3: Water and Buffer
- Page ID
- 158568
\( \newcommand{\vecs}[1]{\overset { \scriptstyle \rightharpoonup} {\mathbf{#1}} } \)
\( \newcommand{\vecd}[1]{\overset{-\!-\!\rightharpoonup}{\vphantom{a}\smash {#1}}} \)
\( \newcommand{\dsum}{\displaystyle\sum\limits} \)
\( \newcommand{\dint}{\displaystyle\int\limits} \)
\( \newcommand{\dlim}{\displaystyle\lim\limits} \)
\( \newcommand{\id}{\mathrm{id}}\) \( \newcommand{\Span}{\mathrm{span}}\)
( \newcommand{\kernel}{\mathrm{null}\,}\) \( \newcommand{\range}{\mathrm{range}\,}\)
\( \newcommand{\RealPart}{\mathrm{Re}}\) \( \newcommand{\ImaginaryPart}{\mathrm{Im}}\)
\( \newcommand{\Argument}{\mathrm{Arg}}\) \( \newcommand{\norm}[1]{\| #1 \|}\)
\( \newcommand{\inner}[2]{\langle #1, #2 \rangle}\)
\( \newcommand{\Span}{\mathrm{span}}\)
\( \newcommand{\id}{\mathrm{id}}\)
\( \newcommand{\Span}{\mathrm{span}}\)
\( \newcommand{\kernel}{\mathrm{null}\,}\)
\( \newcommand{\range}{\mathrm{range}\,}\)
\( \newcommand{\RealPart}{\mathrm{Re}}\)
\( \newcommand{\ImaginaryPart}{\mathrm{Im}}\)
\( \newcommand{\Argument}{\mathrm{Arg}}\)
\( \newcommand{\norm}[1]{\| #1 \|}\)
\( \newcommand{\inner}[2]{\langle #1, #2 \rangle}\)
\( \newcommand{\Span}{\mathrm{span}}\) \( \newcommand{\AA}{\unicode[.8,0]{x212B}}\)
\( \newcommand{\vectorA}[1]{\vec{#1}} % arrow\)
\( \newcommand{\vectorAt}[1]{\vec{\text{#1}}} % arrow\)
\( \newcommand{\vectorB}[1]{\overset { \scriptstyle \rightharpoonup} {\mathbf{#1}} } \)
\( \newcommand{\vectorC}[1]{\textbf{#1}} \)
\( \newcommand{\vectorD}[1]{\overrightarrow{#1}} \)
\( \newcommand{\vectorDt}[1]{\overrightarrow{\text{#1}}} \)
\( \newcommand{\vectE}[1]{\overset{-\!-\!\rightharpoonup}{\vphantom{a}\smash{\mathbf {#1}}}} \)
\( \newcommand{\vecs}[1]{\overset { \scriptstyle \rightharpoonup} {\mathbf{#1}} } \)
\(\newcommand{\longvect}{\overrightarrow}\)
\( \newcommand{\vecd}[1]{\overset{-\!-\!\rightharpoonup}{\vphantom{a}\smash {#1}}} \)
\(\newcommand{\avec}{\mathbf a}\) \(\newcommand{\bvec}{\mathbf b}\) \(\newcommand{\cvec}{\mathbf c}\) \(\newcommand{\dvec}{\mathbf d}\) \(\newcommand{\dtil}{\widetilde{\mathbf d}}\) \(\newcommand{\evec}{\mathbf e}\) \(\newcommand{\fvec}{\mathbf f}\) \(\newcommand{\nvec}{\mathbf n}\) \(\newcommand{\pvec}{\mathbf p}\) \(\newcommand{\qvec}{\mathbf q}\) \(\newcommand{\svec}{\mathbf s}\) \(\newcommand{\tvec}{\mathbf t}\) \(\newcommand{\uvec}{\mathbf u}\) \(\newcommand{\vvec}{\mathbf v}\) \(\newcommand{\wvec}{\mathbf w}\) \(\newcommand{\xvec}{\mathbf x}\) \(\newcommand{\yvec}{\mathbf