21.5: Biosynthesis of Cholesterol and Steroids
- Page ID
- 15176
\( \newcommand{\vecs}[1]{\overset { \scriptstyle \rightharpoonup} {\mathbf{#1}} } \)
\( \newcommand{\vecd}[1]{\overset{-\!-\!\rightharpoonup}{\vphantom{a}\smash {#1}}} \)
\( \newcommand{\dsum}{\displaystyle\sum\limits} \)
\( \newcommand{\dint}{\displaystyle\int\limits} \)
\( \newcommand{\dlim}{\displaystyle\lim\limits} \)
\( \newcommand{\id}{\mathrm{id}}\) \( \newcommand{\Span}{\mathrm{span}}\)
( \newcommand{\kernel}{\mathrm{null}\,}\) \( \newcommand{\range}{\mathrm{range}\,}\)
\( \newcommand{\RealPart}{\mathrm{Re}}\) \( \newcommand{\ImaginaryPart}{\mathrm{Im}}\)
\( \newcommand{\Argument}{\mathrm{Arg}}\) \( \newcommand{\norm}[1]{\| #1 \|}\)
\( \newcommand{\inner}[2]{\langle #1, #2 \rangle}\)
\( \newcommand{\Span}{\mathrm{span}}\)
\( \newcommand{\id}{\mathrm{id}}\)
\( \newcommand{\Span}{\mathrm{span}}\)
\( \newcommand{\kernel}{\mathrm{null}\,}\)
\( \newcommand{\range}{\mathrm{range}\,}\)
\( \newcommand{\RealPart}{\mathrm{Re}}\)
\( \newcommand{\ImaginaryPart}{\mathrm{Im}}\)
\( \newcommand{\Argument}{\mathrm{Arg}}\)
\( \newcommand{\norm}[1]{\| #1 \|}\)
\( \newcommand{\inner}[2]{\langle #1, #2 \rangle}\)
\( \newcommand{\Span}{\mathrm{span}}\) \( \newcommand{\AA}{\unicode[.8,0]{x212B}}\)
\( \newcommand{\vectorA}[1]{\vec{#1}} % arrow\)
\( \newcommand{\vectorAt}[1]{\vec{\text{#1}}} % arrow\)
\( \newcommand{\vectorB}[1]{\overset { \scriptstyle \rightharpoonup} {\mathbf{#1}} } \)
\( \newcommand{\vectorC}[1]{\textbf{#1}} \)
\( \newcommand{\vectorD}[1]{\overrightarrow{#1}} \)
\( \newcommand{\vectorDt}[1]{\overrightarrow{\text{#1}}} \)
\( \newcommand{\vectE}[1]{\overset{-\!-\!\rightharpoonup}{\vphantom{a}\smash{\mathbf {#1}}}} \)
\( \newcommand{\vecs}[1]{\overset { \scriptstyle \rightharpoonup} {\mathbf{#1}} } \)
\(\newcommand{\longvect}{\overrightarrow}\)
\( \newcommand{\vecd}[1]{\overset{-\!-\!\rightharpoonup}{\vphantom{a}\smash {#1}}} \)
\(\newcommand{\avec}{\mathbf a}\) \(\newcommand{\bvec}{\mathbf b}\) \(\newcommand{\cvec}{\mathbf c}\) \(\newcommand{\dvec}{\mathbf d}\) \(\newcommand{\dtil}{\widetilde{\mathbf d}}\) \(\newcommand{\evec}{\mathbf e}\) \(\newcommand{\fvec}{\mathbf f}\) \(\newcommand{\nvec}{\mathbf n}\) \(\newcommand{\pvec}{\mathbf p}\) \(\newcommand{\qvec}{\mathbf q}\) \(\newcommand{\svec}{\mathbf s}\) \(\newcommand{\tvec}{\mathbf t}\) \(\newcommand{\uvec}{\mathbf u}\) \(\newcommand{\vvec}{\mathbf v}\) \(\newcommand{\wvec}{\mathbf w}\) \(\newcommand{\xvec}{\mathbf x}\) \(\newcommand{\yvec}{\mathbf y}\) \(\newcommand{\zvec}{\mathbf z}\) \(\newcommand{\rvec}{\mathbf r}\) \(\newcommand{\mvec}{\mathbf m}\) \(\newcommand{\zerovec}{\mathbf 