14.5: Practical Applications of Monoclonal and Polyclonal Antibodies
- Page ID
- 31869
\( \newcommand{\vecs}[1]{\overset { \scriptstyle \rightharpoonup} {\mathbf{#1}} } \)
\( \newcommand{\vecd}[1]{\overset{-\!-\!\rightharpoonup}{\vphantom{a}\smash {#1}}} \)
\( \newcommand{\id}{\mathrm{id}}\) \( \newcommand{\Span}{\mathrm{span}}\)
( \newcommand{\kernel}{\mathrm{null}\,}\) \( \newcommand{\range}{\mathrm{range}\,}\)
\( \newcommand{\RealPart}{\mathrm{Re}}\) \( \newcommand{\ImaginaryPart}{\mathrm{Im}}\)
\( \newcommand{\Argument}{\mathrm{Arg}}\) \( \newcommand{\norm}[1]{\| #1 \|}\)
\( \newcommand{\inner}[2]{\langle #1, #2 \rangle}\)
\( \newcommand{\Span}{\mathrm{span}}\)
\( \newcommand{\id}{\mathrm{id}}\)
\( \newcommand{\Span}{\mathrm{span}}\)
\( \newcommand{\kernel}{\mathrm{null}\,}\)
\( \newcommand{\range}{\mathrm{range}\,}\)
\( \newcommand{\RealPart}{\mathrm{Re}}\)
\( \newcommand{\ImaginaryPart}{\mathrm{Im}}\)
\( \newcommand{\Argument}{\mathrm{Arg}}\)
\( \newcommand{\norm}[1]{\| #1 \|}\)
\( \newcommand{\inner}[2]{\langle #1, #2 \rangle}\)
\( \newcommand{\Span}{\mathrm{span}}\) \( \newcommand{\AA}{\unicode[.8,0]{x212B}}\)
\( \newcommand{\vectorA}[1]{\vec{#1}} % arrow\)
\( \newcommand{\vectorAt}[1]{\vec{\text{#1}}} % arrow\)
\( \newcommand{\vectorB}[1]{\overset { \scriptstyle \rightharpoonup} {\mathbf{#1}} } \)
\( \newcommand{\vectorC}[1]{\textbf{#1}} \)
\( \newcommand{\vectorD}[1]{\overrightarrow{#1}} \)
\( \newcommand{\vectorDt}[1]{\overrightarrow{\text{#1}}} \)
\( \newcommand{\vectE}[1]{\overset{-\!-\!\rightharpoonup}{\vphantom{a}\smash{\mathbf {#1}}}} \)
\( \newcommand{\vecs}[1]{\overset { \scriptstyle \rightharpoonup} {\mathbf{#1}} } \)
\( \newcommand{\vecd}[1]{\overset{-\!-\!\rightharpoonup}{\vphantom{a}\smash {#1}}} \)
\(\newcommand{\avec}{\mathbf a}\) \(\newcommand{\bvec}{\mathbf b}\) \(\newcommand{\cvec}{\mathbf c}\) \(\newcommand{\dvec}{\mathbf d}\) \(\newcommand{\dtil}{\widetilde{\mathbf d}}\) \(\newcommand{\evec}{\mathbf e}\) \(\newcommand{\fvec}{\mathbf f}\) \(\newcommand{\nvec}{\mathbf n}\) \(\newcommand{\pvec}{\mathbf p}\) \(\newcommand{\qvec}{\mathbf q}\) \(\newcommand{\svec}{\mathbf s}\) \(\newcommand{\tvec}{\mathbf t}\) \(\newcommand{\uvec}{\mathbf u}\) \(\newcommand{\vvec}{\mathbf v}\) \(\newcommand{\wvec}{\mathbf w}\) \(\newcommand{\xvec}{\mathbf x}\) \(\newcommand{\yvec}{\mathbf y}\) \(\newcommand{\zvec}{\mathbf z}\) \(\newcommand{\rvec}{\mathbf r}\) \(\newcommand{\mvec}{\mathbf m}\) \(\newcommand{\zerovec}{\mathbf 0}\) \(\newcommand{\onevec}{\mathbf 1}\) \(\newcommand{\real}{\mathbb R}\) \(\newcommand{\twovec}[2]{\left[\begin{array}{r}#1 \\ #2 \end{array}\right]}\) \(\newcommand{\ctwovec}[2]{\left[\begin{array}{c}#1 \\ #2 \end{array}\right]}\) \(\newcommand{\threevec}[3]{\left[\begin{array}{r}#1 \\ #2 \\ #3 \end{array}\right]}\) \(\newcommand{\cthreevec}[3]{\left[\begin{array}{c}#1 \\ #2 \\ #3 \end{array}\right]}\) \(\newcommand{\fourvec}[4]{\left[\begin{array}{r}#1 \\ #2 \\ #3 \\ #4 \end{array}\right]}\) \(\newcommand{\cfourvec}[4]{\left[\begin{array}{c}#1 \\ #2 \\ #3 \\ #4 \end{array}\right]}\) \(\newcommand{\fivevec}[5]{\left[\begin{array}{r}#1 \\ #2 \\ #3 \\ #4 \\ #5 \\ \end{array}\right]}\) \(\newcommand{\cfivevec}[5]{\left[\begin{array}{c}#1 \\ #2 \\ #3 \\ #4 \\ #5 \\ \end{array}\right]}\) \(\newcommand{\mattwo}[4]{\left[\begin{array}{rr}#1 \amp #2 \\ #3 \amp #4 \\ \end{array}\right]}\) \(\newcommand{\laspan}[1]{\text{Span}\{#1\}}\) \(\newcommand{\bcal}{\cal