y}\) \(\newcommand{\zvec}{\mathbf z}\) \(\newcommand{\rvec}{\mathbf r}\) \(\newcommand{\mvec}{\mathbf m}\) \(\newcommand{\zerovec}{\mathbf 0}\) \(\newcommand{\onevec}{\mathbf 1}\) \(\newcommand{\real}{\mathbb R}\) \(\newcommand{\twovec}[2]{\left[\begin{array}{r}#1 \\ #2 \end{array}\right]}\) \(\newcommand{\ctwovec}[2]{\left[\begin{array}{c}#1 \\ #2 \end{array}\right]}\) \(\newcommand{\threevec}[3]{\left[\begin{array}{r}#1 \\ #2 \\ #3 \end{array}\right]}\) \(\newcommand{\cthreevec}[3]{\left[\begin{array}{c}#1 \\ #2 \\ #3 \end{array}\right]}\) \(\newcommand{\fourvec}[4]{\left[\begin{array}{r}#1 \\ #2 \\ #3 \\ #4 \end{array}\right]}\) \(\newcommand{\cfourvec}[4]{\left[\begin{array}{c}#1 \\ #2 \\ #3 \\ #4 \end{array}\right]}\) \(\newcommand{\fivevec}[5]{\left[\begin{array}{r}#1 \\ #2 \\ #3 \\ #4 \\ #5 \\ \end{array}\right]}\) \(\newcommand{\cfivevec}[5]{\left[\begin{array}{c}#1 \\ #2 \\ #3 \\ #4 \\ #5 \\ \end{array}\right]}\) \(\newcommand{\mattwo}[4]{\left[\begin{array}{rr}#1 \amp #2 \\ #3 \amp #4 \\ \end{array}\right]}\) \(\newcommand{\laspan}[1]{\text{Span}\{#1\}}\) \(\newcommand{\bcal}{\cal B}\) \(\newcommand{\ccal}{\cal C}\) \(\newcommand{\scal}{\cal S}\) \(\newcommand{\wcal}{\cal W}\) \(\newcommand{\ecal}{\cal E}\) \(\newcommand{\coords}[2]{\left\{#1\right\}_{#2}}\) \(\newcommand{\gray}[1]{\color{gray}{#1}}\) \(\newcommand{\lgray}[1]{\color{lightgray}{#1}}\) \(\newcommand{\rank}{\operatorname{rank}}\) \(\newcommand{\row}{\text{Row}}\) \(\newcommand{\col}{\text{Col}}\) \(\renewcommand{\row}{\text{Row}}\) \(\newcommand{\nul}{\text{Nul}}\) \(\newcommand{\var}{\text{Var}}\) \(\newcommand{\corr}{\text{corr}}\) \(\newcommand{\len}[1]{\left|#1\right|}\) \(\newcommand{\bbar}{\overline{\bvec}}\) \(\newcommand{\bhat}{\widehat{\bvec}}\) \(\newcommand{\bperp}{\bvec^\perp}\) \(\newcommand{\xhat}{\widehat{\xvec}}\) \(\newcommand{\vhat}{\widehat{\vvec}}\) \(\newcommand{\uhat}{\widehat{\uvec}}\) \(\newcommand{\what}{\widehat{\wvec}}\) \(\newcommand{\Sighat}{\widehat{\Sigma}}\) \(\newcommand{\lt}{<}\) \(\newcommand{\gt}{>}\) \(\newcommand{\amp}{&}\) \(\definecolor{fillinmathshade}{gray}{0.9}\)- Define pH and explain how it is measured.
- Understand the logarithmic nature of the pH scale.
- Explain how pH influences biological systems and biochemical reactions.
- Describe the role and mechanism of buffer systems in maintaining pH homeostasis.
- Differentiate between strong and weak acids/bases and their dissociation behavior.