0}\) \(\newcommand{\onevec}{\mathbf 1}\) \(\newcommand{\real}{\mathbb R}\) \(\newcommand{\twovec}[2]{\left[\begin{array}{r}#1 \\ #2 \end{array}\right]}\) \(\newcommand{\ctwovec}[2]{\left[\begin{array}{c}#1 \\ #2 \end{array}\right]}\) \(\newcommand{\threevec}[3]{\left[\begin{array}{r}#1 \\ #2 \\ #3 \end{array}\right]}\) \(\newcommand{\cthreevec}[3]{\left[\begin{array}{c}#1 \\ #2 \\ #3 \end{array}\right]}\) \(\newcommand{\fourvec}[4]{\left[\begin{array}{r}#1 \\ #2 \\ #3 \\ #4 \end{array}\right]}\) \(\newcommand{\cfourvec}[4]{\left[\begin{array}{c}#1 \\ #2 \\ #3 \\ #4 \end{array}\right]}\) \(\newcommand{\fivevec}[5]{\left[\begin{array}{r}#1 \\ #2 \\ #3 \\ #4 \\ #5 \\ \end{array}\right]}\) \(\newcommand{\cfivevec}[5]{\left[\begin{array}{c}#1 \\ #2 \\ #3 \\ #4 \\ #5 \\ \end{array}\right]}\) \(\newcommand{\mattwo}[4]{\left[\begin{array}{rr}#1 \amp #2 \\ #3 \amp #4 \\ \end{array}\right]}\) \(\newcommand{\laspan}[1]{\text{Span}\{#1\}}\) \(\newcommand{\bcal}{\cal B}\) \(\newcommand{\ccal}{\cal C}\) \(\newcommand{\scal}{\cal S}\) \(\newcommand{\wcal}{\cal W}\) \(\newcommand{\ecal}{\cal E}\) \(\newcommand{\coords}[2]{\left\{#1\right\}_{#2}}\) \(\newcommand{\gray}[1]{\color{gray}{#1}}\) \(\newcommand{\lgray}[1]{\color{lightgray}{#1}}\) \(\newcommand{\rank}{\operatorname{rank}}\) \(\newcommand{\row}{\text{Row}}\) \(\newcommand{\col}{\text{Col}}\) \(\renewcommand{\row}{\text{Row}}\) \(\newcommand{\nul}{\text{Nul}}\) \(\newcommand{\var}{\text{Var}}\) \(\newcommand{\corr}{\text{corr}}\) \(\newcommand{\len}[1]{\left|#1\right|}\) \(\newcommand{\bbar}{\overline{\bvec}}\) \(\newcommand{\bhat}{\widehat{\bvec}}\) \(\newcommand{\bperp}{\bvec^\perp}\) \(\newcommand{\xhat}{\widehat{\xvec}}\) \(\newcommand{\vhat}{\widehat{\vvec}}\) \(\newcommand{\uhat}{\widehat{\uvec}}\) \(\newcommand{\what}{\widehat{\wvec}}\) \(\newcommand{\Sighat}{\widehat{\Sigma}}\) \(\newcommand{\lt}{<}\) \(\newcommand{\gt}{>}\) \(\newcommand{\amp}{&}\) \(\definecolor{fillinmathshade}{gray}{0.9}\)By William (Bill) W. Christie and Henry Jakubowski.
This section is an abbreviated and modified version of material from the Lipid Web, an introduction to the chemistry and biochemistry of individual lipid classes, written by William Christie.
-
Structural Understanding:
- Describe the molecular structure of cholesterol, including its tetracyclic cyclopenta[a]phenanthrene nucleus, iso‑octyl side chain, and functional groups (e.g., the 3β‑hydroxyl group and the Δ5 double bond).
- Explain how the structure of cholesterol relates to its orientation and interaction within the lipid bilayer (e.g., using the diamond lattice model to illustrate atomic positions and interactions).
