5.2: Organizers-Other Organizers
- Page ID
- 21540
\( \newcommand{\vecs}[1]{\overset { \scriptstyle \rightharpoonup} {\mathbf{#1}} } \)
\( \newcommand{\vecd}[1]{\overset{-\!-\!\rightharpoonup}{\vphantom{a}\smash {#1}}} \)
\( \newcommand{\dsum}{\displaystyle\sum\limits} \)
\( \newcommand{\dint}{\displaystyle\int\limits} \)
\( \newcommand{\dlim}{\displaystyle\lim\limits} \)
\( \newcommand{\id}{\mathrm{id}}\) \( \newcommand{\Span}{\mathrm{span}}\)
( \newcommand{\kernel}{\mathrm{null}\,}\) \( \newcommand{\range}{\mathrm{range}\,}\)
\( \newcommand{\RealPart}{\mathrm{Re}}\) \( \newcommand{\ImaginaryPart}{\mathrm{Im}}\)
\( \newcommand{\Argument}{\mathrm{Arg}}\) \( \newcommand{\norm}[1]{\| #1 \|}\)
\( \newcommand{\inner}[2]{\langle #1, #2 \rangle}\)
\( \newcommand{\Span}{\mathrm{span}}\)
\( \newcommand{\id}{\mathrm{id}}\)
\( \newcommand{\Span}{\mathrm{span}}\)
\( \newcommand{\kernel}{\mathrm{null}\,}\)
\( \newcommand{\range}{\mathrm{range}\,}\)
\( \newcommand{\RealPart}{\mathrm{Re}}\)
\( \newcommand{\ImaginaryPart}{\mathrm{Im}}\)
\( \newcommand{\Argument}{\mathrm{Arg}}\)
\( \newcommand{\norm}[1]{\| #1 \|}\)
\( \newcommand{\inner}[2]{\langle #1, #2 \rangle}\)
\( \newcommand{\Span}{\mathrm{span}}\) \( \newcommand{\AA}{\unicode[.8,0]{x212B}}\)
\( \newcommand{\vectorA}[1]{\vec{#1}} % arrow\)
\( \newcommand{\vectorAt}[1]{\vec{\text{#1}}} % arrow\)
\( \newcommand{\vectorB}[1]{\overset { \scriptstyle \rightharpoonup} {\mathbf{#1}} } \)
\( \newcommand{\vectorC}[1]{\textbf{#1}} \)
\( \newcommand{\vectorD}[1]{\overrightarrow{#1}} \)
\( \newcommand{\vectorDt}[1]{\overrightarrow{\text{#1}}} \)
\( \newcommand{\vectE}[1]{\overset{-\!-\!\rightharpoonup}{\vphantom{a}\smash{\mathbf {#1}}}} \)
\( \newcommand{\vecs}[1]{\overset { \scriptstyle \rightharpoonup} {\mathbf{#1}} } \)
\(\newcommand{\longvect}{\overrightarrow}\)
\( \newcommand{\vecd}[1]{\overset{-\!-\!\rightharpoonup}{\vphantom{a}\smash {#1}}} \)
\(\newcommand{\avec}{\mathbf a}\) \(\newcommand{\bvec}{\mathbf b}\) \(\newcommand{\cvec}{\mathbf c}\) \(\newcommand{\dvec}{\mathbf d}\) \(\newcommand{\dtil}{\widetilde{\mathbf d}}\) \(\newcommand{\evec}{\mathbf e}\) \(\newcommand{\fvec}{\mathbf f}\) \(\newcommand{\nvec}{\mathbf n}\) \(\newcommand{\pvec}{\mathbf p}\) \(\newcommand{\qvec}{\mathbf q}\) \(\newcommand{\svec}{\mathbf s}\) \(\newcommand{\tvec}{\mathbf t}\) \(\newcommand{\uvec}{\mathbf u}\) \(\newcommand{\vvec}{\mathbf v}\) \(\newcommand{\wvec}{\mathbf w}\) \(\newcommand{\xvec}{\mathbf x}\) \(\newcommand{\yvec}{\mathbf y}\) \(\newcommand{\zvec}{\mathbf z}\) \(\newcommand{\rvec}{\mathbf r}\) \(\newcommand{\mvec}{\mathbf m}\) \(\newcommand{\zerovec}{\mathbf 0}\) \(\newcommand{\onevec}{\mathbf 1}\) \(\newcommand{\real}{\mathbb R}\) \(\newcommand{\twovec}[2]{\left[\begin{array}{r}#1 \\ #2 \end{array}\right]}\) \(\newcommand{\ctwovec}[2]{\left[\begin{array}{c}#1 \\ #2 \end{array}\right]}\) \(\newcommand{\threevec}[3]{\left[\begin{array}{r}#1 \\ #2 \\ #3 \end{array}\right]}\) \(\newcommand{\cthreevec}[3]{\left[\begin{array}{c}#1 \\ #2 \\ #3 \end{array}\right]}\) \(\newcommand{\fourvec}[4]{\left[\begin{array}{r}#1 \\ #2 \\ #3 \\ #4 \end{array}\right]}\) \(\newcommand{\cfourvec}[4]{\left[\begin{array}{c}#1 \\ #2 \\ #3 \\ #4 \end{array}\right]}\) \(\newcommand{\fivevec}[5]{\left[\begin{array}{r}#1 \\ #2 \\ #3 \\ #4 \\ #5 \\ \end{array}\right]}\) \(\newcommand{\cfivevec}[5]{\left[\begin{array}{c}#1 \\ #2 \\ #3 \\ #4 \\ #5 \\ \end{array}\right]}\) \(\newcommand{\mattwo}[4]{\left[\begin{array}{rr}#1 \amp #2 \\ #3 \amp #4 \\ \end{array}\right]}\) \(\newcommand{\laspan}[1]{\text{Span}\{#1\}}\) \(\newcommand{\bcal}{\cal B}\) \(\newcommand{\ccal}{\cal C}\) \(\newcommand{\scal}{\cal S}\) \(\newcommand{\wcal}{\cal W}\) \(\newcommand{\ecal}{\cal E}\) \(\newcommand{\coords}[2]{\left\{#1\right\}_{#2}}\) \(\newcommand{\gray}[1]{\color{gray}{#1}}\) \(\newcommand{\lgray}[1]{\color{lightgray}{#1}}\) \(\newcommand{\rank}{\operatorname{rank}}\) \(\newcommand{\row}{\text{Row}}\) \(\newcommand{\col}{\text{Col}}\) \(\renewcommand{\row}{\text{Row}}\) \(\newcommand{\nul}{\text{Nul}}\) \(\newcommand{\var}{\text{Var}}\) \(\newcommand{\corr}{\text{corr}}\) \(\newcommand{\len}[1]{\left|#1\right|}\) \(\newcommand{\bbar}{\overline{\bvec}}\) \(\newcommand{\bhat}{\widehat{\bvec}}\) \(\newcommand{\bperp}{\bvec^\perp}\) \(\newcommand{\xhat}{\widehat{\xvec}}\) \(\newcommand{\vhat}{\widehat{\vvec}}\) \(\newcommand{\uhat}{\widehat{\uvec}}\) \(\newcommand{\what}{\widehat{\wvec}}\) \(\newcommand{\Sighat}{\widehat{\Sigma}}\) \(\newcommand{\lt}{<}\) \(\newcommand{\gt}{>}\) \(\newcommand{\amp}{&}\) \(\definecolor{fillinmathshade}{gray}{0.9}\)Organizers
One of the most interesting things about building animal bodies is the diversity we see across and within bodies. For example, even though the Hox gene patterning in fruit flies and mice is very similar, the end results (an adult mouse or an adult fly) are extremely different. We see intriguing diversity within bodies as well. For example, a thoracic segment in a fruit fly might express a single Hox gene across a segment, yet parts of this segment take on many different forms. Much of this differentiation is ruled by local organizers and master control genes. These are locally expressed genes that are often expressed in many segments in similar spots. Depending on the Hox gene expressed in that segment, they will activate different body parts. For example, we will look later on at wing master control genes -these genes build wings in thoracic segments and different structures, like gin traps, in other segments. The type of wing - elytra, haltere, flying wing, etc. is also dependent on the position of the segment in the body and the species it is expressed in. For example, dragonflies have two flying wings, while the first wing in a beetle is a protective elytron, and the second wing in a fly is a proprioceptive organ.
