \scriptstyle \rightharpoonup} {\mathbf{#1}} } \)
\( \newcommand{\vectorC}[1]{\textbf{#1}} \)
\( \newcommand{\vectorD}[1]{\overrightarrow{#1}} \)
\( \newcommand{\vectorDt}[1]{\overrightarrow{\text{#1}}} \)
\( \newcommand{\vectE}[1]{\overset{-\!-\!\rightharpoonup}{\vphantom{a}\smash{\mathbf {#1}}}} \)
\( \newcommand{\vecs}[1]{\overset { \scriptstyle \rightharpoonup} {\mathbf{#1}} } \)
\(\newcommand{\longvect}{\overrightarrow}\)
\( \newcommand{\vecd}[1]{\overset{-\!-\!\rightharpoonup}{\vphantom{a}\smash {#1}}} \)
\(\newcommand{\avec}{\mathbf a}\) \(\newcommand{\bvec}{\mathbf b}\) \(\newcommand{\cvec}{\mathbf c}\) \(\newcommand{\dvec}{\mathbf d}\) \(\newcommand{\dtil}{\widetilde{\mathbf d}}\) \(\newcommand{\evec}{\mathbf e}\) \(\newcommand{\fvec}{\mathbf f}\) \(\newcommand{\nvec}{\mathbf n}\) \(\newcommand{\pvec}{\mathbf p}\) \(\newcommand{\qvec}{\mathbf q}\) \(\newcommand{\svec}{\mathbf s}\) \(\newcommand{\tvec}{\mathbf t}\) \(\newcommand{\uvec}{\mathbf u}\) \(\newcommand{\vvec}{\mathbf v}\) \(\newcommand{\wvec}{\mathbf w}\) \(\newcommand{\xvec}{\mathbf x}\) \(\newcommand{\yvec}{\mathbf y}\) \(\newcommand{\zvec}{\mathbf z}\) \(\newcommand{\rvec}{\mathbf r}\) \(\newcommand{\mvec}{\mathbf m}\) \(\newcommand{\zerovec}{\mathbf 0}\) \(\newcommand{\onevec}{\mathbf 1}\) \(\newcommand{\real}{\mathbb R}\) \(\newcommand{\twovec}[2]{\left[\begin{array}{r}#1 \\ #2 \end{array}\right]}\) \(\newcommand{\ctwovec}[2]{\left[\begin{array}{c}#1 \\ #2 \end{array}\right]}\) \(\newcommand{\threevec}[3]{\left[\begin{array}{r}#1 \\ #2 \\ #3 \end{array}\right]}\) \(\newcommand{\cthreevec}[3]{\left[\begin{array}{c}#1 \\ #2 \\ #3 \end{array}\right]}\) \(\newcommand{\fourvec}[4]{\left[\begin{array}{r}#1 \\ #2 \\ #3 \\ #4 \end{array}\right]}\) \(\newcommand{\cfourvec}[4]{\left[\begin{array}{c}#1 \\ #2 \\ #3 \\ #4 \end{array}\right]}\) \(\newcommand{\fivevec}[5]{\left[\begin{array}{r}#1 \\ #2 \\ #3 \\ #4 \\ #5 \\ \end{array}\right]}\) \(\newcommand{\cfivevec}[5]{\left[\begin{array}{c}#1 \\ #2 \\ #3 \\ #4 \\ #5 \\ \end{array}\right]}\) \(\newcommand{\mattwo}[4]{\left[\begin{array}{rr}#1 \amp #2 \\ #3 \amp #4 \\ \end{array}\right]}\) \(\newcommand{\laspan}[1]{\text{Span}\{#1\}}\) \(\newcommand{\bcal}{\cal B}\) \(\newcommand{\ccal}{\cal C}\) \(\newcommand{\scal}{\cal S}\) \(\newcommand{\wcal}{\cal W}\) \(\newcommand{\ecal}{\cal E}\) \(\newcommand{\coords}[2]{\left\{#1\right\}_{#2}}\) \(\newcommand{\gray}[1]{\color{gray}{#1}}\) \(\newcommand{\lgray}[1]{\color{lightgray}{#1}}\) \(\newcommand{\rank}{\operatorname{rank}}\) \(\newcommand{\row}{\text{Row}}\) \(\newcommand{\col}{\text{Col}}\) \(\renewcommand{\row}{\text{Row}}\) \(\newcommand{\nul}{\text{Nul}}\) \(\newcommand{\var}{\text{Var}}\) \(\newcommand{\corr}{\text{corr}}\) \(\newcommand{\len}[1]{\left|#1\right|}\) \(\newcommand{\bbar}{\overline{\bvec}}\) \(\newcommand{\bhat}{\widehat{\bvec}}\) \(\newcommand{\bperp}{\bvec^\perp}\) \(\newcommand{\xhat}{\widehat{\xvec}}\) \(\newcommand{\vhat}{\widehat{\vvec}}\) \(\newcommand{\uhat}{\widehat{\uvec}}\) \(\newcommand{\what}{\widehat{\wvec}}\) \(\newcommand{\Sighat}{\widehat{\Sigma}}\) \(\newcommand{\lt}{<}\) \(\newcommand{\gt}{>}\) \(\newcommand{\amp}{&}\) \(\definecolor{fillinmathshade}{gray}{0.9}\)MEDIA
One plate of each of the following media:
- Trypticase Soy agar,
- Columbia CNA agar, and
- MacConkey agar.
