10.8: Testing the Effectiveness of Antimicrobial Chemicals and Drugs
- Page ID
- 75870
- Describe why the phenol coefficient is used
- Describe how the Kirby-Bauer disk diffusion test determines the susceptibility of a microbe to an antibacterial drug.
- Explain the significance of the minimal inhibitory concentration and the minimal bactericidal concentration relative to the effectiveness of an antimicrobial drug.
- Compare and contrast the disk-diffusion, use-dilution, and in-use methods for testing the effectiveness of antiseptics, disinfectants, and sterilants
The effectiveness of various chemical disinfectants is reflected in the terms used to describe them. Chemical disinfectants are grouped by the power of their activity, with each category reflecting the types of microbes and viruses its component disinfectants are effective against. High-level germicides have the ability to kill vegetative cells, fungi, viruses, and endospores, leading to sterilization, with extended use. Intermediate-level germicides, as their name suggests, are less effective against endospores and certain viruses, and low-level germicides kill only vegetative cells and certain enveloped viruses, and are ineffective against endospores.
However, several environmental conditions influence the potency of an antimicrobial agent and its effectiveness. For example, length of exposure is particularly important, with longer exposure increasing efficacy. Similarly, the concentration of the chemical agent is also important, with higher concentrations being more effective than lower ones. Temperature, pH, and other factors can also affect the potency of a disinfecting agent.
One method to determine the effectiveness of a chemical agent includes swabbing surfaces before and after use to confirm whether a sterile field was maintained during use. Additional tests are described in the sections that follow. These tests allow for the maintenance of appropriate disinfection protocols in clinical settings, controlling microbial growth to protect patients, health-care workers, and the community.
Phenol Coefficient
The effectiveness of a disinfectant or antiseptic can be determined in a number of ways. Historically, a chemical agent’s effectiveness was often compared with that of phenol, the first chemical agent used by Joseph Lister. In 1903, British chemists Samuel Rideal (1863–1929) and J. T. Ainslie Walker (1868–1930) established a protocol to compare the effectiveness of a variety of chemicals with that of phenol, using as their test organisms Staphylococcus aureus (a gram-positive bacterium) and Salmonella enterica serovar Typhi (a gram-negative bacterium). They exposed the test bacteria to the antimicrobial chemical solutions diluted in water for 7.5 minutes. They then calculated a phenol coefficient for each chemical for each of the two bacteria tested. A phenol coefficient of 1.0 means that the chemical agent has about the same level of effectiveness as phenol. A chemical agent with a phenol coefficient of less than 1.0 is less effective than phenol. An example is formalin, with phenol coefficients of 0.3 (S. aureus) and 0.7 (S. enterica serovar Typhi). A chemical agent with a phenol coefficient greater than 1.0 is more effective than phenol, such as chloramine, with phenol coefficients of 133 and 100, respectively. Although the phenol coefficient was once a useful measure of effectiveness, it is no longer commonly used because the conditions and organisms used were arbitrarily chosen.
What are the differences between the three levels of disinfectant effectiveness?
Disk-Diffusion (Kirby-Bauer) Method
The Kirby-Bauer disk diffusion test has long been used as a starting point for determining the susceptibility of specific microbes to various antimicrobial drugs or chemicals. The Kirby-Bauer assay starts with a Mueller-Hinton agar plate on which a confluent lawn (bacteria is spread across the entire surface of the plate) is inoculated with a sample or patient’s isolated bacterial pathogen. Filter paper disks impregnated with known amounts of antibacterial drugs or chemicals to be tested are then placed on the agar plate. As the bacterial inoculum grows, drug or chemical diffuses from the circular disk into the agar and interacts with the growing bacteria. Antimicrobial activity is observed as a clear circular zone of inhibition around the drug/chemical-impregnated disk. The diameter of the zone of inhibition, measured in millimeters and compared to a standardized chart, determines the susceptibility or resistance of the bacterial pathogen to the drug or chemical.(Figure \(\PageIndex{1}\))
There are multiple factors that determine the size of a zone of inhibition in this assay, including drug solubility (whether the agent is water soluble and able to diffuse in the agar), rate of drug diffusion through agar, the thickness of the agar medium, and the drug concentration impregnated into the disk. Due to a lack of standardization of these factors, interpretation of the Kirby-Bauer disk diffusion assay provides only limited information on susceptibility and resistance to the drugs tested. The assay cannot distinguish between bacteriostatic and bactericidal activities, and differences in zone sizes cannot be used to compare drug potencies or efficacies. Comparison of zone sizes to a standardized chart will only provide information on the antibacterials to which a bacterial pathogen is susceptible or resistant. However it is common to correlate larger zones to increased inhibition effectiveness of the chemical agent.