-
Membrane Organization and Function:
- Analyze the distribution of cholesterol among various cellular membranes (plasma membrane, endoplasmic reticulum, Golgi apparatus, mitochondria) and explain the functional significance of its uneven distribution.
- Explain how cholesterol modulates membrane fluidity, thickness, and permeability by interacting with phospholipids (e.g., phosphatidylcholine, sphingomyelin) and how these interactions lead to the formation of ordered microdomains (rafts).
-
Biosynthesis of Cholesterol:
- Outline the cholesterol biosynthetic pathway from acetyl-CoA via the mevalonate pathway, highlighting key intermediates (e.g., HMG‑CoA, mevalonate, squalene, lanosterol) and the role of critical enzymes such as HMG‑CoA reductase.
- Discuss the energetic cost and oxygen requirements of cholesterol synthesis, and describe how this process is tightly regulated (including feedback by sterol levels and regulation via SREBP and SCAP).
-
Cholesterol Esterification and Transport:
- Explain the biosynthesis and hydrolysis of cholesterol esters, including the roles of enzymes such as lecithin:cholesterol acyltransferase (LCAT) and acyl-CoA:cholesterol acyltransferase (ACAT).
- Describe how cholesterol esters are packaged into lipoproteins (e.g., HDL, LDL, VLDL) and the importance of these processes in cholesterol transport and storage.
-
Regulation and Homeostasis:
- Discuss the mechanisms that maintain cholesterol homeostasis in cells, including the roles of intracellular cholesterol pools, feedback inhibition of biosynthetic enzymes, and the regulation of cholesterol uptake (via LDL receptors) and efflux (via ABC transporters).
- Evaluate the role of oxysterols (oxygenated cholesterol derivatives) in modulating cholesterol synthesis and immune responses through nuclear receptor activation (e.g., LXR).
-
Pathophysiological Implications:
- Analyze how disruptions in cholesterol metabolism contribute to human diseases, such as cardiovascular disease, atherosclerosis, gallstone formation, and various neurodegenerative disorders.
- Assess the impact of pharmacological agents (e.g., statins) on cholesterol biosynthesis and discuss current and emerging therapeutic targets within the cholesterol metabolic pathway.
-
Comparative and Integrated Perspectives:
- Compare cholesterol metabolism in animals with that of plant sterols (phytosterols), noting both similarities and key differences in structure, biosynthesis, and functional roles.
- Integrate knowledge of cholesterol structure, synthesis, regulation, and transport to predict how alterations in these processes may influence cell signaling, membrane dynamics, and overall human health.
These learning goals aim to ensure that students can critically analyze the multifaceted roles of cholesterol and its derivatives in cellular biochemistry and understand their broader implications for human physiology and disease.
Sterols: Cholesterol and Cholesterol Esters
In animal tissues, cholesterol (cholest-5-en-3β-ol) is by far the most abundant member of a family of polycyclic compounds known as sterols. It can also be described as a polyisoprenoid or a triterpene from its biosynthetic origin. Cholesterol was first recognized as a component of gallstones as early as 1769, while the great French lipid chemist Michel-Eugène Chevreul isolated it from animal fats in 1815. However, it was not until well into the 20th century that the structure was fully defined by the German Chemist Heinrich Wieland, who received the Nobel Prize in Chemistry for his work in 1927, the first of thirteen so honored for research on cholesterol and its metabolism.
Cholesterol plays a vital role in animal life, and it is essential for the normal functioning of cells both as a structural component of cell membranes and as a precursor of steroid hormones and other key metabolites, including vitamin D and bile acids. It is also important for cell signaling, transport processes, nerve conduction, and the regulation of gene transcription. Every cell in vertebrates is capable of both synthesizing and metabolizing cholesterol, and there is evidence that de novo synthesis is essential, regardless of dietary intake; this process is particularly vital in the brain. However, excess cholesterol can contribute to the pathology of various diseases, notably cardiovascular disease, so cholesterol levels must be balanced to ensure an adequate but not excessive supply.
Cholesterol – Structure, Occurrence, and Function in Membranes
The structure of cholesterol is shown below in Figure \(\PageIndex{1}\).
