1: Chemistry
- Page ID
- 158496
\( \newcommand{\vecs}[1]{\overset { \scriptstyle \rightharpoonup} {\mathbf{#1}} } \)
\( \newcommand{\vecd}[1]{\overset{-\!-\!\rightharpoonup}{\vphantom{a}\smash {#1}}} \)
\( \newcommand{\dsum}{\displaystyle\sum\limits} \)
\( \newcommand{\dint}{\displaystyle\int\limits} \)
\( \newcommand{\dlim}{\displaystyle\lim\limits} \)
\( \newcommand{\id}{\mathrm{id}}\) \( \newcommand{\Span}{\mathrm{span}}\)
( \newcommand{\kernel}{\mathrm{null}\,}\) \( \newcommand{\range}{\mathrm{range}\,}\)
\( \newcommand{\RealPart}{\mathrm{Re}}\) \( \newcommand{\ImaginaryPart}{\mathrm{Im}}\)
\( \newcommand{\Argument}{\mathrm{Arg}}\) \( \newcommand{\norm}[1]{\| #1 \|}\)
\( \newcommand{\inner}[2]{\langle #1, #2 \rangle}\)
\( \newcommand{\Span}{\mathrm{span}}\)
\( \newcommand{\id}{\mathrm{id}}\)
\( \newcommand{\Span}{\mathrm{span}}\)
\( \newcommand{\kernel}{\mathrm{null}\,}\)
\( \newcommand{\range}{\mathrm{range}\,}\)
\( \newcommand{\RealPart}{\mathrm{Re}}\)
\( \newcommand{\ImaginaryPart}{\mathrm{Im}}\)
\( \newcommand{\Argument}{\mathrm{Arg}}\)
\( \newcommand{\norm}[1]{\| #1 \|}\)
\( \newcommand{\inner}[2]{\langle #1, #2 \rangle}\)
\( \newcommand{\Span}{\mathrm{span}}\) \( \newcommand{\AA}{\unicode[.8,0]{x212B}}\)
\( \newcommand{\vectorA}[1]{\vec{#1}} % arrow\)
\( \newcommand{\vectorAt}[1]{\vec{\text{#1}}} % arrow\)
\( \newcommand{\vectorB}[1]{\overset { \scriptstyle \rightharpoonup} {\mathbf{#1}} } \)
\( \newcommand{\vectorC}[1]{\textbf{#1}} \)
\( \newcommand{\vectorD}[1]{\overrightarrow{#1}} \)
\( \newcommand{\vectorDt}[1]{\overrightarrow{\text{#1}}} \)
\( \newcommand{\vectE}[1]{\overset{-\!-\!\rightharpoonup}{\vphantom{a}\smash{\mathbf {#1}}}} \)
\( \newcommand{\vecs}[1]{\overset { \scriptstyle \rightharpoonup} {\mathbf{#1}} } \)
\(\newcommand{\longvect}{\overrightarrow}\)
\( \newcommand{\vecd}[1]{\overset{-\!-\!\rightharpoonup}{\vphantom{a}\smash {#1}}} \)
\(\newcommand{\avec}{\mathbf a}\) \(\newcommand{\bvec}{\mathbf b}\) \(\newcommand{\cvec}{\mathbf c}\) \(\newcommand{\dvec}{\mathbf d}\) \(\newcommand{\dtil}{\widetilde{\mathbf d}}\) \(\newcommand{\evec}{\mathbf e}\) \(\newcommand{\fvec}{\mathbf f}\) \(\newcommand{\nvec}{\mathbf n}\) \(\newcommand{\pvec}{\mathbf p}\) \(\newcommand{\qvec}{\mathbf q}\) \(\newcommand{\svec}{\mathbf s}\) \(\newcommand{\tvec}{\mathbf t}\) \(\newcommand{\uvec}{\mathbf u}\) \(\newcommand{\vvec}{\mathbf v}\) \(\newcommand{\wvec}{\mathbf w}\) \(\newcommand{\xvec}{\mathbf x}\) \(\newcommand{\yvec}{\mathbf y}\) \(\newcommand{\zvec}{\mathbf z}\) \(\newcommand{\rvec}{\mathbf r}\) \(\newcommand{\mvec}{\mathbf m}\) \(\newcommand{\zerovec}{\mathbf 0}\) \(\newcommand{\onevec}{\mathbf 1}\) \(\newcommand{\real}{\mathbb R}\) \(\newcommand{\twovec}[2]{\left[\begin{array}{r}#1 \\ #2 \end{array}\right]}\) \(\newcommand{\ctwovec}[2]{\left[\begin{array}{c}#1 \\ #2 \end{array}\right]}\) \(\newcommand{\threevec}[3]{\left[\begin{array}{r}#1 \\ #2 \\ #3 \end{array}\right]}\) \(\newcommand{\cthreevec}[3]{\left[\begin{array}{c}#1 \\ #2 \\ #3 \end{array}\right]}\) \(\newcommand{\fourvec}[4]{\left[\begin{array}{r}#1 \\ #2 \\ #3 \\ #4 \end{array}\right]}\) \(\newcommand{\cfourvec}[4]{\left[\begin{array}{c}#1 \\ #2 \\ #3 \\ #4 \end{array}\right]}\) \(\newcommand{\fivevec}[5]{\left[\begin{array}{r}#1 \\ #2 \\ #3 \\ #4 \\ #5 \\ \end{array}\right]}\) \(\newcommand{\cfivevec}[5]{\left[\begin{array}{c}#1 \\ #2 \\ #3 \\ #4 \\ #5 \\ \end{array}\right]}\) \(\newcommand{\mattwo}[4]{\left[\begin{array}{rr}#1 \amp #2 \\ #3 \amp #4 \\ \end{array}\right]}\) \(\newcommand{\laspan}[1]{\text{Span}\{#1\}}\) \(\newcommand{\bcal}{\cal B}\) \(\newcommand{\ccal}{\cal C}\) \(\newcommand{\scal}{\cal S}\) \(\newcommand{\wcal}{\cal W}\) \(\newcommand{\ecal}{\cal E}\) \(\newcommand{\coords}[2]{\left\{#1\right\}_{#2}}\) \(\newcommand{\gray}[1]{\color{gray}{#1}}\) \(\newcommand{\lgray}[1]{\color{lightgray}{#1}}\) \(\newcommand{\rank}{\operatorname{rank}}\) \(\newcommand{\row}{\text{Row}}\) \(\newcommand{\col}{\text{Col}}\) \(\renewcommand{\row}{\text{Row}}\) \(\newcommand{\nul}{\text{Nul}}\) \(\newcommand{\var}{\text{Var}}\) \(\newcommand{\corr}{\text{corr}}\) \(\newcommand{\len}[1]{\left|#1\right|}\) \(\newcommand{\bbar}{\overline{\bvec}}\) \(\newcommand{\bhat}{\widehat{\bvec}}\) \(\newcommand{\bperp}{\bvec^\perp}\) \(\newcommand{\xhat}{\widehat{\xvec}}\) \(\newcommand{\vhat}{\widehat{\vvec}}\) \(\newcommand{\uhat}{\widehat{\uvec}}\) \(\newcommand{\what}{\widehat{\wvec}}\) \(\newcommand{\Sighat}{\widehat{\Sigma}}\) \(\newcommand{\lt}{<}\) \(\newcommand{\gt}{>}\) \(\newcommand{\amp}{&}\) \(\definecolor{fillinmathshade}{gray}{0.9}\)- Explain the significance of biochemistry in understanding life processes.
- Describe the origin of elements and their role in biochemical structures.
- Differentiate between atoms, molecules, and compounds.
- Understand the octet rule and how it influences chemical bonding.
- Identify and compare different types of chemical bonds and their biological significance.
- Recognize the importance of key biochemical elements (C, H, O, N) in sustaining life.
- Biochemistry - The study of the chemical processes and substances occurring within living organisms.
- Atom - The smallest unit of matter that retains the properties of an element.
- Molecule - A combination of two or more atoms bonded together.
- Compound - A molecule composed of different types of atoms.
- Element - A pure substance made entirely of one type of atom.
- Atomic Number - The number of protons in an atom’s nucleus.
- Mass Number - The total number of protons and neutrons in an atom’s nucleus.
- Isotope - Variants of an element with different numbers of neutrons.
- Octet Rule - The principle stating that atoms are most stable when they have eight electrons in their outer shell.
- Covalent Bond - A chemical bond where atoms share electrons.
- Ionic Bond - A bond formed by the transfer of electrons, creating charged ions.
- Hydrogen Bond - A weak attraction between a hydrogen atom and an electronegative atom.