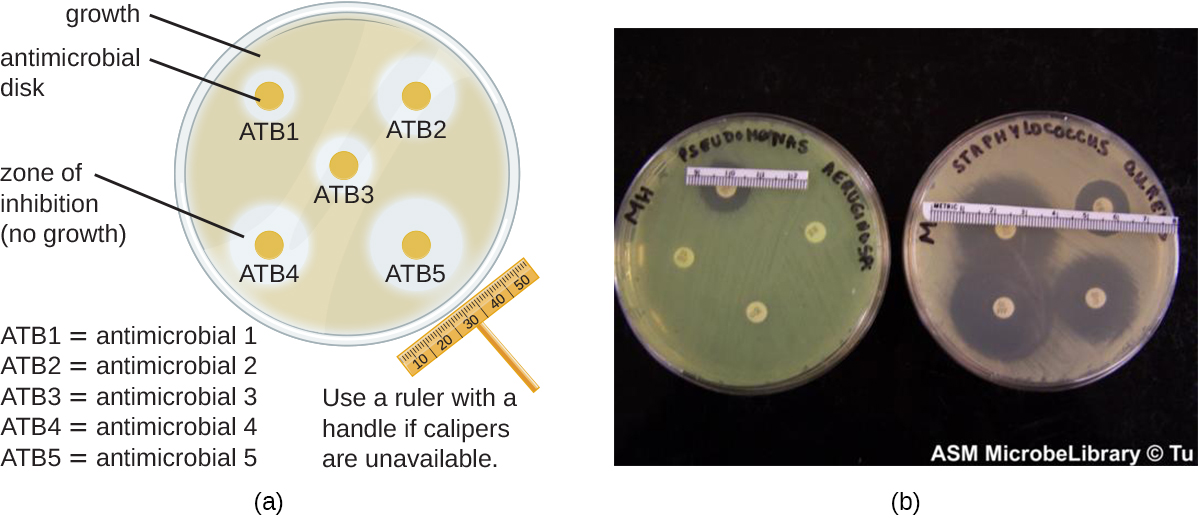
When comparing the activities of two disinfectants against the same microbe, using the disk-diffusion assay, and assuming both are water soluble and can easily diffuse in the agar, would a more effective disinfectant have a larger zone of inhibition or a smaller one?
How does one use the information from a Kirby-Bauer assay to predict the therapeutic effectiveness of an antimicrobial drug in a patient?
Unfortunately, infectious diseases don’t take a time-out for lab work. As a result, physicians rarely have the luxury of conducting susceptibility testing before they write a prescription. Instead, they rely primarily on the empirical evidence (i.e., the signs and symptoms of disease) and their professional experience to make an educated guess as to the diagnosis, causative agent(s), and drug most likely to be effective. This approach allows treatment to begin sooner so the patient does not have to wait for lab test results. In many cases, the prescription is effective; however, in an age of increased antimicrobial resistance, it is becoming increasingly more difficult to select the most appropriate empiric therapy. Selecting an inappropriate empiric therapy not only puts the patient at risk but may promote greater resistance to the drug prescribed.
Recently, studies have shown that antibiograms are useful tools in the decision-making process of selecting appropriate empiric therapy. An antibiogram is a compilation of local antibiotic susceptibility data broken down by bacterial pathogen. In a November 2014 study published in the journal Infection Control and Hospital Epidemiology, researchers determined that 85% of the prescriptions ordered in skilled nursing facilities were decided upon empirically, but only 35% of those prescriptions were deemed appropriate when compared with the eventual pathogen identification and susceptibility profile obtained from the clinical laboratory. However, in one nursing facility where use of antibiograms was implemented to direct selection of empiric therapy, appropriateness of empiric therapy increased from 32% before antibiogram implementation to 45% after implementation of antibiograms.1 Although these data are preliminary, they do suggest that health-care facilities can reduce the number of inappropriate prescriptions by using antibiograms to select empiric therapy, thus benefiting patients and minimizing opportunities for antimicrobial resistance to develop.