- Identify major biological buffer systems and their importance in the human body.
- pH: A measure of hydrogen ion (H⁺) concentration in a solution.
- pKa: The pH at which a weak acid is half dissociated; indicates acid strength.
- Buffer: A solution that resists pH changes by neutralizing added acids or bases.
- Titration Point: The stage in a titration where the amount of added acid or base fully neutralizes the solution.
- Buffer Zone: The range of pH values where a buffer effectively stabilizes pH.
- Strong Acid/Base: A substance that fully dissociates in water, drastically altering pH.
- Weak Acid/Base: A substance that only partially dissociates, helping to maintain pH balance.
Water: The Solvent of Life
Water is often referred to as the "solvent of life" because of its unparalleled ability to dissolve a wide range of substances essential for biological functions. This property makes water an indispensable component of cells, tissues, and bodily fluids. In biological systems, water serves as a medium for chemical reactions, facilitates nutrient transport, and aids in the removal of waste products.
Hydrogen bonding is a crucial interaction in water and many biological molecules. A hydrogen bond forms when a hydrogen atom covalently bonded to an electronegative atom (such as oxygen or nitrogen) experiences an attraction to another electronegative atom. These bonds are weak individually but collectively play a significant role in determining the properties of water and biological macromolecules. For example, the hydrogen bonds between water molecules give it a high surface tension, allowing insects like water striders to walk on water. In DNA, hydrogen bonds hold the two strands of the double helix together, ensuring genetic stability. The three-dimensional shape of macromolecules like proteins and nucleic acids is heavily influenced by hydrogen bonding and interactions with water. For example, the secondary and tertiary structures of proteins depend on hydrogen bonds, which stabilize alpha-helices and beta-sheets. In DNA, hydrogen bonds between base pairs maintain the double-helical structure necessary for genetic function.
Water is a polar molecule, meaning it has a partial positive charge on the hydrogen atoms and a partial negative charge on the oxygen atom. This polarity allows water molecules to attract one another through hydrogen bonds (Figure 3.1). Additionally, water can interact with other polar substances, enabling it to dissolve ionic compounds like sodium chloride (table salt) and hydrophilic molecules such as sugars and amino acids. A polar covalent bond occurs when electrons are shared unequally between atoms, resulting in partial charges. Water molecules exhibit this property, allowing them to engage in interactions with other polar substances. For instance, in protein folding, the interactions between polar groups help establish the molecule's final three-dimensional shape, which is crucial for function (Figure 3.2). Similarly, in cell membranes, the arrangement of phospholipids—where the polar heads interact with water while nonpolar tails remain shielded—helps maintain structural integrity.

Figure 3.1: Water molecules are polar covalently bound and are interacting with other water molecules due to their partial charges.

Figure 3.2: Water molecules interact with protein structures, helping the fold for the final three-dimensional structure.
Water's polarity also leads to hydrophobic interactions, where nonpolar molecules (such as lipids) do not dissolve in water. Instead, they cluster together to minimize their exposure to water. This property is crucial in biological systems:
- Cell Membranes: Phospholipid bilayers form because the hydrophobic tails face inward, avoiding water, while the hydrophilic heads interact with the aqueous environment.
- Fat Metabolism: Nonpolar fats do not mix with water and require emulsifiers (like bile salts) for digestion. If you get tar or grease on your hands, water alone will not wash it away because these substances are nonpolar. However, vegetable oil can dissolve the tar, as it is also nonpolar, allowing it to be wiped away.
Oxygen and nitrogen are highly electronegative elements that frequently participate in hydrogen bonding. These elements are found in many biological molecules such as proteins, nucleic acids, and carbohydrates. Their presence in these molecules allows for critical interactions, influencing molecular structure, stability, and function. For example, the presence of oxygen in carbohydrates allows them to readily dissolve in water, making them easily transportable in the bloodstream. Water plays an essential role in many biochemical reactions. It is involved in both the reactants and products of numerous metabolic processes. Two key reactions include:
- Hydrolysis: Water is used to break down macromolecules. For example, during digestion, polysaccharides are broken into simple sugars by adding water.