- Van der Waals Forces - Weak intermolecular forces due to temporary dipoles.
- Hydrophilic - Molecules that are attracted to water.
- Hydrophobic - Molecules that repel water.
Why Study Biochemistry?
Biochemistry is the study of the molecules and chemical processes that make up life. It helps us understand how life originates, functions, and sustains itself. Have you ever wondered what you and a distant star have in common? The answer is stardust! Everything around us, including our bodies, is made from elements that were formed in the cores of stars billions of years ago (Figure 1.1). When these stars exploded, they scattered their elements across the universe, eventually forming planets and life. This means that the atoms in your body were once part of a star. But how does this stardust become life? Let's break it down step by step.

Figure 1.1: The Origin of Elements. A visual representation of nucleosynthesis in stars, showing how elements are formed and dispersed through supernovae. Image generated via ChatGPT
An atom is the smallest unit of matter that retains the properties of an element. Atoms interact to form molecules and compounds, which are essential for life. A molecule is combination of two or more atoms (e.g., O₂ – oxygen molecule), whereas a compound is a molecule composed of different types of atoms (e.g., H₂O – water). An atom can also be referred to as an element. An element is a substance made entirely of one type of atom. Each element has a unique number of protons, called the atomic number (Figure 1.2). For example, Hydrogen (H) has an atomic number of 1, which means it has 1 proton, whereas Carbon (C) has an atomic number of 6, which means it has 6 protons. The mass number is the total of protons and neutrons in an atom’s nucleus. Different atoms have different masses due to their varying numbers of neutrons.

Figure 1.2: Atomic Structure. Diagram of an atom with labeled protons, neutrons, and electrons, including electron shells. Image of the atomic structure featuring Helium atom.
Isotopes are atoms of the same element that differ in the number of neutrons (Figure 1.3). While isotopes have the same chemical properties, they have different mass numbers. For example, Carbon has two stable isotopes: Carbon-12 (12C) and Carbon-13 (13C), but it also has an unstable isotope, Carbon-14 (14C), which undergoes radioactive decay. Some isotopes are radioactive because their nuclei are unstable. These radioisotopes decay over time, releasing energy. Scientists use this decay to measure the age of fossils and rocks. For example, Carbon-14 decays to Nitrogen-14 in 5730 years (Figure 1.4), whereas Potassium-40 decays to Argon-40 in 1.29 billion years. This process is like a ticking clock, helping us understand the history of our planet and evolution.
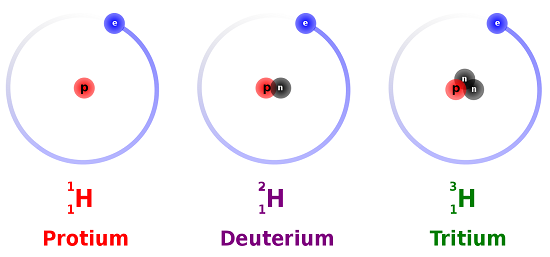
Figure 1.3: Figure The three most stable isotopes of hydrogen: protium (A = 1), deuterium (A = 2), and tritium (A = 3). (CC SA-BY 3.0; Balajijagadesh via Wikipedia).

Figure 1.4: A radioactive isotope of carbon (carbon-14) has six protons and eight neutrons. It decays to a stable isotope of nitrogen (nitrogen-14), which has seven protons and seven neutrons. Radioactive decay releases radiation. (The particular type of radiation that occurs in this example is called beta minus decay, β-.) Carbon-14 decays at a predictable rate, with half of it decaying every 5730 years. Because carbon is abundant in organisms, this predictable decay rate is commonly used for dating fossils—image from CDC (public domain).
Introduction to the Octet Rule
The octet rule states that atoms are most stable when they have eight electrons in their outermost shell (valence shell). This is why atoms tend to gain, lose, or share electrons—to achieve a full outer shell and reach a stable state, like the noble gases. The octet rule is a fundamental principle in chemistry that helps explain why atoms interact the way they do. It states that atoms are most stable when they have a full outer shell of electrons, typically eight for most elements. This drives atoms to either share, lose, or gain electrons to achieve a stable electronic configuration, similar to noble gases, which are naturally inert. For example, sodium (Na) has one valence electron, making it highly reactive as it seeks to lose that electron and become a stable Na⁺ ion. On the other hand, chlorine (Cl) has seven valence electrons and needs one more to complete its outer shell, making it eager to gain an electron and become Cl⁻. When sodium donates its electron to chlorine, they form an ionic bond, creating table salt (NaCl). This concept is crucial in understanding how molecules form and function in biological systems. In human biochemistry, ions like Na⁺, K⁺, and Cl⁻ play key roles in nerve signal transmission, muscle contraction, and maintaining fluid balance. Sodium-potassium pumps (Na⁺/K⁺ ATPase) work because sodium and potassium ions achieve stability by losing or gaining electrons, allowing nerve impulses to travel through neurons. Calcium (Ca²⁺) ions are essential for muscle contractions, as they move across membranes to trigger responses in muscle cells. Oxygen (O₂), which we breathe, forms a stable molecule by sharing electrons between two oxygen atoms in a double bond.