B}\) \(\newcommand{\ccal}{\cal C}\) \(\newcommand{\scal}{\cal S}\) \(\newcommand{\wcal}{\cal W}\) \(\newcommand{\ecal}{\cal E}\) \(\newcommand{\coords}[2]{\left\{#1\right\}_{#2}}\) \(\newcommand{\gray}[1]{\color{gray}{#1}}\) \(\newcommand{\lgray}[1]{\color{lightgray}{#1}}\) \(\newcommand{\rank}{\operatorname{rank}}\) \(\newcommand{\row}{\text{Row}}\) \(\newcommand{\col}{\text{Col}}\) \(\renewcommand{\row}{\text{Row}}\) \(\newcommand{\nul}{\text{Nul}}\) \(\newcommand{\var}{\text{Var}}\) \(\newcommand{\corr}{\text{corr}}\) \(\newcommand{\len}[1]{\left|#1\right|}\) \(\newcommand{\bbar}{\overline{\bvec}}\) \(\newcommand{\bhat}{\widehat{\bvec}}\) \(\newcommand{\bperp}{\bvec^\perp}\) \(\newcommand{\xhat}{\widehat{\xvec}}\) \(\newcommand{\vhat}{\widehat{\vvec}}\) \(\newcommand{\uhat}{\widehat{\uvec}}\) \(\newcommand{\what}{\widehat{\wvec}}\) \(\newcommand{\Sighat}{\widehat{\Sigma}}\) \(\newcommand{\lt}{<}\) \(\newcommand{\gt}{>}\) \(\newcommand{\amp}{&}\) \(\definecolor{fillinmathshade}{gray}{0.9}\)Learning Objectives
- Compare the method of development, use, and characteristics of monoclonal and polyclonal antibodies
- Explain the nature of antibody cross-reactivity and why this is less of a problem with monoclonal antibodies
In addition to being crucial for our normal immune response, antibodies provide powerful tools for research and diagnostic purposes. The high specificity of antibodies makes them an excellent tool for detecting and quantifying a broad array of targets, from drugs to serum proteins to microorganisms. With in vitro assays, antibodies can be used to precipitate soluble antigens, agglutinate (clump) cells, opsonize and kill bacteria with the assistance of complement, and neutralize drugs, toxins, and viruses.
Measuring Specificity
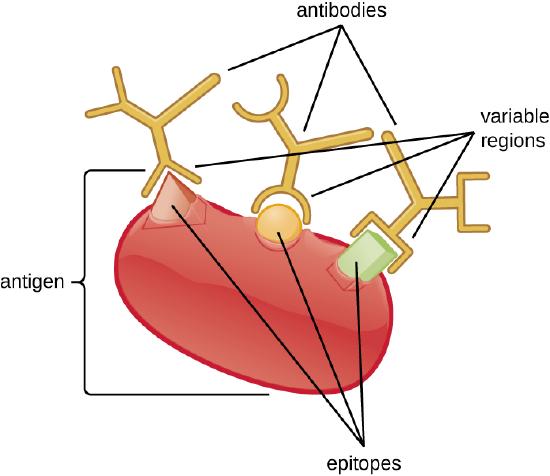
An antibody’s specificity results from the antigen-binding site formed within the variable regions—regions of the antibody that have unique patterns of amino acids that can only bind to target antigens with a molecular sequence that provides complementary charges and noncovalent bonds. There are limitations to antibody specificity, however. Some antigens are so chemically similar that cross-reactivity occurs; in other words, antibodies raised against one antigen bind to a chemically similar but different antigen. Consider an antigen that consists of a single protein with multiple epitopes (Figure \(\PageIndex{1}\)). This single protein may stimulate the production of many different antibodies, some of which may bind to chemically identical epitopes on other proteins.
Cross-reactivity is more likely to occur between antibodies and antigens that have low affinity or avidity. Affinity, which can be determined experimentally, is a measure of the binding strength between an antibody's binding site and an epitope, whereas avidity is the total strength of all the interactions in an antibody-antigen complex (which may have more than one bonding site). Avidity is influenced by affinity as well as the structural arrangements of the epitope and the variable regions of the antibody. If an antibody has a high affinity/avidity for a specific antigen, it is less likely to cross-react with an antigen for which it has a lower affinity/avidity.
Exercise \(\PageIndex{1}\)
- What property makes antibodies useful for research and clinical diagnosis?
- What is cross-reactivity and why does it occur?