- Dehydration Synthesis: Water is removed to link smaller molecules into larger macromolecules. For instance, amino acids bond to form proteins by releasing water molecules.
Water has an exceptionally high heat capacity due to the energy required to break and form hydrogen bonds. This property makes water effective in regulating temperature in biological systems. Water helps buffer temperature changes, preventing sudden fluctuations that could harm cellular processes. Large bodies of water absorb heat during the day and release it slowly at night, stabilizing local climates.
Cohesion refers to water molecules sticking together due to hydrogen bonding, while adhesion refers to water molecules sticking to other surfaces (Figure 3.3). These properties are vital in biological processes. In Plants, Cohesion and adhesion enable capillary action, allowing water to travel from roots to leaves through xylem vessels. Without these forces, plants would not be able to transport water against gravity, leading to dehydration and death. In Animals, Surface tension (a result of cohesion) helps organisms like insects walk on water. Additionally, cohesion is essential in blood circulation, where it assists in maintaining blood pressure and the movement of fluids in capillaries. In Biochemistry, Adhesion helps water interact with biomolecules, aiding in solubility and transport. For example, adhesion allows water to form hydration shells around ions and proteins, facilitating biochemical reactions inside cells. In Drug Studies, Many pharmaceutical drugs rely on water solubility for effective absorption in the body. Adhesion allows drugs to dissolve in biological fluids, ensuring proper distribution and efficacy. Cohesion helps in the consistency of solutions used for injections or intravenous fluids.
Overall, Water's unique properties, including its solvent capabilities, hydrogen bonding, high heat capacity, and cohesive and adhesive nature, make it indispensable for life. Whether facilitating biochemical reactions, maintaining cellular structures, or stabilizing ecosystems, water plays a fundamental role in sustaining biological functions across all forms of life.

Figure 3.3: Cohesion vs adhesion. FeltyRacketeer6. CC BY-SA 4.0
-----------------------------------------------------
Understanding pH and Its Measurement
pH is a measure of how acidic or basic a solution is, based on the concentration of hydrogen ions (H⁺) (Figure 3.4). It is calculated using the logarithmic scale pH = -log10[H⁺], meaning that a change of one unit represents a ten-fold difference in hydrogen ion concentration. For example:
- A solution with a pH of 3 has 100 times more H⁺ ions than a solution with a pH of 5.
- A shift from pH 6 to pH 7 means the solution has 10 times fewer H⁺ ions, becoming less acidic.
- A shift from pH 10 to pH 13 indicates a 1,000-fold decrease in H⁺ ions, making the solution significantly more basic.

Figure 3.4: Image of pH scale diagram generated by ChatGPT.

Figure 3.5: Image of strong vs weak acid making buffers.
Most life forms thrive within a narrow pH range. For instance, human blood maintains a pH around 7.35-7.45. Deviations beyond this range can be harmful, as biochemical reactions depend on pH stability. Like temperature, pH extremes can lead to protein denaturation—a process where proteins lose their three-dimensional structure and functionality. At extreme acidic or basic pH values, the hydrogen or hydroxide ions interfere with the hydrogen bonds and ionic interactions that maintain the structure of proteins. This disruption unfolds proteins, rendering them inactive and potentially leading to cellular damage or death. For example:
- Stomach enzymes, such as pepsin, function optimally at very low pH (~2) but would denature at neutral pH.
- Blood proteins, such as hemoglobin, require a strict pH range (~7.4) to function correctly; even small deviations can impair oxygen transport.
To prevent drastic pH changes, biological systems rely on buffers, which stabilize pH by absorbing excess hydrogen (H⁺) or hydroxide (OH⁻) ions. Buffers help maintain homeostasis, ensuring that enzymatic and biochemical reactions occur under optimal conditions. One key property of a buffer is its pKa, which indicates the strength of an acid or base. The pKa value is the pH at which half of the acid or base molecules are dissociated into their ionized form.