The octet rule helps explain why certain elements are essential for life and why imbalances in these ions can lead to health problems such as electrolyte disorders.
Take a look at the Periodic Table on Figure 1.2 and determine the number of valence electrons for the elements below:
- Sodium (Na) has 1 valence electron and will tend to lose it to become Na⁺.
- Chlorine (Cl) has 7 valence electrons and will tend to gain 1 to become Cl⁻.
- Oxygen (O) has 6 valence electrons and will tend to gain 2 to become O²⁻.
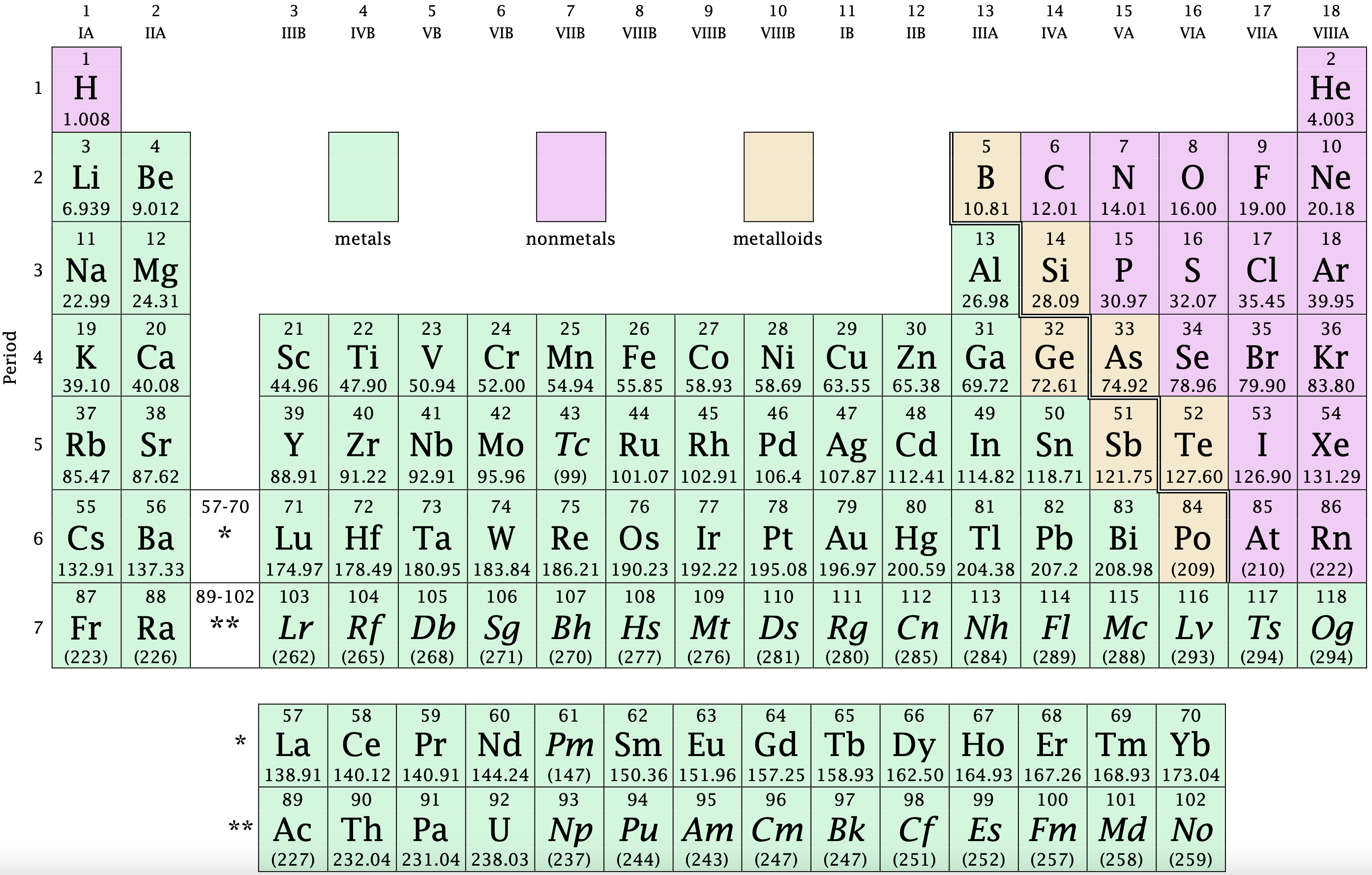
Figure 1.5: The Periodic Table is shared under a CC BY-NC-SA 4.0 license and was authored, remixed, and/or curated by LibreTexts.