Producing Polyclonal Antibodies
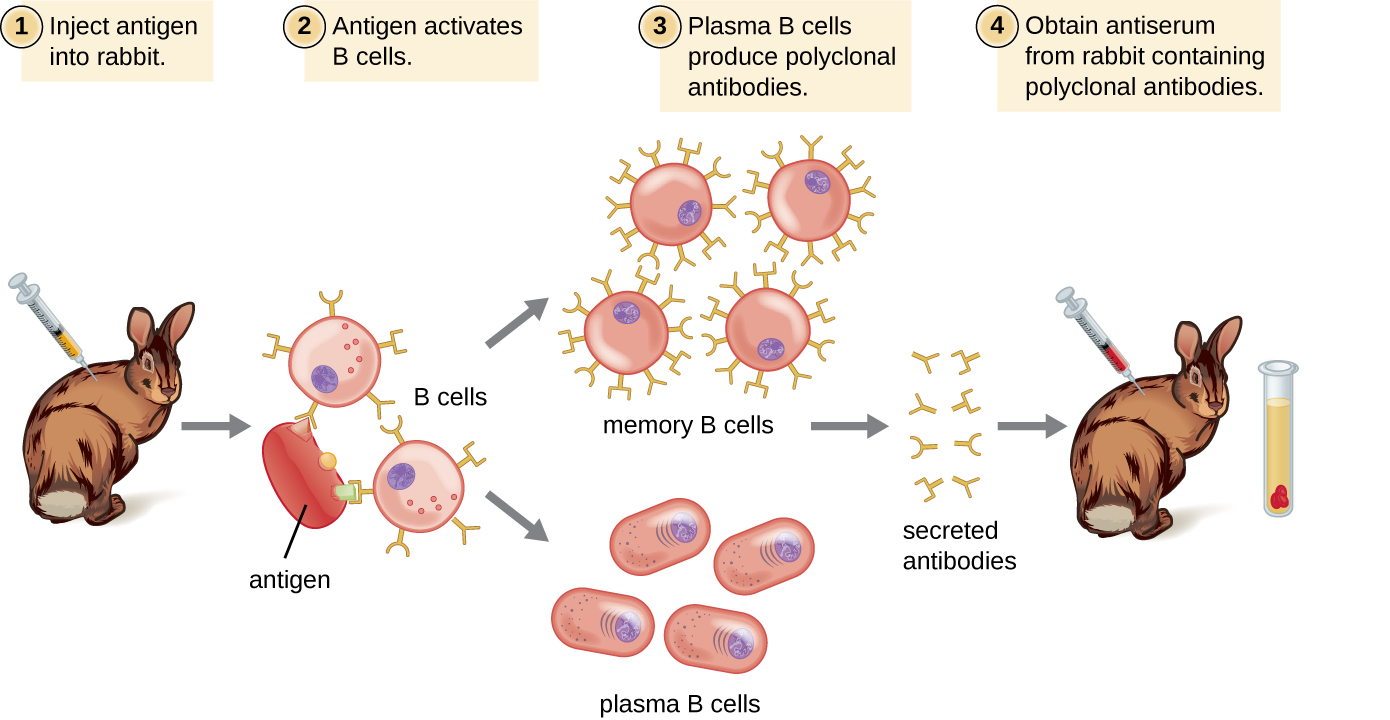
Antibodies used for research and diagnostic purposes are often obtained by injecting a lab animal such as a rabbit or a goat with a specific antigen. Within a few weeks, the animal’s immune system will produce high levels of antibodies specific for the antigen. These antibodies can be harvested in an antiserum, which is whole serum collected from an animal following exposure to an antigen. Because most antigens are complex structures with multiple epitopes, they result in the production of multiple antibodies in the lab animal. This so-called polyclonal antibody response is also typical of the response to infection by the human immune system. Antiserum drawn from an animal will thus contain antibodies from multiple clones of B cells, with each B cell responding to a specific epitope on the antigen (Figure \(\PageIndex{2}\)).
Lab animals are usually injected at least twice with antigen when being used to produce antiserum. The second injection will activate memory cells that make class IgG antibodies against the antigen. The memory cells also undergo affinity maturation, resulting in a pool of antibodies with higher average affinity. Affinity maturation occurs because of mutations in the immunoglobulin gene variable regions, resulting in B cells with slightly altered antigen-binding sites. On re-exposure to the antigen, those B cells capable of producing antibody with higher affinity antigen-binding sites will be stimulated to proliferate and produce more antibody than their lower-affinity peers. An adjuvant, which is a chemical that provokes a generalized activation of the immune system that stimulates greater antibody production, is often mixed with the antigen prior to injection.
Antiserum obtained from animals will not only contain antibodies against the antigen artificially introduced in the laboratory, but it will also contain antibodies to any other antigens to which the animal has been exposed during its lifetime. For this reason, antisera must first be “purified” to remove other antibodies before using the antibodies for research or diagnostic assays.
Clinical Uses of Polyclonal Antisera
Polyclonal antisera are used in many clinical tests that are designed to determine whether a patient is producing antibodies in response to a particular pathogen. While these tests are certainly powerful diagnostic tools, they have their limitations, because they are an indirect means of determining whether a particular pathogen is present. Tests based on a polyclonal response can sometimes lead to a false-positive result—in other words, a test that confirms the presence of an antigen that is, in fact, not present. Antibody-based tests can also result in a false-negative result, which occurs when the test fails to detect an antibody that is, in fact, present.
The accuracy of antibody tests can be described in terms of test sensitivity and test specificity. Test sensitivity is the probability of getting a positive test result when the patient is indeed infected. If a test has high sensitivity, the probability of a false negative is low. Test specificity, on the other hand, is the probability of getting a negative test result when the patient is not infected. If a test has high specificity, the probability of a false positive is low.
False positives often occur due to cross-reactivity, which can occur when epitopes from a different pathogen are similar to those found on the pathogen being tested for. For this reason, antibody-based tests are often used only as screening tests; if the results are positive, other confirmatory tests are used to make sure that the results were not a false positive.
For example, a blood sample from a patient suspected of having hepatitis C can be screened for the virus using antibodies that bind to antigens on hepatitis C virus. If the patient is indeed infected with hepatitis C virus, the antibodies will bind to the antigens, yielding a positive test result. If the patient is not infected with hepatitic C virus, the antibodies will generally not bind to anything and the test should be negative; however, a false positive may occur if the patient has been previously infected by any of a variety of pathogens that elicit antibodies that cross-react with the hepatitis C virus antigens. Antibody tests for hepatitis C have high sensitivity (a low probability of a false negative) but low specificity (a high probability of a false positive). Thus, patients who test positive must have a second, confirmatory test to rule out the possibility of a false positive. The confirmatory test is a more expensive and time-consuming test that directly tests for the presence of hepatitis C viral RNA in the blood. Only after the confirmatory test comes back positive can the patient be definitively diagnosed with a hepatitis C infection. Antibody-based tests can result in a false negative if, for any reason, the patient’s immune system has not produced detectable levels of antibodies. For some diseases, it may take several weeks following infection before the immune system produces enough antibodies to cross the detection threshold of the assay. In immunocompromised patients, the immune system may not be capable of producing a detectable level of antibodies.
