7.2: The Electron Transport Chain (ETC)
- Page ID
- 16451
All cells use an electron transport chain (ETC) to oxidize substrates in exergonic reactions. The electron flow from reduced substrates through an ETC is like the movement of electrons between the poles of a battery. In the case of the battery, the electron flow releases free energy to power a motor, light, cell phone, etc. In the mitochondrial ETC, electrons flow when the reduced electron (NADH, FADH2) are oxidized. In plants and other photosynthetic organisms, an ETC serves to oxidize NADPH (a phosphorylated version of the electron carrier NADH). In both cases, free energy released when the redox reactions of an ETC are coupled to the active transport of protons (H+ ions) across a membrane. The result is a chemical gradient of H+ ions as well as a pH gradient. Since protons are charged, the proton gradient is also an electrical gradient. In a kind of shorthand, we say that the free energy once in reduced substrates is now in an electrochemical gradient. That gradient free energy is captured in ATP synthesis reactions coupled to the flow (diffusion) of protons back across the membrane in the process called oxidative phosphorylation. In aerobic respiration, electrons are ultimately transferred from components at the end of the ETC to a final electron acceptor molecular oxygen, O2, making water. In photosynthesis, electron transfer reduces CO2 to sugars.
The Chemiosmotic Mechanism explained how the creation of an electrochemical gradient and how gradient free energy ends up in ATP. For this insight, Peter Mitchell won the Nobel Prize in Chemistry in 1978. You can read Mitchell’s original proposal of the chemiosmosis model of mitochondrial ATP synthesis in Mitchell P (1961) Coupling of phosphorylation to electron and hydrogen transfer by a chemiosmotic type of mechanism. Nature 191:144-148. Here we focus on the details of respiration as it occurs in the mitochondria of eukaryotic cells. The end products of electron transport are NAD+, FAD, water and protons. The protons end up outside the mitochondrial matrix because they are pumped across the cristal membrane using the free energy of electron transport.
Electron transport and oxidative phosphorylation are summarized in the illustration below.
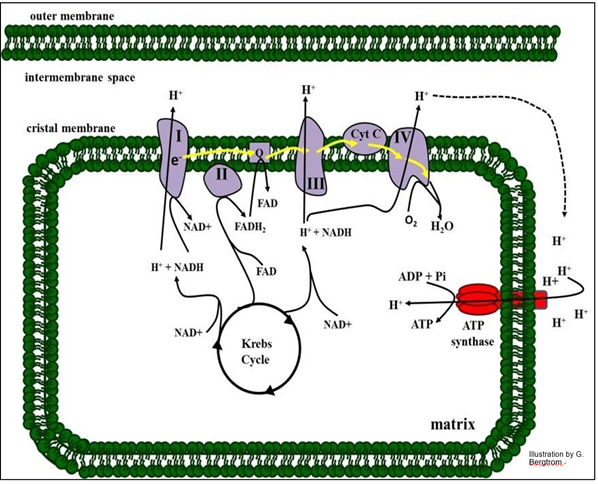
161 Electron Transport Oxidizes Reduced Electron Carriers
162 Finding the Free Energy of Electron Transport
163 Separating Electron Transport from Oxidative Phosphorylation
Roman numbered protein complexes along with Coenzyme Q (just “Q” in the drawing) and cytochrome C (Cyt c) constitute the ETC), the sequence of reactions that oxidize NADH or FADH2 to NAD+ and FAD (respectively). The electrons from these reduced electron carriers are transferred from one ETC complex to the next. At the end of chain, electrons, protons and oxygen unite in complex IV to make water. As you might expect, under standard conditions in a closed system, electron transport is downhill, with an overall release of free energy (negative DGo) at equilibrium.
In the illustration above, we can see three sites in the respiratory ETC that function as H+ pumps. At these sites, the negative change in free energy of electron transfer is large and coupled to the action of a pump. The result is that protons accumulate outside the matrix of the mitochondrion. Because the outer mitochondrial membrane is freely permeable to protons, the electrochemical gradient is in effect between the cytoplasm and the mitochondrial matrix. Proton flow back into the mitochondrial matrix through lollipop- shaped ATP synthase complexes releases the gradient free energy that is harnessed as chemical energy.
164 Proton Pumps Store Free Energy of the ETC in Proton Gradients