Types of Chemical Bonds and Their Importance in Biochemistry and Medicine
Chemical bonds are the forces that hold atoms together in molecules, and they determine the structure and function of biological macromolecules such as proteins, DNA, and enzymes. The three main types of chemical bonds are covalent, ionic, and hydrogen bonds, each with distinct properties. Covalent bonds involve the sharing of electrons between atoms, forming strong and stable connections. These are essential in organic molecules, like the carbon-carbon bonds in DNA and proteins. Within covalent bonding, there are polar covalent bonds, where electrons are shared unequally, and nonpolar covalent bonds, where electrons are shared equally (Figure 1.6 and 1.7). The strength of covalent bonds ensures that enzymes can perform complex reactions without breaking apart easily. Many drugs target covalent interactions in enzymes, like aspirin, which forms a covalent bond with an enzyme to reduce inflammation. This difference plays a role in molecular polarity, which affects solubility and interactions with other molecules.
- Nonpolar Covalent Bonds: Electrons are shared equally (e.g., O₂, H₂, CH₄).
- Polar Covalent Bonds: Electrons are shared unequally, leading to partial charges (e.g., H₂O, NH₃).
Figure 1.6: Pauling scale electronegativities of elements. Licensed CC BY-SA 4.0.

Figure 1.7: The Electron Distribution in a Nonpolar Covalent Bond, a Polar Covalent Bond, and an Ionic Bond Using Lewis Electron Structures. Electron-rich (negatively charged) regions are shown in blue; electron-poor (positively charged) regions are shown in red. (CC BY-SA-NC)
Ionic bonds, on the other hand, form when electrons are completely transferred from one atom to another, creating positively and negatively charged ions. These bonds are weaker in aqueous solutions because water molecules can surround and separate the ions. In medicine, this is significant because many drugs are formulated as salts (such as sodium or potassium salts) to improve solubility in the bloodstream. Na⁺ and Cl⁻ form salt (NaCl), which dissolves in water to help maintain fluid balance in cells. Calcium ions (Ca²⁺) are crucial in blood clotting, muscle contractions, and nerve transmission. Many medications work by interacting with ionic sites on proteins, affecting how enzymes function.
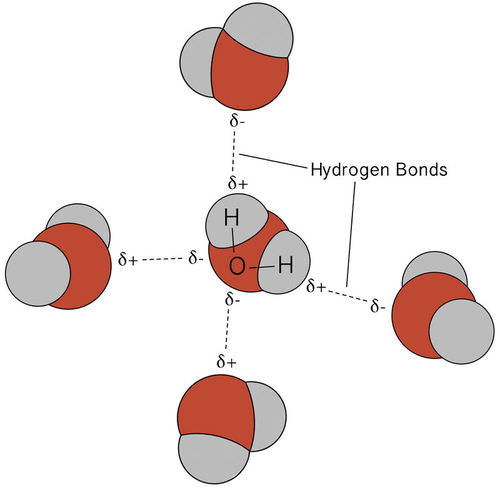
Hydrogen bonds are weak but critical for biological function. They occur between molecules with polar covalent bonds, such as water molecules, and play a key role in maintaining the three-dimensional structures of proteins and nucleic acids (Figure 1.8). In drug studies, hydrogen bonding is essential because it determines how a drug interacts with its target protein. Many drugs function by mimicking natural molecules and forming hydrogen bonds with enzymes or receptors, enhancing their effectiveness. For example, DNA base pairing relies on hydrogen bonding between adenine and thymine and cytosine and guanine. The double helix structure of DNA is stabilized by hydrogen bonds between base pairs (A-T and G-C). The shape of enzymes depends on hydrogen bonding, which determines how they interact with substrates. Many drugs form hydrogen bonds with their target molecules, improving their effectiveness and specificity. Any disruption to these bonds can lead to mutations, which can have serious consequences in diseases like cancer.