Cholesterol consists of a tetracyclic cyclopenta[a]phenanthrene structure with an iso-octyl side-chain at carbon 17. The four rings (A, B, C, D) have trans ring junctions, and the side chain and two methyl groups (C-18 and C-19) are at an angle to the rings above the plane with β stereochemistry (as for the hydroxyl group on C-3 also); there is a double bond between carbons 5 and 6. Thus, the molecule has a rigid planar four-ring nucleus with a flexible tail. Of the two recognized numbering systems in use, one originally described by Fieser and Fieser in 1959 and a second by IUPAC-IUB in 1989, the first appears to be preferred by most current authors.
Most of the atoms in cholesterol can be placed on the diamond lattice, which is the structure showing the position of each carbon atom in the network solid diamond. In that structure, each carbon is connected to four other carbons using sp3-hybridized atomic orbitals and a tetrahedral geometry. A representation of the diamond lattice is shown below. It consists of a series of interconnected boat conformations of cyclohexane propagating in the xyz direction. The structures of two different fused cyclohexanes, trans- and cis-decalins, as well as the structure of three fused cyclohexanes, adamantane, are shown in Figure \(\PageIndex{2}\). Superimposing sp3-connected atoms onto a diamond lattice allows improved visualization of the real and allowed structures of more complicated molecules.
Figure \(\PageIndex{3}\) shows a superposition of 5-α-cholestane, a reduced and non-hydroxylated form of cholesterol, onto the diamond lattice. The pink cyclopentane D ring of 5-α-cholestane, which is distorted from the 1090 bond angle for sp3-hybridized carbon atoms, does not fit exactly onto the lattice. Cholesterol, with its double bond, would also deviate from the ideal position on the diamond lattice, given the two sp2-hybridized carbons in the double bond.
Cholesterol is a ubiquitous component of all animal tissues (and of some fungi), produced by every nucleated animal cell, where much of it is located in the membranes, although it is not evenly distributed. The highest proportion of unesterified cholesterol is found in the plasma membrane, accounting for roughly 30-50% of the lipid in the membrane or 60-80% of the cholesterol in the cell. In comparison, mitochondria and the endoplasmic reticulum contain significantly less (~5% in the latter), while the Golgi contains an intermediate amount. Cholesterol is also enriched in early and recycling endosomes, but not in late endosomes. It may surprise some to learn that the brain contains more cholesterol than any other organ, comprising roughly a quarter of the total free cholesterol in the human body, with 70-80% of this cholesterol found in the myelin sheath. Of all the organic constituents of blood, only glucose is present in a higher molar concentration than cholesterol. In animal tissues, it occurs in the free form, esterified to long-chain fatty acids (such as cholesterol esters), and in other covalent and non-covalent linkages, including associations with plasma lipoproteins. In plants, it tends to be a minor component only of a complex mixture of structurally related 'phytosterols'. However, there are exceptions; nevertheless, it is important as a precursor to some plant hormones.
Animals in general synthesize a high proportion of their cholesterol requirement, but they can also ingest and absorb appreciable amounts from foods. On the other hand, many invertebrates, including insects, crustaceans, and some molluscs, are unable to synthesize cholesterol and must obtain it from their diet; for example, spiny lobsters must obtain exogenous cholesterol to produce essential sex hormones. Similarly, it must be supplied from exogenous sources to the primitive nematode Caenorhabditis elegans, where it does not appear to have a major role in membrane structure, other than perhaps in the function of ion channels, although it is essential for the production of steroidal hormones required for larval development; the novel lipid phosphoethanolamine glucosylceramide regulates its uptake. Some species can convert dietary plant sterols, such as β-sitosterol, into cholesterol. Prokaryotes lack cholesterol entirely except for some pathogens that acquire it from eukaryotic hosts to ensure their intracellular survival (e.g., Borrelia sp.); bacterial hopanoids are often considered to be sterol surrogates.