Dilution Tests
As discussed, the limitations of the Kirby-Bauer disk diffusion test do not allow for a direct comparison of antibacterial potencies to guide selection of the best therapeutic choice. However, antibacterial dilution tests can be used to determine a particular drug’s minimal inhibitory concentration (MIC), the lowest concentration of drug that inhibits visible bacterial growth, and minimal bactericidal concentration (MBC), the lowest drug concentration that kills ≥99.9% of the starting inoculum. Determining these concentrations helps identify the correct drug for a particular pathogen. For the macrobroth dilution assay, a dilution series of the drug in broth is made in test tubes and the same number of cells of a test bacterial strain is added to each tube (Figure \(\PageIndex{2}\)). The MIC is determined by examining the tubes to find the lowest drug concentration that inhibits visible growth; this is observed as turbidity (cloudiness) in the broth. Tubes with no visible growth are then inoculated onto agar media without antibiotic to determine the MBC. Generally, serum levels of an antibacterial should be at least three to five times above the MIC for treatment of an infection.

The MIC assay can also be performed using 96-well microdilution trays, which allow for the use of small volumes and automated dispensing devices, as well as the testing of multiple antimicrobials and/or microorganisms in one tray (Figure \(\PageIndex{3}\)). MICs are interpreted as the lowest concentration that inhibits visible growth, the same as for the macrobroth dilution in test tubes. Growth may also be interpreted visually or by using a spectrophotometer or similar device to detect turbidity or a color change if an appropriate biochemical substrate that changes color in the presence of bacterial growth is also included in each well.

The Etest is an alternative method used to determine MIC, and is a combination of the Kirby-Bauer disk diffusion test and dilution methods. Similar to the Kirby-Bauer assay, a confluent lawn of a bacterial isolate is inoculated onto the surface of an agar plate. Rather than using circular disks impregnated with one concentration of drug, however, commercially available plastic strips that contain a gradient of an antibacterial are placed on the surface of the inoculated agar plate (Figure \(\PageIndex{4}\)). As the bacterial inoculum grows, antibiotic diffuses from the plastic strips into the agar and interacts with the bacterial cells. Because the rate of drug diffusion is directly related to concentration, an elliptical zone of inhibition is observed with the Etest drug gradient, rather than a circular zone of inhibition observed with the Kirby-Bauer assay. To interpret the results, the intersection of the elliptical zone with the gradient on the drug-containing strip indicates the MIC. Because multiple strips containing different antimicrobials can be placed on the same plate, the MIC of multiple antimicrobials can be determined concurrently and directly compared. However, unlike the macrobroth and microbroth dilution methods, the MBC cannot be determined with the Etest.

Compare and contrast MIC and MBC.
Use-Dilution Test
Other methods are also used for measuring the effectiveness of a chemical agent in clinical settings. The use-dilution test is commonly used to determine a chemical’s disinfection effectiveness on an inanimate surface. For this test, a cylinder of stainless steel is dipped in a culture of the targeted microorganism and then dried. The cylinder is then dipped in solutions of disinfectant at various concentrations for a specified amount of time. Finally, the cylinder is transferred to a new test tube containing fresh sterile medium that does not contain disinfectant, and this test tube is incubated. Bacterial survival is demonstrated by the presence of turbidity in the medium, whereas killing of the target organism on the cylinder by the disinfectant will produce no turbidity.
The Association of Official Agricultural Chemists International (AOAC), a nonprofit group that establishes many protocol standards, has determined that a minimum of 59 of 60 replicates must show no growth in such a test to achieve a passing result, and the results must be repeatable from different batches of disinfectant and when performed on different days. Disinfectant manufacturers perform use-dilution tests to validate the efficacy claims for their products, as designated by the EPA.
Is the use-dilution test performed in a clinical setting? Why?