Figure 1.8: Multiple hydrogen bonds occur simultaneously in water because of its bent shape and the presence of two hydrogen atoms per molecule. Image by Allison Soult, Ph.D. (Department of Chemistry, University of Kentucky).
Among the various types of chemical bonds and interactions, Van der Waals interactions are usually the least taught in basic chemistry courses. However, these weak forces play a critical role in biological systems, drug interactions, and even the physical abilities of certain animals. Van der Waals forces are weak attractions that occur due to temporary shifts in electron density, creating momentary partial charges in otherwise nonpolar molecules (Figure 1.9). This temporary charge can influence nearby molecules, leading to subtle but significant conformational changes in proteins, enzymes, and other biomolecules. A useful analogy to understand Van der Waals interactions is to think of a magnet pulling all electrons to one side of a molecule. Even though the molecule may not normally have a charge, this temporary shift creates a weak attraction between it and a neighboring molecule. These induced dipoles allow nonpolar molecules, which typically do not interact strongly, to exhibit weak attractive forces. While individually weak (with bond strengths between 0.4-4 kJ/mol), these forces become significant when many such interactions occur together, contributing to molecular stability and shape. One of the most famous real-world examples of Van der Waals interactions is how geckos climb walls. Geckos have microscopic hair-like structures, called setae, on their feet (Figure 1.10). These setae branch out into even finer structures that maximize contact with surfaces, allowing the gecko to take advantage of Van der Waals forces. Even though these interactions are weak, the sheer number of tiny contacts between the gecko’s feet and a surface generates enough attraction to support the animal’s weight—allowing it to cling to walls and even walk upside down on ceilings.

Figure 1.9: A molecule, represented as the magnet, that can contribute to shifting electrons of another molecule.

Figure 1.10: Here is an image of a gecko climbing a wall using Van der Waals forces. Image generated via ChatGPT.
In biochemistry and medicine, Van der Waals interactions contribute to the binding specificity between drugs and their targets. Many drugs fit into enzyme active sites using a combination of hydrogen bonds, ionic interactions, and Van der Waals forces. These weak forces also help proteins fold into their correct functional shapes, ensuring biological activity. If these interactions are disrupted—such as in protein misfolding diseases like Alzheimer's—serious health consequences can occur. Therefore, despite often being overlooked, Van der Waals interactions play a vital role in both molecular stability and biological function.
Hydrophilic vs. Hydrophobic Interactions in Enzyme and Drug Studies
Hydrophilic and hydrophobic interactions also play a crucial role in the structure and function of biological molecules. Hydrophilic molecules, which have a charge or polar groups, interact well with water and dissolve easily in aqueous environments. In contrast, hydrophobic molecules, which are nonpolar, tend to repel water and cluster together to minimize contact with it (Figure 1.11). This distinction is vital in biochemistry because the way molecules interact with water determines their function in the body.

Figure 1.11: The Attraction between Lipid molecules and water molecules will separate. This is driven by an increase in entropy (disorder) due to water exclusion
In enzymes and drug development, hydrophilic and hydrophobic properties influence how drugs are absorbed, distributed, and interact with their targets. For instance, cell membranes are composed of phospholipids, which have amphipathic properties—meaning they have both hydrophilic heads and hydrophobic tails. This structure forms the lipid bilayer, creating a selective barrier that controls what enters and exits the cell. Drugs designed to cross cell membranes must often have hydrophobic properties to penetrate the lipid layer, while others need to be hydrophilic to dissolve in blood plasma. Understanding these interactions allows scientists to modify drug structures for better delivery and effectiveness. Additionally, proteins fold in a way that places hydrophobic amino acids inside, away from water, while hydrophilic amino acids remain on the outside. This folding is crucial for enzyme function, and any disruption—such as in genetic diseases or misfolded proteins in Alzheimer's—can have severe consequences.