Another limitation of using antibody production as an indicator of disease is that antibodies in the blood will persist long after the infection has been cleared. Depending on the type of infection, antibodies will be present for many months; sometimes, they may be present for the remainder of the patient’s life. Thus, a positive antibody-based test only means that the patient was infected at some point in time; it does not prove that the infection is active.
In addition to their role in diagnosis, polyclonal antisera can activate complement, detect the presence of bacteria in clinical and food industry settings, and perform a wide array of precipitation reactions that can detect and quantify serum proteins, viruses, or other antigens. However, with the many specificities of antibody present in a polyclonal antiserum, there is a significant likelihood that the antiserum will cross-react with antigens to which the individual was never exposed. Therefore, we must always account for the possibility of false-positive results when working with a polyclonal antiserum.
Exercise \(\PageIndex{2}\)
- What is a false positive and what are some reasons that false positives occur?
- What is a false negative and what are some reasons that false positives occur?
- If a patient tests negative on a highly sensitive test, what is the likelihood that the person is infected with the pathogen?
Producing Monoclonal Antibodies
Some types of assays require better antibody specificity and affinity than can be obtained using a polyclonal antiserum. To attain this high specificity, all of the antibodies must bind with high affinity to a single epitope. This high specificity can be provided by monoclonal antibodies (mAbs). Table \(\PageIndex{1}\) compares some of the important characteristics of monoclonal and polyclonal antibodies.
Unlike polyclonal antibodies, which are produced in live animals, monoclonal antibodies are produced in vitro using tissue-culture techniques. mAbs are produced by immunizing an animal, often a mouse, multiple times with a specific antigen. B cells from the spleen of the immunized animal are then removed. Since normal B cells are unable to proliferate forever, they are fused with immortal, cancerous B cells called myeloma cells, to yield hybridoma cells. All of the cells are then placed in a selective medium that allows only the hybridomas to grow; unfused myeloma cells cannot grow, and any unfused B cells die off. The hybridomas, which are capable of growing continuously in culture while producing antibodies, are then screened for the desired mAb. Those producing the desired mAb are grown in tissue culture; the culture medium is harvested periodically and mAbs are purified from the medium. This is a very expensive and time-consuming process. It may take weeks of culturing and many liters of media to provide enough mAbs for an experiment or to treat a single patient. Making mAbs is expensive (Figure \(\PageIndex{3}\)).
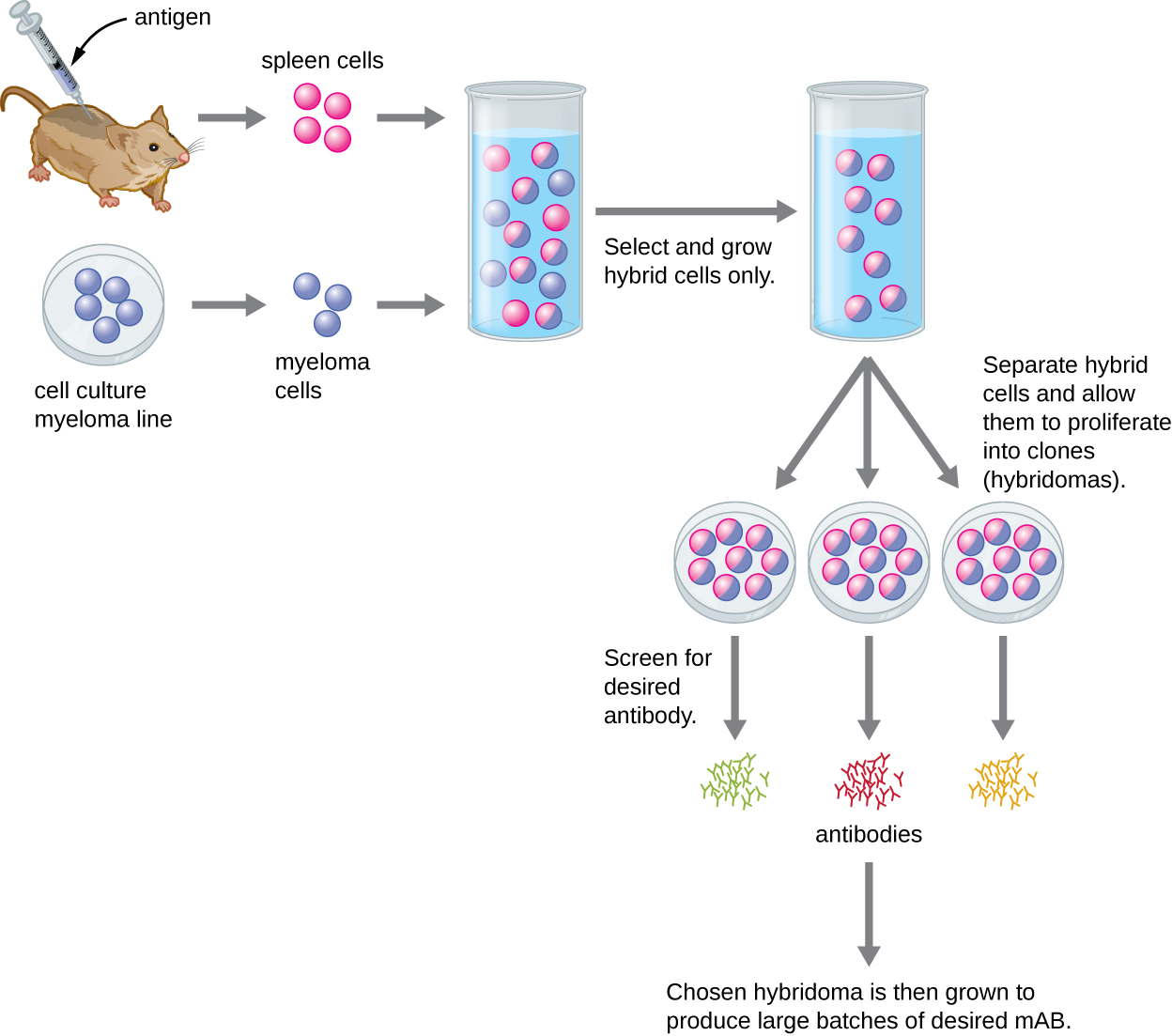
Table \(\PageIndex{1}\): Characteristics of Polyclonal and Monoclonal Antibodies
| Monoclonal Antibodies | Polyclonal Antibodies |
|---|---|
| Expensive production | Inexpensive production |
| Long production time | Rapid production |
| Large quantities of specific antibodies | Large quantities of nonspecific antibodies |
| Recognize a single epitope on an antigen | Recognize multiple epitopes on an antigen |
| Production is continuous and uniform once the hybridoma is made | Different batches vary in composition |
Clinical Uses of Monoclonal Antibodies
Since the most common methods for producing monoclonal antibodies use mouse cells, it is necessary to create humanized monoclonal antibodies for human clinical use. Mouse antibodies cannot be injected repeatedly into humans, because the immune system will recognize them as being foreign and will respond to them with neutralizing antibodies. This problem can be minimized by genetically engineering the antibody in the mouse B cell. The variable regions of the mouse light and heavy chain genes are ligated to human constant regions, and the chimeric gene is then transferred into a host cell. This allows production of a mAb that is mostly “human” with only the antigen-binding site being of mouse origin.