Cholesterol plays vital structural roles in membranes and lipid metabolism, with an extraordinary diversity of biological functions, including cell signaling, morphogenesis, lipid digestion and absorption in the intestines, reproduction, stress responses, sodium and water balance, and calcium and phosphorus metabolism. We can only touch on a few of these functions in this web page. It is a biosynthetic precursor of bile acids, vitamin D, and steroid hormones (glucocorticoids, estrogens, progesterones, androgens, and aldosterone), and it is found in covalent linkage to specific membrane proteins or proteolipids ('hedgehog' proteins), which have vital functions in embryonic development. Additionally, it plays a significant role in the development and functioning of the central nervous system. On the other hand, excess cholesterol in cells can be toxic, and a complex web of enzymes is essential to maintain the optimum concentrations. Because plasma cholesterol levels can be a major contributory factor to atherogenesis, media coverage has created what has been termed a ‘cholesterophobia’ in the population at large.
One of the primary functions of cholesterol is to regulate the fluidity of membranes by interacting with their complex lipid components, specifically phospholipids such as phosphatidylcholine and sphingomyelin. As an amphiphilic molecule, cholesterol can intercalate between phospholipids in lipid bilayers to span about half a bilayer. In its three-dimensional structure, it is, in essence, a planar molecule that can interact on both sides. The tetracyclic ring structure is compact and very rigid. Additionally, the location of the hydroxyl group facilitates the orientation of the molecule within a membrane bilayer. The positions of the methyl groups appear to maximize interactions with other lipid constituents. The structure of cholesterol as it would appear on the diamond lattice is shown in Figure \(\PageIndex{4}\).
As the α-face of the cholesterol nucleus (facing down) is 'smooth', it can make good contact with the saturated fatty-acyl chains of phospholipids down to about their tenth methylene group; the β-face (facing up) is made 'rough' by the projection of methyl groups from carbons 10 and 13. The interaction is primarily mediated by van der Waals and hydrophobic interactions, with a contribution from hydrogen bonding between the cholesterol hydroxyl group and the polar head group and interfacial regions of the phospholipids, especially sphingomyelin. Intercalated cholesterol may also disrupt electrostatic interactions between the ionic phosphocholine head groups of nearby membrane phospholipids, leading to increased mobility of the head groups. Indeed, there is evidence that cholesterol forms stoichiometric complexes with the saturated fatty acyl groups of sphingomyelin and, to a lesser extent, phosphatidylcholine.
Experiments with mutant cell lines and specific inhibitors of cholesterol biosynthesis suggest that an equatorial hydroxyl group at C-3 of sterols is essential for the growth of mammalian cells. The Δ5 double bond ensures that the molecule adopts a planar conformation, and this feature also appears to be essential for cell growth, as is the flexible iso-octyl side-chain. The C-18 methyl group is crucial for the proper orientation of the sterol. While plant sterols appear to be able to substitute for cholesterol in supporting many of the bulk properties of membranes in mammalian cells in vitro, cholesterol is essential for other purposes.
In the absence of cholesterol, a membrane composed of unsaturated lipids is in a fluid state that is characterized by a substantial degree of lipid chain disorder, i.e., it constitutes a liquid-disordered phase. The function of cholesterol is to increase the degree of order (cohesion and packing) in membranes, leading to the formation of a liquid-ordered phase. In contrast, it renders bilayers composed of more saturated lipids, which would otherwise be in a solid gel state, more fluid. Thus, cholesterol can promote and stabilize a liquid-ordered phase over a substantial range of temperatures and sterol concentrations. Further, high cholesterol concentrations in membranes reduce their passive permeability to solutes. These effects enable membranes to bend or withstand mechanical stresses, and they permit the fine-tuning of membrane lipid composition and organization, and regulate critical cell functions. Simplistically, the higher cholesterol concentrations in the plasma membrane support its barrier function by increasing membrane thickness and reducing its permeability to small molecules. In contrast, the endoplasmic reticulum has increased membrane flexibility due to its lower cholesterol concentration, which enables the insertion and folding of proteins into its lipid bilayer. While mitochondrial membranes have a low cholesterol content overall, this may be concentrated in nanodomains at regions of high curvature in the inner mitochondrial membrane, with links to nucleoprotein complexes (nucleoids).