This type of programming is known as serial homology. We will be coming back to this concept later, but it is a conceptually important idea in Evo-Devo. Serial homologs are a special type of homolog wherein the same type of tissue expressing a core set of genetic regulators (an organizer) is found in multiple spots along a body axis. One easy to understand example of serial homologs are arms and legs. Developmentally, they are nearly identical for the first part of their growth, they only grow substantial differences later. They express the same core limb genes (as we will soon consider) and have the same bones, albeit in slightly different shapes. Serial homologs are incredibly important in our understanding of evolution because they are genetically "cheap," since they use the same core gene regulatory network, but they can provide novel functions -like tool use plus locomotion. The fate of a serial homolog often depends on the Hox genes that are expressed in the region of the serial homolog organizer.
Organizer outputs are not only affected by the Hox code, they can also themselves set up the Hox code (as in the case of Nanos and Bicoid) or can set up a different axis (like the dorsoventral axis of a frog or the anteroposterior axis of a limb). Now that you have a firm grasp of gene regulatory networks, we can take a closer look at a famous organizer we have already considered - the Spemann-Mangold organizer of the frog gastrula.
The genetics of the Spemann-Mangold organizer and the notochord
If you need a quick refresher on the Spemann-Mangold organizer, check out Cleavage and Gastrulation. Briefly, the Spemann-Mangold organizer is mesoderm found at the position of the grey crescent, the dorsal pole of the frog (or newt) embryo. This organizer, as it develops into the notochord, induces the formation of dorsal structures like the central nervous system and spine. Amphibians with an extra Spemann-Mangold organizer grow a second A/P axis (conjoined twin tadpoles) and amphibians missing a Spemann-Mangold organizer develop into a "belly piece."
Using genetics, we can explain two huge questions: 1) What causes the organizer to develop? and 2) How does it induce the formation of dorsal structures? The first step in organizer formation is cortical rotation itself, as shown in Figure 1 of Cortical Rotation. In cortical rotation, microtubules rotate the outer cortex of the fertilized egg relative to the inner mass. The vegetal pole has several localized proteins and mRNAs tethered to its cytoskeleton. In the inner mass this includes VegT and Vg17,8. The outer cortex has vegetally localized Dishevelled (Dsh) protein, a component of the Wnt pathway, which is transported towards the animal pole during cortical rotation9. The displacement of Dsh creates a new zone - a part of the embryo with Dsh, but no Vg1 or VegT (Figure 8)10. Dsh helps localize and stabilize b-catenin, the transcription factor of the canonical Wnt pathway. Now we have Vg1 and VegT at the vegetal pole and Dsh and b-catenin at the future dorsal end.