-
Strong acids (pKa <3) fully dissociate in water and drastically lower pH (e.g., HCl).
-
Weak acids (pKa 3-7) partially dissociate, creating buffer systems (e.g., acetic acid, pKa ~4.76).
-
Weak bases (pKa 7-11) only partially accept protons (e.g., ammonia, NH3, pKa ~9.3).
-
Strong bases (pKa >11) readily accept protons and sharply increase pH (e.g., NaOH).
A buffer consists of a weak acid and its conjugate base (or a weak base and its conjugate acid) (Figure 3.5). It resists pH changes by either donating or accepting protons (H⁺) in response to fluctuations in the environment. For example, when Acetic acid (CH₃COOH) partially ionizes in water: CH₃COOH ⇌ CH₃COO⁻ + H⁺. If an acid (H⁺) is added, the acetate ion (CH₃COO⁻) binds to the excess protons, forming more acetic acid and preventing a pH drop. If a base (OH⁻) is added, acetic acid donates protons to neutralize it, preventing a pH increase. This balance allows buffer solutions to resist pH changes when small amounts of acids or bases are introduced.
The buffer zone refers to the range of pH values over which a buffer solution can effectively maintain pH stability (Figure 3.6 and 3.7). This range is typically ±1 pH unit around the pKa of the buffer system. Within this zone, the weak acid and its conjugate base are in equilibrium, allowing them to neutralize small amounts of added acid (H⁺) or base (OH⁻) without significantly altering the overall pH. The titration point (also known as the equivalence point or tilting point) occurs when the amount of added strong acid or base completely reacts with the buffer components, surpassing the buffer zone and leading to a rapid drop in pH or a drastic increase in pH. At this stage, the buffering capacity is overwhelmed, and the system can no longer resist shifts in pH.
Luckily, our body employs multiple buffer systems to maintain different pH environments in various biological fluids. Each buffer system works within its optimal buffer zone to ensure stability in its respective environment. This redundancy is crucial because different physiological processes generate acids and bases at various rates. Proteins, such as hemoglobin and albumin, have amino acid side chains that can accept or donate protons, stabilizing pH. The histidine residue in hemoglobin, for example, plays a crucial role in buffering blood pH. By having multiple buffer systems in place, the body can efficiently regulate pH across multiple biological compartments. Each system operates within a specific buffer zone to regulate pH in different compartments:
- Blood (pH 7.35-7.45) – The bicarbonate buffer system (H₂CO₃/HCO₃⁻) maintains blood pH by balancing CO₂ exhalation with carbonic acid formation. This system works efficiently because CO₂ can be expelled through respiration, shifting the equilibrium as needed. The bicarbonate buffer system is the primary regulator of blood pH, maintaining it around 7.4. It operates based on the following equilibrium: H₂CO₃ ⇌ HCO₃⁻ + H⁺. If pH drops (more acidic), bicarbonate (HCO₃⁻) absorbs H⁺, shifting the reaction toward carbonic acid (H₂CO₃), which then decomposes into CO₂ and H₂O, expelled through respiration. If pH rises (more basic), carbonic acid releases H⁺ to balance the system.
- Intracellular fluids (pH ~6.8-7.2) – The phosphate buffer system (H₂PO₄⁻/HPO₄²⁻) stabilizes pH inside cells where metabolic reactions produce acids. The phosphate buffer system is crucial for intracellular pH regulation. It works via the equilibrium: H₂PO₄⁻ ⇌ HPO₄²⁻ + H⁺. This buffer is essential in urine and cellular fluids, helping to maintain pH stability in metabolic processes.
- Urine (pH ~4.5-8.0) – The phosphate and ammonia buffer systems help excrete excess H⁺, adjusting urine pH depending on dietary and metabolic conditions.
