4.3: NAD and NADP
- Page ID
- 4629
Nicotinamide adenine dinucleotide (NAD) and its relative nicotinamide adenine dinucleotide phosphate (NADP) are two of the most important coenzymes in the cell. NADP is simply NAD with a third phosphate group attached as shown at the bottom of the figure.
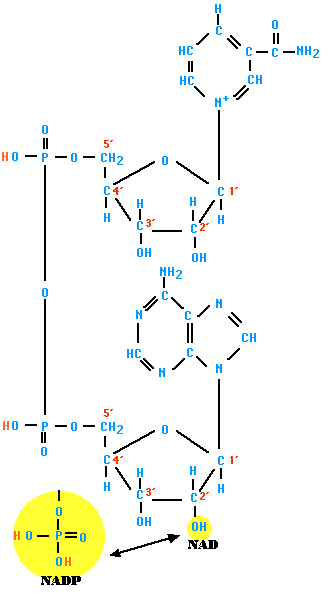
Because of the positive charge on the nitrogen atom in the nicotinamide ring (upper right), the oxidized forms of these important redox reagents are often depicted as NAD+ and NADP+ respectively.
In cells, most oxidations are accomplished by the removal of hydrogen atoms. Both of these coenzymes play crucial roles in this. Each molecule of NAD+ (or NADP+) can acquire two electrons; that is, be reduced by two electrons. However, only one proton accompanies the reduction. The other proton produced as two hydrogen atoms are removed from the molecule being oxidized is liberated into the surrounding medium. For NAD, the reaction is thus:
\[\ce{NAD^{+} + 2H -> NADH + H^{+}}\]
NAD and NADP uses
NAD participates in many redox reactions in cells, including those in glycolysis and most of those in the citric acid cycle of cellular respiration.
NADP is the reducing agent produced by the light reactions of photosynthesis and is consumed in the Calvin cycle of photosynthesis and used in many other anabolic reactions in both plants and animals.
Under the conditions existing in a normal cell, the hydrogen atoms shown in red are dissociated from these acidic substances.