In-Use Test
An in-use test can determine whether an actively used solution of disinfectant in a clinical setting is microbially contaminated (Figure \(\PageIndex{5}\)). A 1-mL sample of the used disinfectant is diluted into 9 mL of sterile broth medium that also contains a compound to inactivate the disinfectant. Ten drops, totaling approximately 0.2 mL of this mixture, are then inoculated onto each of two agar plates. One plate is incubated at 37 °C for 3 days and the other is incubated at room temperature for 7 days. The plates are monitored for growth of microbial colonies. Growth of five or more colonies on either plate suggests that viable microbial cells existed in the disinfectant solution and that it is contaminated. Such in-use tests monitor the effectiveness of disinfectants in the clinical setting.
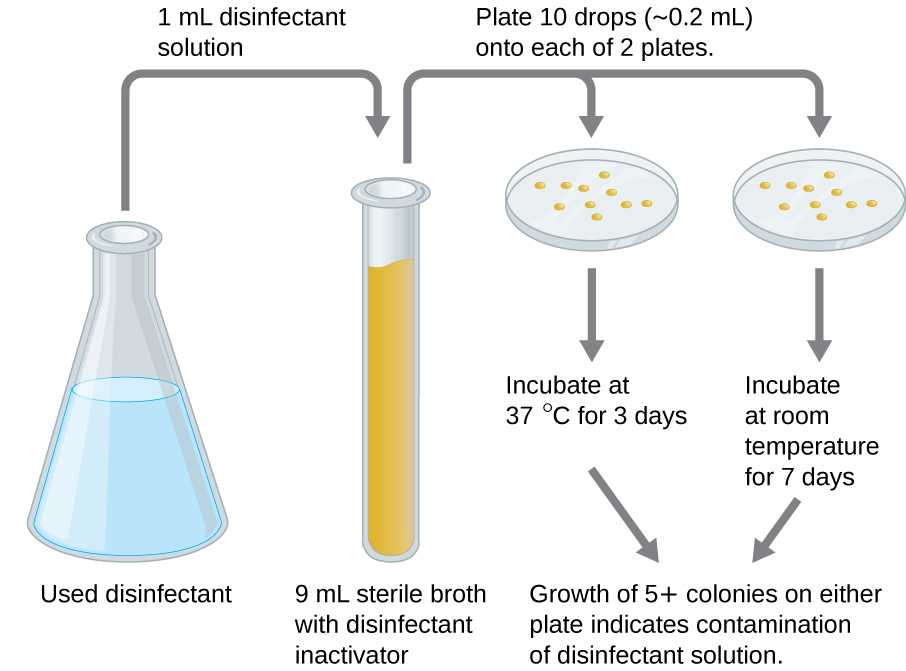
What does a positive in-use test indicate?
Despite antibiotic treatment, Roberta’s symptoms worsened. She developed pyelonephritis, a severe kidney infection, and was rehospitalized in the intensive care unit (ICU). Her condition continued to deteriorate, and she developed symptoms of septic shock. At this point, her physician ordered a culture from her urine to determine the exact cause of her infection, as well as a drug sensitivity test to determine what antibiotics would be effective against the causative bacterium. The results of this test indicated resistance to a wide range of antibiotics, including the carbapenems, a class of antibiotics that are used as the last resort for many types of bacterial infections. This was an alarming outcome, suggesting that Roberta’s infection was caused by a so-called superbug: a bacterial strain that has developed resistance to the majority of commonly used antibiotics. In this case, the causative agent belonged to the carbapenem-resistant Enterobacteriaceae (CRE), a drug-resistant family of bacteria normally found in the digestive system . When CRE is introduced to other body systems, as might occur through improperly cleaned surgical instruments, catheters, or endoscopes, aggressive infections can occur.
CRE infections are notoriously difficult to treat, with a 40%–50% fatality rate. To treat her kidney infection and septic shock, Roberta was treated with dialysis, intravenous fluids, and medications to maintain blood pressure and prevent blood clotting. She was also started on aggressive treatment with intravenous administration of a new drug called tigecycline, which has been successful in treating infections caused by drug-resistant bacteria.
After several weeks in the ICU, Roberta recovered from her CRE infection. However, public health officials soon noticed that Roberta’s case was not isolated. Several patients who underwent similar procedures at the same hospital also developed CRE infections, some dying as a result. Ultimately, the source of the infection was traced to the duodenoscopes used in the procedures. Despite the hospital staff meticulously following manufacturer protocols for disinfection, bacteria, including CRE, remained within the instruments and were introduced to patients during procedures.