Comparison of Bond Strengths and Their Importance in Drug Development
Understanding bond strength is crucial in drug design, as the type of bonds formed between a drug and its biological target determines how strongly and how long the drug remains active in the body. Different types of chemical bonds—covalent, ionic, hydrogen, hydrophobic interactions, and Van der Waals forces—play distinct roles in drug effectiveness, metabolism, and elimination. The stronger the bond, the more stable the interaction; however, in some cases, transient and reversible interactions are more desirable to allow for controlled biological effects.
Covalent bonds are the strongest, with bond energies ranging from 100-1000 kJ/mol. These bonds form when two atoms share electrons, making them nearly irreversible in biological conditions. Drugs that form covalent bonds with their targets often have long-lasting effects because breaking the bond requires enzymatic intervention or protein turnover. One of the most well-known covalent inhibitors is Aspirin (Acetylsalicylic Acid), which irreversibly inhibits cyclooxygenase (COX-1 and COX-2) enzymes. By forming a covalent bond with a serine residue in the active site, aspirin permanently disables the enzyme, reducing inflammation and pain until new enzymes are synthesized. Another example is Clopidogrel (Plavix), an antiplatelet drug that covalently binds to the P2Y12 receptor on platelets, preventing clot formation in heart disease patients. Similarly, some cancer drugs, like Ibrutinib, use covalent bonding to target specific proteins, ensuring prolonged inhibition of cancer cell signaling.
Ionic bonds, with bond strengths around 20 kJ/mol, occur due to the attraction between positively and negatively charged molecules. Many drugs exploit ionic interactions to bind to their targets while still allowing for reversible binding, which is critical for regulated biological activity. For instance, Atorvastatin (Lipitor), a cholesterol-lowering drug, binds ionically to HMG-CoA reductase, the enzyme responsible for cholesterol synthesis. This interaction effectively blocks the enzyme's function but allows for controlled drug clearance, preventing excessive cholesterol reduction. Beta-lactam antibiotics, like Penicillin, also use ionic interactions to bind to bacterial enzymes (transpeptidases), disrupting cell wall synthesis and killing bacteria.
Hydrogen bonds, which range in strength from 12-30 kJ/mol, are vital for increasing drug selectivity and specificity. These bonds form between a hydrogen atom and electronegative elements like oxygen or nitrogen, stabilizing drug-protein interactions. A classic example is Remdesivir, an antiviral drug designed to treat COVID-19. It forms multiple hydrogen bonds with the RNA-dependent RNA polymerase (RdRp) enzyme of SARS-CoV-2, mimicking natural nucleotides and disrupting viral replication. Similarly, Oseltamivir (Tamiflu), an anti-influenza drug, relies on hydrogen bonding to fit snugly into the active site of neuraminidase, preventing viral spread.
Hydrophobic interactions, though weaker than covalent and ionic bonds, are essential in drugs that interact with cell membranes or hydrophobic protein pockets. These interactions occur due to water exclusion, forcing nonpolar molecules together, with bond strengths typically less than 40 kJ/mol. Drugs like Tamoxifen, used in breast cancer treatment, bind to estrogen receptors via hydrophobic interactions, blocking estrogen signaling and slowing cancer growth. Similarly, Statins (such as Simvastatin and Atorvastatin) fit into the hydrophobic active site of HMG-CoA reductase, reducing cholesterol synthesis.
Van der Waals forces, the weakest of all at 0.4-4 kJ/mol, occur due to temporary dipole-induced attractions. Although weak, these interactions contribute significantly to drug-receptor binding stability and fine-tuning molecular fit. One example is Benzodiazepines (like Diazepam/Valium and Alprazolam/Xanax), which bind to GABA-A receptors through a combination of hydrogen bonds and Van der Waals forces. These interactions help modulate receptor activity, producing anxiolytic (anti-anxiety) effects. Another example is HIV protease inhibitors (such as Ritonavir and Lopinavir), which use Van der Waals interactions to snugly fit into the hydrophobic pocket of HIV protease, preventing viral replication.
A deep understanding of bond strength is essential in drug design because different therapeutic goals require different types of binding interactions. Covalent inhibitors offer long-lasting effects but must be used cautiously to avoid off-target toxicity. Ionic and hydrogen bonds allow for strong, yet reversible interactions, ensuring drug effectiveness while preventing excessive buildup. Hydrophobic and Van der Waals forces help improve drug specificity, allowing precise molecular targeting. By fine-tuning these interactions, pharmaceutical scientists can develop drugs with optimal binding, selectivity, and duration of action, leading to more effective and safer treatments for diseases.