Humanized mAbs have been successfully used to treat cancer with minimal side effects. For example, the humanized monoclonal antibody drug Herceptin has been helpful for the treatment of some types of breast cancer. There have also been a few preliminary trials of humanized mAb for the treatment of infectious diseases, but none of these treatments are currently in use. In some cases, mAbs have proven too specific to treat infectious diseases, because they recognize some serovars of a pathogen but not others. Using a cocktail of multiple mAbs that target different strains of the pathogen can address this problem. However, the great cost associated with mAb production is another challenge that has prevented mAbs from becoming practical for use in treating microbial infections.1
One promising technology for inexpensive mAbs is the use of genetically engineered plants to produce antibodies (or plantibodies). This technology transforms plant cells into antibody factories rather than relying on tissue culture cells, which are expensive and technically demanding. In some cases, it may even be possible to deliver these antibodies by having patients eat the plants rather than by extracting and injecting the antibodies. For example, in 2013, a research group cloned antibody genes into plants that had the ability to neutralize an important toxin from bacteria that can cause severe gastrointestinal disease.2 Eating the plants could potentially deliver the antibodies directly to the toxin.
Exercise \(\PageIndex{3}\)
- How are humanized monoclonal antibodies produced?
- What does the “monoclonal” of monoclonal antibodies mean?
Using Monoclonal Antibodies to Combat Ebola
During the 2014–2015 Ebola outbreak in West Africa, a few Ebola-infected patients were treated with ZMapp, a drug that had been shown to be effective in trials done in rhesus macaques only a few months before.3 ZMapp is a combination of three mAbs produced by incorporating the antibody genes into tobacco plants using a viral vector. By using three mAbs, the drug is effective across multiple strains of the virus. Unfortunately, there was only enough ZMapp to treat a tiny number of patients.
While the current technology is not adequate for producing large quantities of ZMapp, it does show that plantibodies—plant-produced mAbs—are feasible for clinical use, potentially cost effective, and worth further development. The last several years have seen an explosion in the number of new mAb-based drugs for the treatment of cancer and infectious diseases; however, the widespread use of such drugs is currently inhibited by their exorbitant cost, especially in underdeveloped parts of the world, where a single dose might cost more than the patient’s lifetime income. Developing methods for cloning antibody genes into plants could reduce costs dramatically.
Use of Antibodies in Testing and Identification
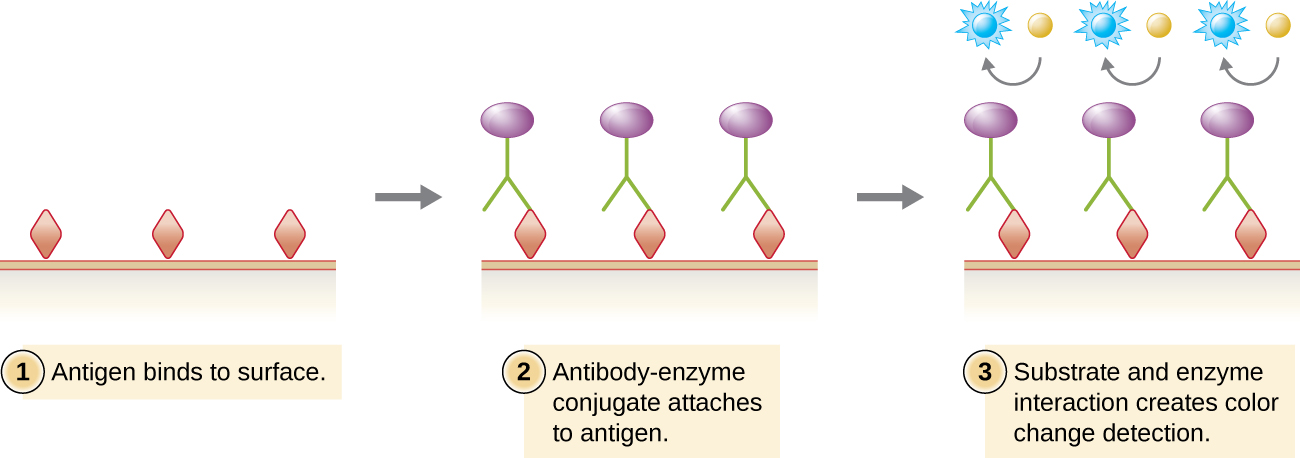
Enzyme immunoassays (EIAs) use antibodies to detect the presence of antigens. However, the assays are conducted in microtiter plates or in vivo. There are many different types of EIAs, but they all involve an antibody molecule whose constant region binds an enzyme, leaving the variable region free to bind its specific antigen. The addition of a substrate for the enzyme allows the antigen to be visualized or quantified (Figure \(\PageIndex{4}\)).