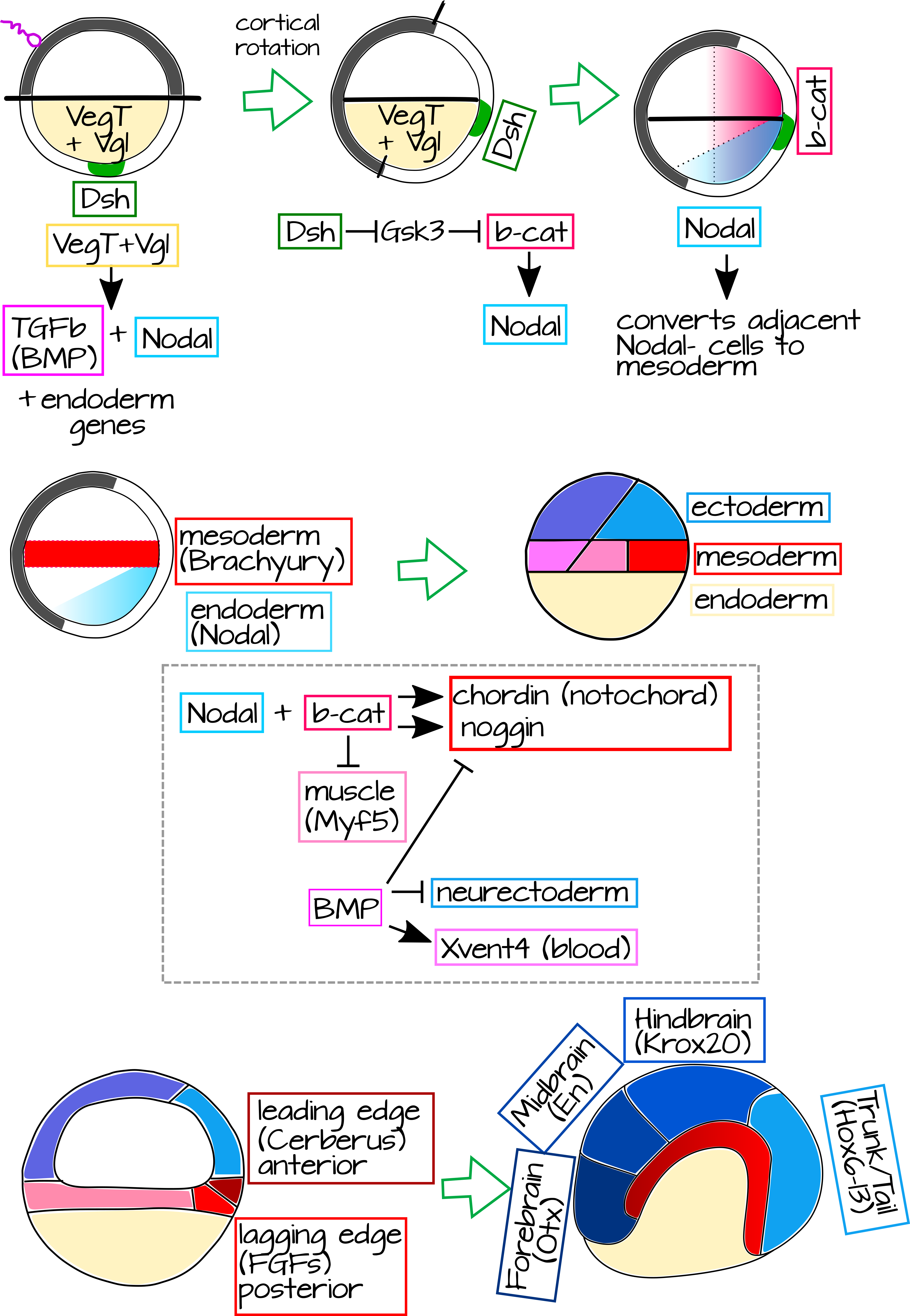 |
Figure 8: Genetics of the Spemann-Mangold organizer. Maternally derived proteins set up an Animal/Vegetal axis in the unfertilized egg, including VegT and Vg1 in the Vegetal pole inner cortex (orange). After fertilization, cortical rotation brings the Vegetal pole outer cortex protein Dishevelled (Dsh, green) into contact with the Animal inner cortex, driving the accumulation of b-catenin in dorsal cells. Vegetal pole proteins activate the expression of TGFb (BMP) and Nodal. Nodal converts the adjacent Nodal- cells into mesoderm (red), resulting in a stripe of mesoderm just above the vegetal pole. Meanwhile, b-catenin enhances Nodal expression in the dorsal-most part of the embryo - resulting in high Nodal expression in the dorsal vegetal inner cortex. This, plus b-catenin, drives the expression of organizer genes like Chordin and Noggin, while repressing ventral mesoderm fates like muscle. Chordin and Noggin specify the notochord, which will elongate and induce the dorsal ectoderm (all ectoderm is blue) that lies over it to become neurectoderm. BMP represses organizer activity on the ventral side of the embryo, in particular ventral ectoderm will form skin, not neurectoderm. The notochord itself has a leading (anterior) and lagging (posterior) edge which express different genes and will induce different parts of the central nervous system. An editable svg file of this figure is available at https://scholarlycommons.pacific.edu/open-images/27/ |
As cleavage proceeds, VegT and Vg1 induce the expression of Nodal (specifically Xenopus nodal related protein or Xnr) in the vegetal-most cells. These cells will become the endoderm. Nodal, a homolog of TGFb, signals to nearby cells lacking Nodal (Nodal- cells), these cells will become the mesoderm. Nodal expression is also induced by b-catenin. This makes a gradient of Nodal that is higher at the dorsal side, where it is induced by VegT and Vg1 as well as b-catenin. While b-catenin is stabilized in a wide swath of dorsal tissue, Nodal expression is restricted to the vegetal side of the embryo where it is expressed all over, but highest where b-catenin is present (Figure 8). Where Nodal signaling meets mesodermal cells with high b-catenin, we see the expression of a new suite of genes - the organizer genes Chordin, Noggin, Frizzled, and more (Figure 8)11.