Carbapenem-resistant Enterobacteriaceae infections due to contaminated endoscopes have become a high-profile problem in recent years. Several CRE outbreaks have been traced to endoscopes, including a case at Ronald Reagan UCLA Medical Center in early 2015 in which 179 patients may have been exposed to a contaminated endoscope. Seven of the patients developed infections, and two later died. Several lawsuits have been filed against Olympus, the manufacturer of the endoscopes. Some claim that Olympus did not obtain FDA approval for design changes that may have led to contamination, and others claim that the manufacturer knowingly withheld information from hospitals concerning defects in the endoscopes.
Lawsuits like these raise difficult-to-answer questions about liability. Invasive procedures are inherently risky, but negative outcomes can be minimized by strict adherence to established protocols. Who is responsible, however, when negative outcomes occur due to flawed protocols or faulty equipment? Can hospitals or health-care workers be held liable if they have strictly followed a flawed procedure? Should manufacturers be held liable—and perhaps be driven out of business—if their lifesaving equipment fails or is found defective? What is the government’s role in ensuring that use and maintenance of medical equipment and protocols are fail-safe?
Protocols for cleaning or sterilizing medical equipment are often developed by government agencies like the FDA, and other groups, like the AOAC, a nonprofit scientific organization that establishes many protocols for standard use globally. These procedures and protocols are then adopted by medical device and equipment manufacturers. Ultimately, the end-users (hospitals and their staff) are responsible for following these procedures and can be held liable if a breach occurs and patients become ill from improperly cleaned equipment.
Unfortunately, protocols are not infallible, and sometimes it takes negative outcomes to reveal their flaws. In 2008, the FDA had approved a disinfection protocol for endoscopes, using glutaraldehyde (at a lower concentration when mixed with phenol), o-phthalaldehyde, hydrogen peroxide, peracetic acid, and a mix of hydrogen peroxide with peracetic acid. However, subsequent CRE outbreaks from endoscope use showed that this protocol alone was inadequate.
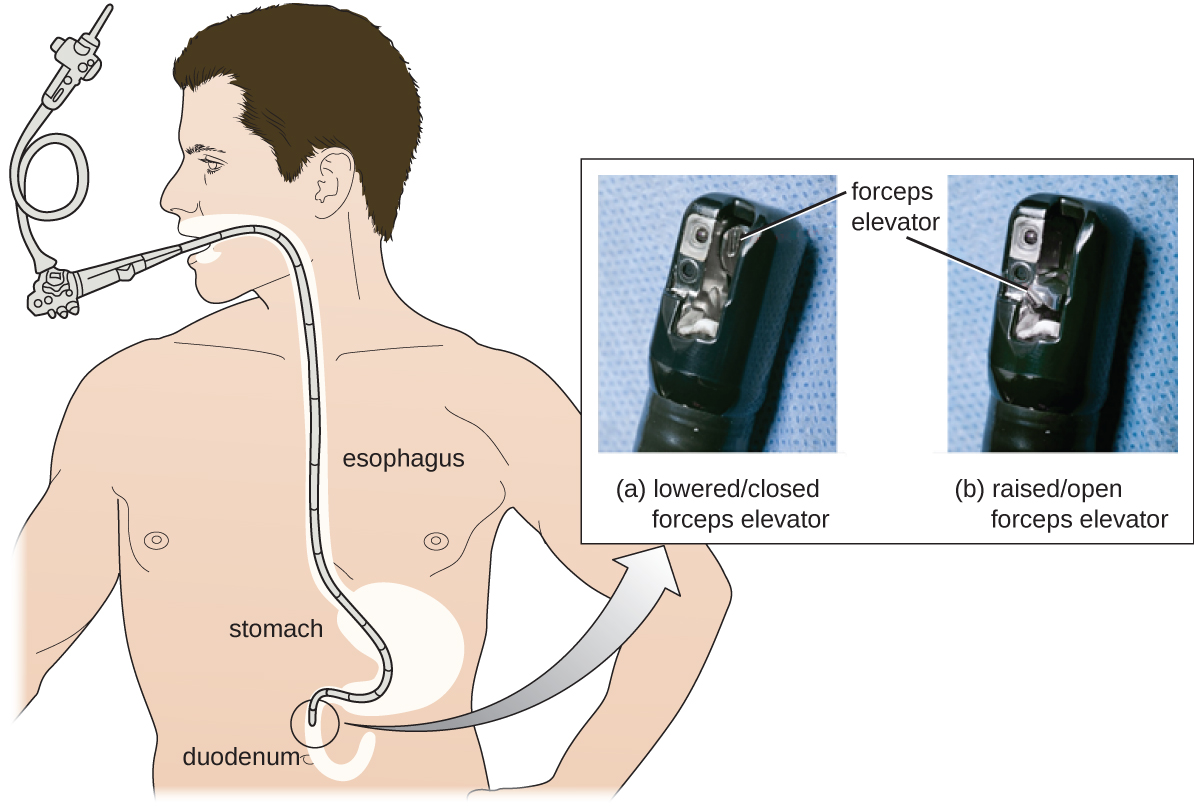
As a result of CRE outbreaks, hospitals, manufacturers, and the FDA are investigating solutions. Many hospitals are instituting more rigorous cleaning procedures than those mandated by the FDA. Manufacturers are looking for ways to redesign duodenoscopes to minimize hard-to-reach crevices where bacteria can escape disinfectants, and the FDA is updating its protocols. In February 2015, the FDA added new recommendations for careful hand cleaning of the duodenoscope elevator mechanism (the location where microbes are most likely to escape disinfection), and issued more careful documentation about quality control of disinfection protocols (Figure \(\PageIndex{7}\)).