In EIAs, the substrate for the enzyme is most often a chromogen, a colorless molecule that is converted into a colored end product. The most widely used enzymes are alkaline phosphatase and horseradish peroxidase for which appropriate substrates are readily available. In some EIAs, the substrate is a fluorogen, a nonfluorescent molecule that the enzyme converts into a fluorescent form. EIAs that utilize a fluorogen are called fluorescent enzyme immunoassays (FEIAs). Fluorescence can be detected by either a fluorescence microscope or a spectrophotometer.
The MMR Titer
The MMR vaccine is a combination vaccine that provides protection against measles, mumps, and rubella (German measles). Most people receive the MMR vaccine as children and thus have antibodies against these diseases. However, for various reasons, even vaccinated individuals may become susceptible to these diseases again later in life. For example, some children may receive only one round of the MMR vaccine instead of the recommended two. In addition, the titer of protective antibodies in an individual’s body may begin to decline with age or as the result of some medical conditions.
To determine whether the titer of antibody in an individual’s bloodstream is sufficient to provide protection, an MMR titer test can be performed. The test is a simple immunoassay that can be done quickly with a blood sample. The results of the test will indicate whether the individual still has immunity or needs another dose of the MMR vaccine.
Submitting to an MMR titer is often a pre-employment requirement for healthcare workers, especially those who will frequently be in contact with young children or immunocompromised patients. Were a healthcare worker to become infected with measles, mumps, or rubella, the individual could easily pass these diseases on to susceptible patients, leading to an outbreak. Depending on the results of the MMR titer, healthcare workers might need to be revaccinated prior to beginning work.
Immunostaining
One powerful use of EIA is immunostaining, in which antibody-enzyme conjugates enhance microscopy. Immunohistochemistry (IHC) is used for examining whole tissues. As seen in Figure \(\PageIndex{5}\), a section of tissue can be stained to visualize the various cell types. In this example, a mAb against CD8 was used to stain CD8 cells in a section of tonsil tissue. It is now possible to count the number of CD8 cells, determine their relative numbers versus the other cell types present, and determine the location of these cells within this tissue. Such data would be useful for studying diseases such as AIDS, in which the normal function of CD8 cells is crucial for slowing disease progression.
Immunocytochemistry (ICC) is another valuable form of immunostaining. While similar to IHC, in ICC, extracellular matrix material is stripped away, and the cell membrane is etched with alcohol to make it permeable to antibodies. This allows antibodies to pass through the cell membrane and bind to specific targets inside the cell. Organelles, cytoskeletal components, and other intracellular structures can be visualized in this way. While some ICC techniques use EIA, the enzyme can be replaced with a fluorescent molecule, making it a fluorescent immunoassay.

Exercise \(\PageIndex{4}\)
- What is the difference between immunohistochemistry and immunocytochemistry?
- What must be true of the product of the enzymatic reaction used in immunohistochemistry?
Enzyme-linked Immunosorbent Assays (ELISAs)
The enzyme-linked immunosorbent assays (ELISAs) are widely used EIAs. In the direct ELISA, antigens are immobilized in the well of a microtiter plate. An antibody that is specific for a particular antigen and is conjugated to an enzyme is added to each well. If the antigen is present, then the antibody will bind. After washing to remove any unbound antibodies, a colorless substrate (chromogen) is added. The presence of the enzyme converts the substrate into a colored end product (Figure \(\PageIndex{4}\)). While this technique is faster because it only requires the use of one antibody, it has the disadvantage that the signal from a direct ELISA is lower (lower sensitivity).
In a sandwich ELISA, the goal is to use antibodies to precisely quantify specific antigen present in a solution, such as antigen from a pathogen, a serum protein, or a hormone from the blood or urine to list just a few examples. The first step of a sandwich ELISA is to add the primary antibody to all the wells of a microtiter plate (Figure \(\PageIndex{6}\)). The antibody sticks to the plastic by hydrophobic interactions. After an appropriate incubation time, any unbound antibody is washed away. Comparable washes are used between each of the subsequent steps to ensure that only specifically bound molecules remain attached to the plate. A blocking protein is then added (e.g., albumin or the milk protein casein) to bind the remaining nonspecific protein-binding sites in the well. Some of the wells will receive known amounts of antigen to allow the construction of a standard curve, and unknown antigen solutions are added to the other wells. The primary antibody captures the antigen and, following a wash, the secondary antibody is added, which is a polyclonal antibody that is conjugated to an enzyme. After a final wash, a colorless substrate (chromogen) is added, and the enzyme converts it into a colored end product. The color intensity of the sample caused by the end product is measured with a spectrophotometer. The amount of color produced (measured as absorbance) is directly proportional to the amount of enzyme, which in turn is directly proportional to the captured antigen. ELISAs are extremely sensitive, allowing antigen to be quantified in the nanogram (10–9 g) per mL range.
In an indirect ELISA, we quantify antigen-specific antibody rather than antigen. We can use indirect ELISA to detect antibodies against many types of pathogens, including Borrelia burgdorferi (Lyme disease) and HIV. There are three important differences between indirect and direct ELISAs as shown in Figure \(\PageIndex{7}\). Rather than using antibody to capture antigen, the indirect ELISA starts with attaching known antigen (e.g., peptides from HIV) to the bottom of the microtiter plate wells. After blocking the unbound sites on the plate, patient serum is added; if antibodies are present (primary antibody), they will bind the antigen. After washing away any unbound proteins, the secondary antibody with its conjugated enzyme is directed against the primary antibody (e.g., antihuman immunoglobulin). The secondary antibody allows us to quantify how much antigen-specific antibody is present in the patient’s serum by the intensity of the color produced from the conjugated enzyme-chromogen reaction.