These organizer genes are transcription factors and members of signal transduction pathways. Some of them will specify the organizer mesoderm as "dorsal" (notochord and somites) and some of them will signal to the overlying ectoderm during gastrulation and specify it as "neural" rather than epidermal. The physical mechanics of this process are fascinating, and we can study them later if you are interested, but for now I will focus just on the genetics and briefly summarize the tissue movements. As you saw before, mesodermal organizer cells undergo involution at the dorsal lip of the blastopore and crawl along the overlying ectodermal cells to form a thick layer beneath them. Most of the mesoderm in the gastrulating embryo expresses BMP4, a TGFb family secreted protein. This BMP4 signals to the overlying ectoderm to become epidermis. However, the organizer expresses BMP4 antagonists, like Chordin and Noggin12. This causes the ectoderm above the organizer mesoderm to take on neural fates.
The organizer itself is not a homogenous structure, it is made up of several cell populations that express different signaling genes and transcription factors. For example, the leading edge of the organizer (the part that will invaginate first and become the anterior-most notochord) expresses a Wnt and BMP4 antagonist called Cerberus13. Cerberus and other leading edge genes specify the anteriormost portions of the notochord via gene regulatory networks that will ultimately lead to expression of forebrain gene Otx214 in the overlying ectoderm. Organizer cells trailing this leading edge express the secreted signaling molecule FGF, which drives the expression of midbrain gene Krox20 in the ectoderm above it. Both Otx2 and Krox20 help make the Hox gene expression pattern, along with other organizer genes expressed in an anteroposterior pattern in the notochord14. In this way, the notochord is able to direct the anteroposterior patterning of the neural tube (Figure 8).
Other Organizers
There are a multitude of other organizers in developmental biology and their fundamental properties give us insights into the genetics of evolution. They are specified by external factors, for example by the intersection of Nodal-adjacent and b-catenin expressing cells in the Spemann-Mangold organizer. They act on other tissues via secreted signaling molecules, for example chordin inhibiting BMP4 to specify neurectoderm. And their activity on other tissues can vary over time and/or space, for example the ability of the notochord to induce both anterior and posterior nervous system structures.
The notochord has a third organizing activity on the ectoderm (not to mention it's organizing activity on the mesoderm), this is patterning the D/V axis of the neural tube. During gastrulation, the ectoderm lying over the notochord responds to signals from it to fold up into a tube and sinks under the epidermis. This tube is polarized into the dorsal side (close to the overlying epidermis) and the ventral side (close to the notochord). A Shh signal from the notochord drives Shh secretion from the ventral side. This Shh gradient opposes a dorsal BMP (TGFb family) gradient initiated by epidermal signaling (Figure 9).