Bond Strength Comparison
| Bond Type | Strength (kJ/mol) |
|---|---|
| Covalent Bonds | 100 - 1000 |
| Ionic Bonds | ~20 |
| Hydrogen Bonds | 12 - 30 |
| Hydrophobic Interactions | < 40 |
| Van der Waals Forces | 0.4 - 4 |
Table 1.1: Bond Strength Comparison between covalent, ionic, hydrogen, hydrophobic interactions, and Van der Waals forces
The Importance of Key Elements in Biochemistry and Human Health
The periodic table serves as a fundamental tool in chemistry, organizing elements based on their properties. However, in biochemistry, only a select group of elements play a direct role in sustaining life. These essential elements—carbon (C), hydrogen (H), oxygen (O), nitrogen (N) —form the building blocks of biomolecules, regulate cellular functions, and maintain homeostasis in the human body. Understanding these elements is critical to grasping the biochemical processes that govern health and disease.
Carbon (C) is often referred to as the backbone of life because of its unparalleled ability to form stable covalent bonds with other elements. Its versatility allows it to create long chains, rings, and highly complex biomolecules such as glucose, proteins, fats, and DNA. Every organic molecule that makes up the human body—carbohydrates, lipids, nucleic acids, and proteins—relies on carbon’s ability to form multiple strong bonds. Without carbon, the structure and function of life-sustaining molecules would not be possible.
Hydrogen (H) plays an equally critical role in biological systems, particularly in maintaining pH balance and energy transfer. Hydrogen is a major component of water (H₂O), the most abundant molecule in the human body, and is also present in biological acids and bases. It participates in cellular respiration by driving the electron transport chain, which ultimately produces ATP, the primary energy currency of cells. Moreover, hydrogen bonding is responsible for stabilizing the double-helix structure of DNA, ensuring the proper replication and function of genetic material.
Oxygen (O) is essential for survival, as it drives cellular respiration, the process by which cells extract energy from glucose. In the mitochondria, oxygen serves as the final electron acceptor in the electron transport chain, allowing ATP to be synthesized. Without oxygen, aerobic respiration cannot occur efficiently, leading to decreased energy production and potential cell death. Oxygen also plays a role in immune responses and is a key component of many biologically significant molecules, including hemoglobin, which transports oxygen in the blood.
Nitrogen (N) is another indispensable element in biochemistry, forming the backbone of amino acids, proteins, and nucleotides. It is present in DNA and RNA, where it forms the nitrogenous bases (adenine, thymine, cytosine, guanine, and uracil) that encode genetic information. Additionally, nitrogen is crucial for neurotransmitter production, influencing mood, cognition, and overall brain function. Without nitrogen, life-sustaining molecules such as enzymes, hormones, and structural proteins could not be synthesized.
In summary, these key elements—C, H, O, N—are foundational to life. Their interactions govern the structure and function of biomolecules, drive metabolic processes, and maintain physiological balance. Understanding their roles provides insight into how the human body functions at a molecular level and highlights the delicate biochemical equilibrium required for health and survival. Understanding biochemistry helps us grasp how life works at a molecular level. From the atoms in our bodies to the reactions that keep us alive, everything follows the laws of chemistry. The next time you drink water, eat food, or breathe, remember—you’re interacting with the fundamental forces of biochemistry, made possible by stardust!
------------------------------------------------------------------
- Life is built from elements originating from stars, emphasizing the universality of chemistry in biology.
- Atoms form molecules and compounds through various types of bonds, which influence biological function.
- The octet rule explains atomic stability and drives interactions leading to molecular formation.
- Different types of bonds—covalent, ionic, hydrogen, and Van der Waals—play essential roles in biochemistry and medicine.
- Essential elements (C, H, O, N) are fundamental to biological molecules, metabolic pathways, and life processes.
- How does the concept of stardust help us understand the composition of biological molecules?
- Why do atoms form bonds, and how do different bond types impact biological function?
- How do hydrogen bonds contribute to the stability of DNA and protein structures?
- What would happen if Van der Waals forces were absent in biomolecules?
- Why is understanding chemical bonding important in drug development and medical applications?
- Research a drug that relies on a specific type of chemical bond for its effectiveness. Explain how the bond contributes to its function.
- Examine the periodic table and determine how the position of an element influences its bonding properties and role in biochemistry.