ORGANISMS
A broth culture containing a mixture of one of the following Gram-positive bacteria and one of the following Gram-negative bacteria:
- Possible Gram-positive bacteria:
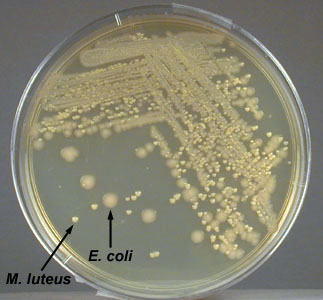
- Micrococcus luteus. A Gram-positive coccus with a tetrad or a sarcina arrangement; produces circular, convex colonies with a yellow, water-insoluble pigment on Trypticase Soy agar.
- Micrococcus luteus growing on TSA
- Close up of Micrococcus luteus growing on TSA
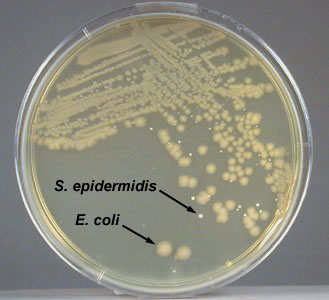
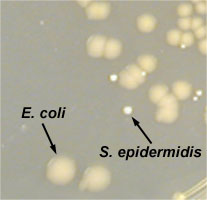
- Staphylococcus epidermidis. A Gram-positive coccus with a staphylococcus arrangement; produces circular, convex, non-pigmented colonies on Trypticase Soy agar.
- Staphylococcus epidermidis growing on TSA
- Close up of Staphylococcus epidermidis growing on TSA
- Micrococcus luteus. A Gram-positive coccus with a tetrad or a sarcina arrangement; produces circular, convex colonies with a yellow, water-insoluble pigment on Trypticase Soy agar.
|
Fig: \(\PageIndex{1A}\): Isolation Plate: Mixture of Escherichia coli and Micrococcus luteus Grown on TSA |
Fig. \(\PageIndex{1B}\): Close Up of Escherichia coli and Micrococcus luteus Grown on TSA |
Fig. \(\PageIndex{1C}\): Isolation Plate: Mixture of Escherichia coli and Staphylococcus epidermidis Grown on TSA |
Fig. \(\PageIndex{1D}\): Close Up of Escherichia coli and Staphylococcus epidermidis Grown on TSA |
|---|---|---|---|
 |
 |
 |
|
| Copyright; Gary E. Kaiser, Ph.D. The Community College of Baltimore County, Catonsville Campus CC-BY-3.0 | |||
- Possible Gram-negative bacteria:
- Escherichia coli. A Gram-negative bacillus; produces irregular, raised, non-pigmented colonies on Trypticase Soy agar.
- Escherichia coli growing on TSA
- Enterobacter aerogenes. A Gram-negative bacillus; produces irregular raised, non-pigmented, possibly mucoid colonies on Trypticase Soy agar.