As with several other tests for antibodies discussed in this chapter, there is always concern about cross-reactivity with antibodies directed against some other antigen, which can lead to false-positive results. Thus, we cannot definitively diagnose an HIV infection (or any other type of infection) based on a single indirect ELISA assay. We must confirm any suspected positive test, which is most often done using either an immunoblot that actually identifies the presence of specific peptides from the pathogen or a test to identify the nucleic acids associated with the pathogen, such as reverse transcriptase PCR (RT-PCR) or a nucleic acid antigen test.
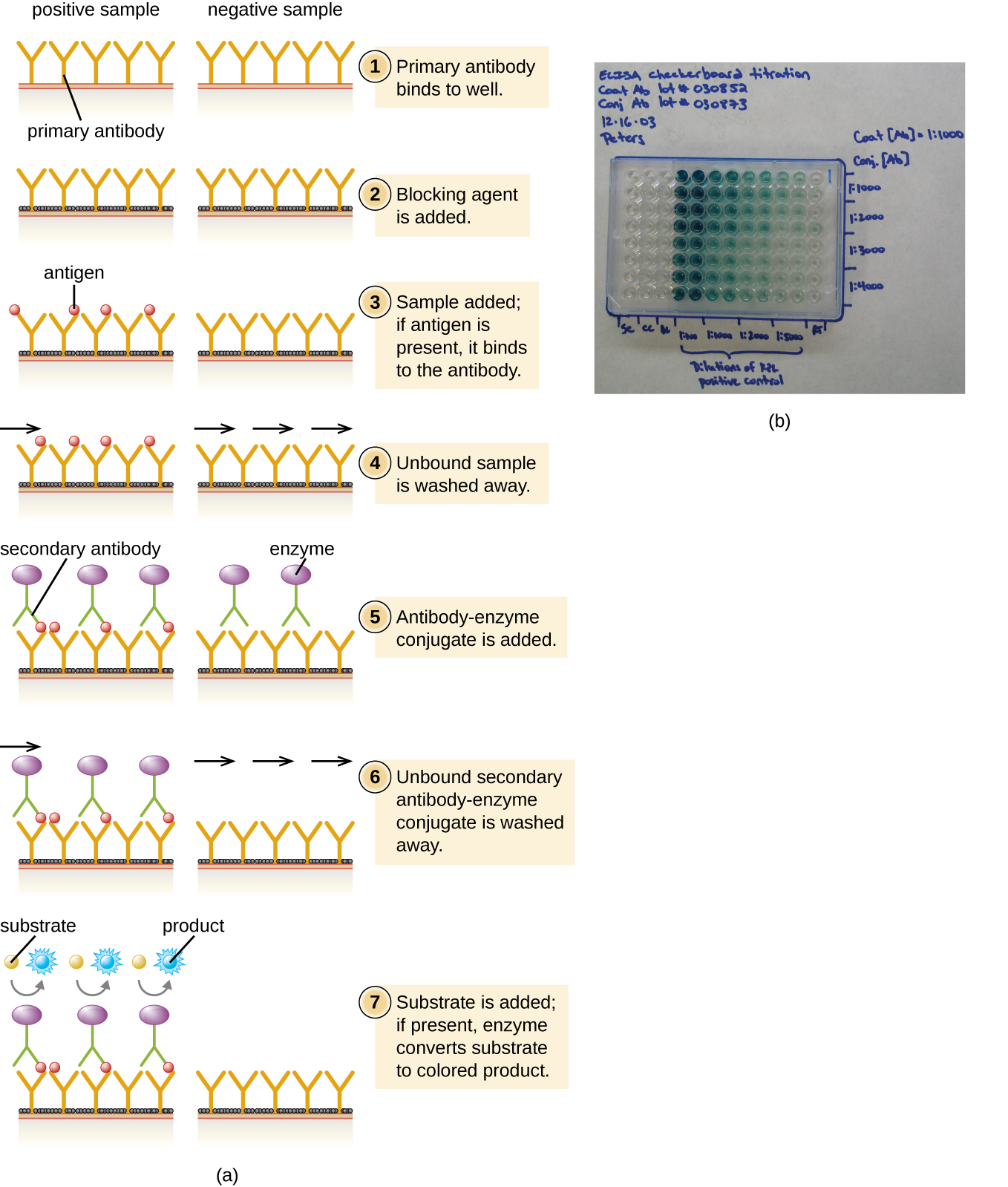
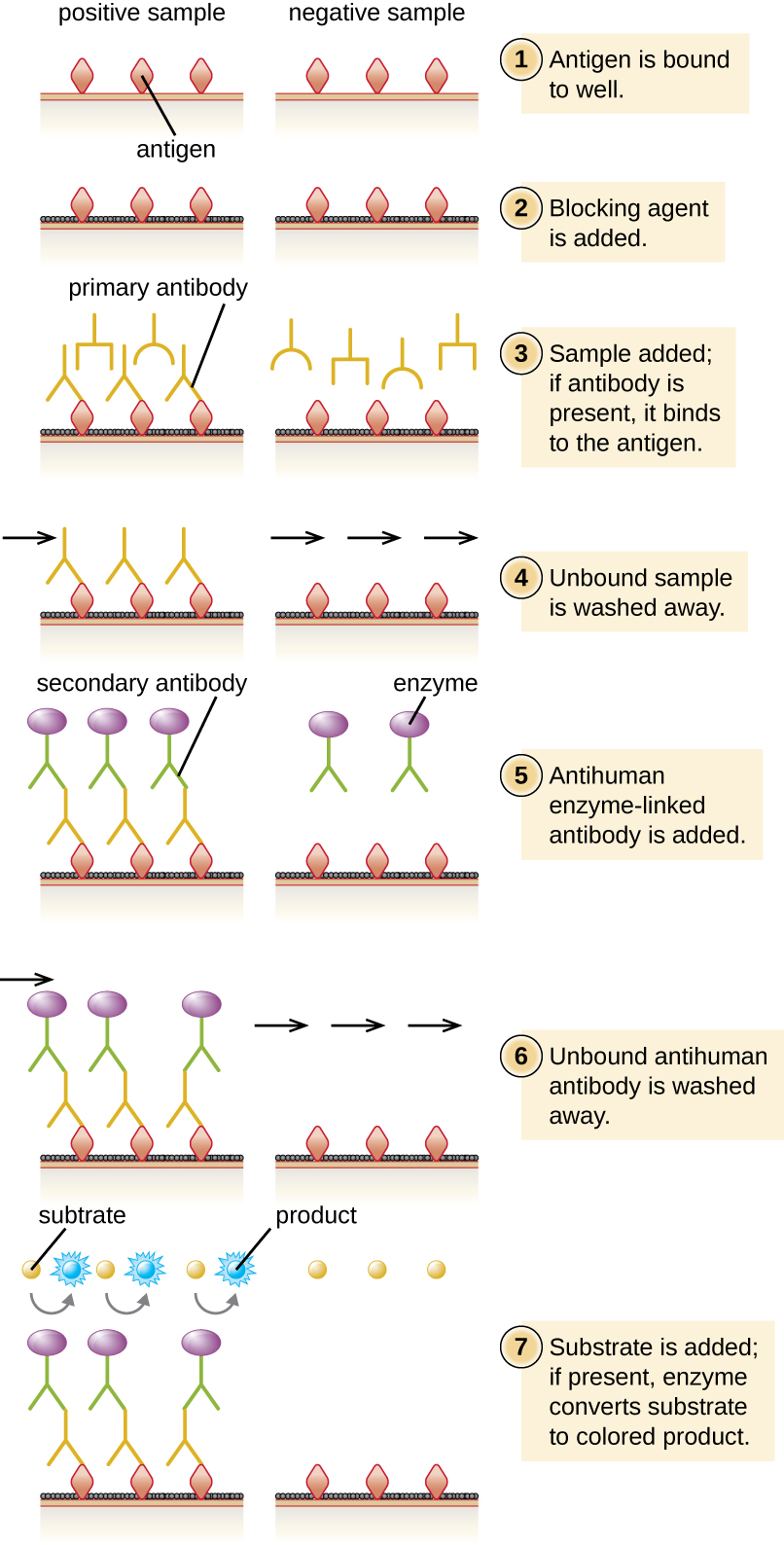
Exercise \(\PageIndex{5}\)
- What is the purpose of the secondary antibody in a direct ELISA?
- What do the direct and indirect ELISAs quantify?
Key Concepts and Summary
- Antibodies bind with high specificity to antigens used to challenge the immune system, but they may also show cross-reactivity by binding to other antigens that share chemical properties with the original antigen.
- Injection of an antigen into an animal will result in a polyclonal antibody response in which different antibodies are produced that react with the various epitopes on the antigen.
- Polyclonal antisera are useful for some types of laboratory assays, but other assays require more specificity. Diagnostic tests that use polyclonal antisera are typically only used for screening because of the possibility of false-positive and false-negative results.
- Monoclonal antibodies provide higher specificity than polyclonal antisera because they bind to a single epitope and usually have high affinity.
- Monoclonal antibodies are typically produced by culturing antibody-secreting hybridomas derived from mice. mAbs are currently used to treat cancer, but their exorbitant cost has prevented them from being used more widely to treat infectious diseases. Still, their potential for laboratory and clinical use is driving the development of new, cost-effective solutions such as plantibodies.