There is no guarantee that new procedures, protocols, or equipment will completely eliminate the risk for infection associated with endoscopes. Yet these devices are used successfully in 500,000–650,000 procedures annually in the United States, many of them lifesaving. At what point do the risks outweigh the benefits of these devices, and who should be held responsible when negative outcomes occur?
Key Concepts and Summary
- Chemical disinfectants are grouped by the types of microbes and infectious agents they are effective against. High-level germicides kill vegetative cells, fungi, viruses, and endospores, and can ultimately lead to sterilization. Intermediate-level germicides cannot kill all viruses and are less effective against endospores. Low-level germicides kill vegetative cells and some enveloped viruses, but are ineffective against endospores.
- The effectiveness of a disinfectant is influenced by several factors, including length of exposure, concentration of disinfectant, temperature, and pH.
- Historically, the effectiveness of a chemical disinfectant was compared with that of phenol at killing Staphylococcus aureus and Salmonella enterica serovar Typhi, and a phenol coefficient was calculated.
- The disk-diffusion method is used to test the effectiveness of a chemical disinfectant against a particular microbe.
- The Kirby-Bauer disk diffusion test helps determine the susceptibility of a microorganism to various antimicrobial drugs. However, the zones of inhibition measured must be correlated to known standards to determine susceptibility and resistance, and do not provide information on bactericidal versus bacteriostatic activity, or allow for direct comparison of drug potencies.
- Antibiograms are useful for monitoring local trends in antimicrobial resistance/susceptibility and for directing appropriate selection of empiric antibacterial therapy.
- There are several laboratory methods available for determining the minimum inhibitory concentration (MIC) of an antimicrobial drug against a specific microbe. The minimal bactericidal concentration (MBC) can also be determined, typically as a follow-up experiment to MIC determination using the tube dilution method.
- The use-dilution test determines the effectiveness of a disinfectant on a surface. In-use tests can determine whether disinfectant solutions are being used correctly in clinical settings.
Footnotes
- J.P. Furuno et al. “Using Antibiograms to Improve Antibiotic Prescribing in Skilled Nursing Facilities.” Infection Control and Hospital Epidemiology 35 no. Suppl S3 (2014):S56–61.
Contributors and Attributions
Nina Parker, (Shenandoah University), Mark Schneegurt (Wichita State University), Anh-Hue Thi Tu (Georgia Southwestern State University), Philip Lister (Central New Mexico Community College), and Brian M. Forster (Saint Joseph’s University) with many contributing authors. Original content via Openstax (CC BY 4.0; Access for free at https://openstax.org/books/microbiology/pages/1-introduction)