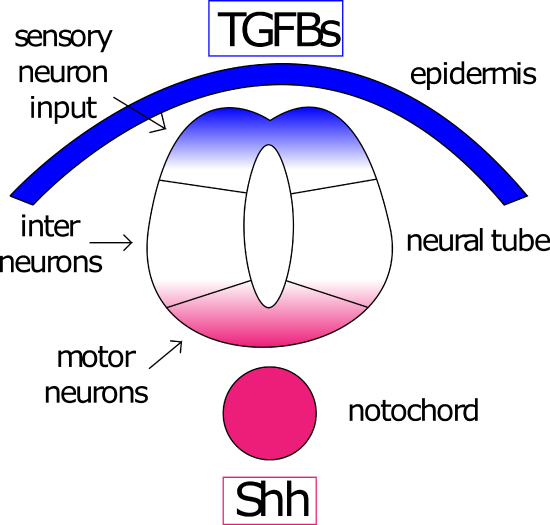 Figure 8: Opposing gradients pattern the neural tube. TGFbs (blue) from the epidermis and Sonic Hedgehog (Shh, red) from the notochord pattern the dorsal/ventral axis of the neural tube. Neurons exposed to a high concentration of TGFbs will become sensory neurons, those exposed to high levels of Shh will become motor neurons. Neurons exposed to low levels of both will become interneurons. Figure by Amanda Lo and Ajna Rivera
Figure 8: Opposing gradients pattern the neural tube. TGFbs (blue) from the epidermis and Sonic Hedgehog (Shh, red) from the notochord pattern the dorsal/ventral axis of the neural tube. Neurons exposed to a high concentration of TGFbs will become sensory neurons, those exposed to high levels of Shh will become motor neurons. Neurons exposed to low levels of both will become interneurons. Figure by Amanda Lo and Ajna Rivera
An editable svg file of this figure is available at https://scholarlycommons.pacific.edu/open-images/28/
These two opposing gradients lead to gradients of active transcription factors responsive to these signal transduction pathways and the expression of genes with cis-regulatory elements responsive to these transcription factors (Figure 9)15. Le Dreu and Marti name 11 distinct domains identified by unique transcription factor expression and neuronal subtype15.
Another famous organizer is one that you encountered in Intro Bio: the tetrapod limb organizer. This is a two-part organizer, with a long thin swath of cells at the distal-most point of the limb expressing and secreting growth-promoting FGFs, and the posterior-most point of the limb expressing Sonic Hedgehog (Shh, Figure 10). The FGF portion of the limb is called the Apical Ectodermal Ridge. It is the signaling center that keeps the limb adding more distal elements onto the end. In this way, the more proximal elements, like the humerus and elbow, develop first. More distal elements, like forearm, wrist, and hands, get laid down later. Interestingly, this system seems to be on a timer, rather than being strictly spatially patterned. If cell-division is inhibited midway through arm development, radius, ulna, and humerus can be small or absent, with a normally proportioned hand and shoulder girdle. This looks somewhat similar to gap mutant in Drosophila, though the developmental mechanism is completely different.
Figure 10: The tetrapod limb organizer. The apical ectodermal ridge (AER, red) secretes FGFs that keep the underlying cells proliferating and maintain Shh signaling from the Zone of Polarizing Activity (ZPA, blue). The Shh protein secreted from ZPA cells forms a gradient along the A/P (thumb/pinky) axis of the limb. Low/no Shh specifies thumb and index finger fates, high Shh specifies pinky and ring finger fates. What happens if we add Shh to the other side of the developing limb? Image from wikimedia commons user Peteruetz under a CC BY-SA 3.0 license
The Shh signaling center is a true organizer. It not only secretes a signaling molecule like the AER, but this signaling molecule acts in a graded fashion. High levels of Shh result in the development of posterior elements of the limb, like the radius or pinky, while low levels result in the development of mid-elements, like the middle fingers, and absence of Shh leads to anterior elements, like the thumb.
In general, signal transduction and transcription factor cascades are responsible for much of the patterning of the developing embryo. This gives us a big hint as to how evolution can act on development to change adult morphology. Small changes in the amount or location of a morphogen can have big effects on the adult. The same "toolkit" of genes is conserved in evolution, but is also reused in many different contexts. For example Shh's role in patterning your Central Nervous System as well as your hands. This reuse of toolbox elements in new contexts gives them new functions - depending on the local activator and repressors found there, the signal transduction pathway genes available, as well as epigenetic chromosomal modifications already present. So far we have mainly focused on gradients and organizers in patterning, but in Patterns we will look at a couple of other ways to pattern an embryo.