- Enzyme immunoassays (EIA) are used to visualize and quantify antigens. They use an antibody conjugated to an enzyme to bind the antigen, and the enzyme converts a substrate into an observable end product. The substrate may be either a chromogen or a fluorogen.
- Immunostaining is an EIA technique for visualizing cells in a tissue (immunohistochemistry) or examining intracellular structures (immunocytochemistry).
- Direct ELISA is used to quantify an antigen in solution. The primary antibody captures the antigen, and the secondary antibody delivers an enzyme. Production of end product from the chromogenic substrate is directly proportional to the amount of captured antigen.
- Indirect ELISA is used to detect antibodies in patient serum by attaching antigen to the well of a microtiter plate, allowing the patient (primary) antibody to bind the antigen and an enzyme-conjugated secondary antibody to detect the primary antibody.
Footnotes
- Saylor, Carolyn, Ekaterina Dadachova and Arturo Casadevall, “Monoclonal Antibody-Based Therapies for Microbial Diseases,” Vaccine 27 (2009): G38-G46.
- Nakanishi, Katsuhiro et al., “Production of Hybrid-IgG/IgA Plantibodies with Neutralizing Activity against Shiga Toxin 1,” PloS One 8, no. 11 (2013): e80712.
- Qiu, Xiangguo et al., “Reversion of Advanced Ebola Virus Disease in Nonhuman Primates with ZMapp,” Nature 514 (2014): 47–53.
Contributors and Attributions
Nina Parker, (Shenandoah University), Mark Schneegurt (Wichita State University), Anh-Hue Thi Tu (Georgia Southwestern State University), Philip Lister (Central New Mexico Community College), and Brian M. Forster (Saint Joseph’s University) with many contributing authors. Original content via Openstax (CC BY 4.0; Access for free at https://openstax.org/books/microbiology/pages/1-introduction)