- Enterobacter aerogenes growing on TSA
- Escherichia coli. A Gram-negative bacillus; produces irregular, raised, non-pigmented colonies on Trypticase Soy agar.
|
Fig: \(\PageIndex{1E}\): Isolation Plate: Mixture of Escherichia coli and Micrococcus luteus Grown on TSA |
Fig. \(\PageIndex{1F}\): Mixture of Klebsiella aerogenes (formerly known as Enterobacter aerogenes) and Staphylococcus epidermidis Grown on TSA |
Fig. \(\PageIndex{1G}\): Pseudomonas aeruginosa Growing on TSA |
|---|---|---|
 |
 |
|
| Copyright; Gary E. Kaiser, Ph.D. The Community College of Baltimore County, Catonsville Campus CC-BY-3.0 | ||
During the next three labs you will attempt to obtain pure cultures of each organism in your mixture and determine which two bacteria you have. Today you will try to separate the bacteria in the mixture in order to obtain isolated colonies; next lab you will identify the two bacteria in your mixture and pick off single isolated colonies of each of the two bacteria in order to get a pure culture of each. The following lab you will prepare microscopy slides of each of the two pure cultures to determine if they are indeed pure.
PROCEDURE (to be done in pairs)
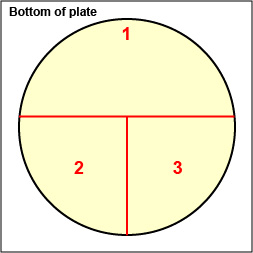
1. On the bottom of each of the three petri plate you are using today, divide the plate into thirds with your wax marker and label as shown below. This will guide your streaking.
2. Although Trypticase Soy agar (TSA), which grows both Gram-positive and Gram-negative bacteria, is not normally used as an isolation medium, we will attempt to obtain isolated colonies of the two organisms in your mixture by using strictly mechanical methods. Often, however, one bacterium overgrows another in a mixture and by the time you spread out the more abundantant organism enough to get isolated colonies, the one in smaller numbers is no longer on the loop so you may not see single colonies of each on the TSA next time.
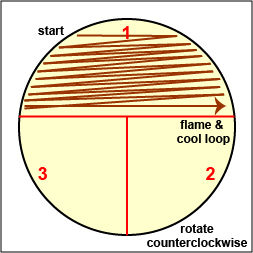
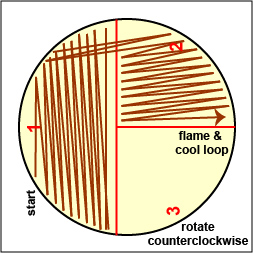
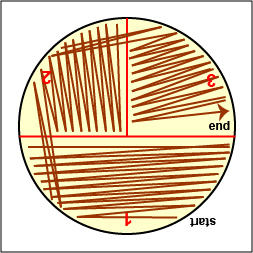
Streak your mixture on a plate of Trypticase Soy agar using one of the two streaking patterns illustrated in Lab 2, Fig. 4 and Fig. 5. agar
|
Fig. \(\PageIndex{2A}\): Label Plate to Guide your Streaking |
Fig. \(\PageIndex{2B}\): Streaking For Isolation, Step-1 |
Fig. \(\PageIndex{2C}\): Streaking For Isolation, Step-2 |
Fig. \(\PageIndex{2D}\): Streaking For Isolation, Step-3 |
 |
 |
 |
 |
| Copyright; Gary E. Kaiser, Ph.D. The Community College of Baltimore County, Catonsville Campus CC-BY-3.0 | |||

Video Review
 |
3. Streak the same mixture for isolation on a plate of Columbia CNA agar (selective for Gram-positive bacteria).
- Micrococcus luteus growing on Columbia CNA agar. See Fig. 8E.
- Staphylococcus epidermidis growing on Columbia CNA agar. See Fig. 8F.
|
Fig: \(\PageIndex{2E}\): Micrococcus luteus Growing on Columbia CNA Agar |
Fig. \(\PageIndex{2f}\): Staphylococcus epidermidis> Growing on Columbia CNA Agar |
|---|---|
| Only Gram-positive bacteria grow on Columbia CNA agar. (Gram-negative bacteria are inhibited by the colistin and nalidixic acid added to the agar.) Micrococcus luteus produces a yellow, water insoluble pigment. | Only Gram-positive bacteria grow on Columbia CNA agar. (Gram-negative bacteria are inhibited by the colistin and nalidixic acid added to the agar.) Staphylococcus epidermidis does not normally produce pigment. |
| Copyright; Gary E. Kaiser, Ph.D. The Community College of Baltimore County, Catonsville Campus CC-BY-3.0 | |
4. Streak the same mixture for isolation on a plate of MacConkey agar (selective for Gram-negative bacteria and differential for certain members of the bacterial family Enterobacteriaceae).