- Stomach (pH ~1.5-3.5) – The gastric environment relies on hydrochloric acid (HCl) for digestion, but mucosal bicarbonate buffering protects the stomach lining from damage.
Overall, understanding pH and buffer systems is essential in biology, medicine, and biotechnology. Buffers prevent harmful pH fluctuations, ensuring biochemical reactions occur efficiently. Whether in blood regulation, enzyme function, or pharmaceutical applications, buffer systems are indispensable for maintaining life’s delicate balance.
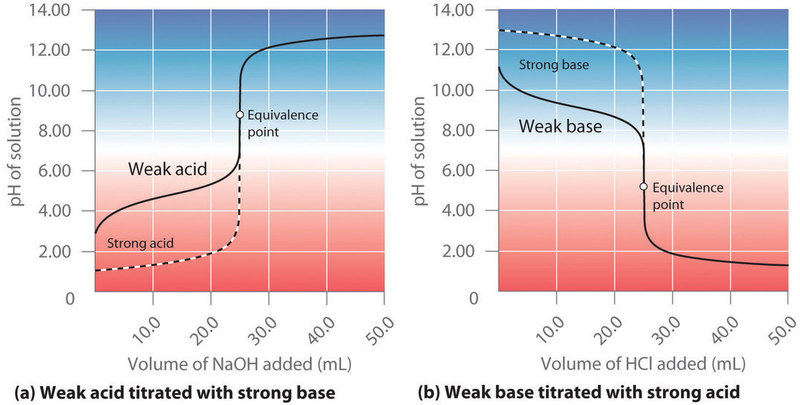
Figure 3.6: Comparison of the buffer zone of a weak base to that of a weak acid. Shared under a CC BY-NC-SA 4.0 license.
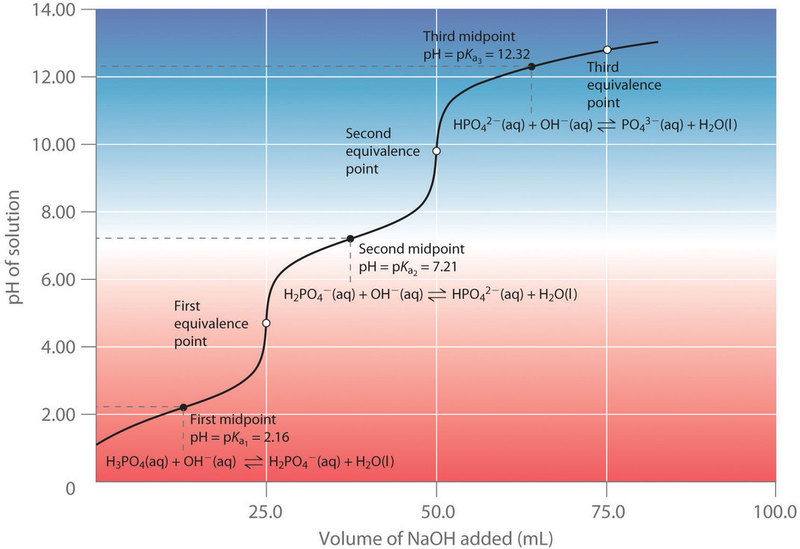
Figure 3.7: Polyprotic titration curve for phosphoric acid, showing multiple buffer zones. Shared under a CC BY-NC-SA 4.0 license.
- How does the logarithmic nature of the pH scale impact biological reactions?
- Why is maintaining a stable pH important for enzymes and proteins?
- What are the differences between strong and weak acids in terms of dissociation?
- How do buffer systems function to resist pH changes?
- What happens when a buffer exceeds its buffer zone?
- pH is a logarithmic scale representing H⁺ concentration.
- Biological processes depend on a stable pH environment.
- Buffers prevent drastic pH changes, maintaining homeostasis.
- The body uses multiple buffers to regulate pH in different physiological compartments.