- Escherichia coli growing on MacConkey agar. See Fig. 8G.
- Klebsiella aerogenes (formerly known as Enterobacter aerogenes) growing on MacConkey agar. See Fig. 8H.
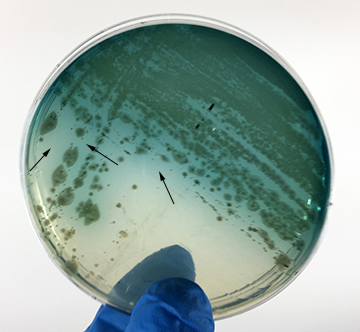
- Pseudomonas aeruginosa growing on MacConkey agar. See Fig. 8I.
|
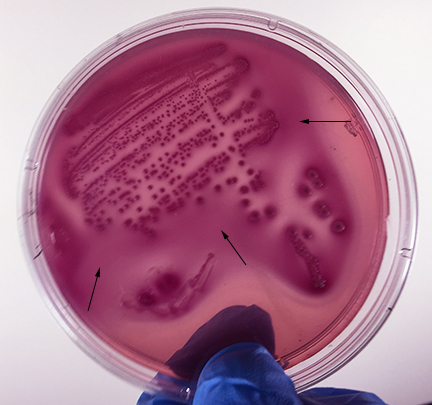
Fig: \(\PageIndex{2F}\): Escherichia coli Growing on MacConkey Agar |
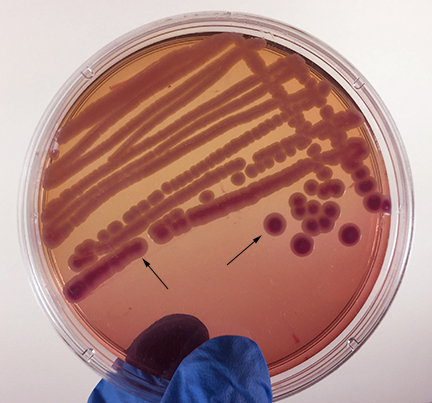
Fig. \(\PageIndex{2G}\): Klebsiella aerogenes (formerly known as Enterobacter aerogenes) Growing on MacConkey Agar |
Fig. \(\PageIndex{H}\): Pseudomonas aeruginosa Growing on MacConkey Agar |
|---|---|---|
 |
 |
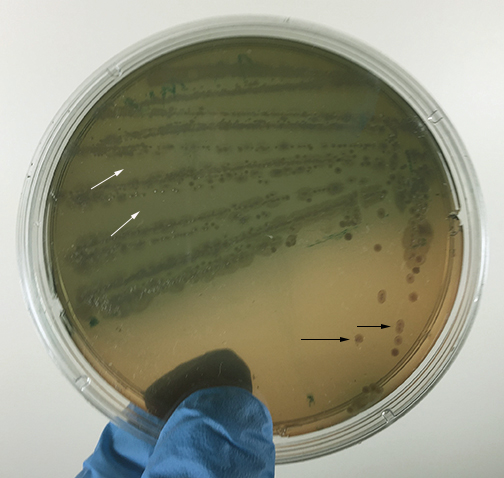 |
| Strong fementation of lactose with high levels of acid production by the bacteria causes the colonies and confluent growth to appear bright pink to red. The resulting acid, at high enough concentrations, can also causes the bile salts in the medium to precipitate out of solution causing a pink precipitate (cloudiness) o appear around the the growth (arrows). | Weak fermentation of lactose by the bacteria causes the colonies and confluent growth to appear pink or pink with red centers (arrows), but without the precipitation of bile salts there is no pink halo around the growth. | If the bacteria do not ferment lactose, the colonies and confluent growth appear relatively colorless and the agar surrounding the bacteria remains relatively transparent. The green, water-soluble pigment produced by Pseudomonas aeruginosa (white arrows) is also evident here. |
| Copyright; Gary E. Kaiser, Ph.D. The Community College of Baltimore County, Catonsville Campus CC-BY-3.0 | ||
5. Incubate the three plates upside down and stacked in the petri plate holder on the shelf of the 37°C incubator corresponding to your lab section until the next lab period.
Contributors and Attributions
Dr. Gary Kaiser (COMMUNITY COLLEGE OF BALTIMORE COUNTY, CATONSVILLE CAMPUS)