In comparison to other lipids, it has been reported that cholesterol can flip rapidly between the leaflets in a bilayer; however, this is not universally accepted, leading to doubts about the trans-bilayer distribution of cholesterol in some biological membranes. However, much recent evidence suggests that the concentration of cholesterol in the inner leaflet of the plasma membrane is much lower than that in the outer leaflet in a range of mammalian cells. This distribution is significant because cholesterol promotes negative curvature of membranes and may be a crucial factor in facilitating membrane fusion during the process of exocytosis. It may also be relevant for regulating various cellular signaling processes at the plasma membrane.
Cholesterol also plays a key role in the lateral organization of membranes and their free volume distribution, which allows for more intimate protein-cholesterol interactions that may regulate the activities of membrane proteins. Many membrane proteins bind strongly to cholesterol, including some that are involved in cellular cholesterol homeostasis or trafficking, and contain a conserved region termed the ‘sterol-sensing domain’. Some proteins bind to cholesterol deep within the hydrophobic core of the membrane via binding sites on the membrane-spanning surfaces or in cavities or pores in the proteins, driven by hydrogen bond formation. Cholesterol has an intimate interaction with G-protein-coupled receptors (GPCRs) to affect ligand binding and activation, either by direct high-affinity binding to the receptor, by changing their oligomerization state, or by inducing changes in the properties of the membrane. For example, it is essential for the stability and function of the β2-adrenergic, oxytocin, and serotonin receptors by increasing the agonist affinities. The inactive state of rhodopsin is stabilized both through indirect effects on plasma membrane curvature and by a direct interaction between lipid and protein. The GPCR neurotransmitter serotonin 1A receptor has ten closely bound cholesterol molecules, which control its organization and positioning. The receptor senses membrane cholesterol via a lysine residue in a so-called 'CRAC' motif in transmembrane helix 2.
Ion pumps, such as the (Na+-K+)-ATPase, which have specific binding sites for cholesterol molecules, are the single most important consumers of ATP in cells and are responsible for the ion gradients across membranes that are essential for many cellular functions; depletion of cholesterol in the plasma membrane deactivates these ion pumps. In the brain, in addition to being essential for the structure of the myelin sheath, cholesterol is a major component of synaptic vesicles and controls their shape and functional properties. In the nucleus of cells, cholesterol plays an important role in the structure and function of chromatin.
The role of cholesterol, together with sphingolipids, in the formation of transient membrane nano-domains known as rafts is crucial for cell function, while the interaction of cholesterol with ceramides is essential for the skin's barrier function.
Cholesterol Biosynthesis
Cholesterol biosynthesis involves a highly complex series of at least thirty different enzymatic reactions, which were largely unraveled by Konrad Bloch and Fyodor Lynen, who received the Nobel Prize for their work on the topic in 1964. When the various regulatory, transport, and genetic studies of recent years are taken into account, it is clear that this subject cannot be treated in depth here. The bare bones of the mechanistic aspects are therefore delineated, which should serve as a guide for further study. In plants, cholesterol synthesis occurs by a somewhat different pathway with cycloartenol as the key intermediate. We will explore the reaction mechanisms of several enzymes on this complex pathway, given its medical importance.
Almost all nucleated cells can synthesize their full complement of cholesterol. The first steps involve the synthesis of the important intermediate mevalonic acid from acetyl-CoA and acetoacetyl-CoA, both of which are derived from acetate, in two enzymatic steps. These precursors are located in the cytosol, as is the first enzyme, 3-hydroxy-3-methylglutaryl (HMG)-CoA synthase. The second enzyme, HMG-CoA reductase, is a particularly important control point and is widely regarded as the rate-limiting step in the overall synthesis of sterols. Its activity is regulated at the transcriptional level and by several additional factors, including a cycle of phosphorylation and dephosphorylation. This and subsequent enzymes are membrane-bound and are located in the endoplasmic reticulum. The enzyme HMG-CoA reductase is among the targets inhibited by the drugs known as ‘statins’, so that patients must then obtain much of their cholesterol from the diet. The first two reactions in the synthesis of cholesterol are shown in Figure \(\PageIndex{5}\).
HMG-CoA Synthase
We saw this reaction in the synthesis of ketone bodies in Chapter 17. This enzyme catalyzes the condensation of acetoacetyl-CoA–CoA (AcAc–CoA) and acetyl–CoA (Ac-CoA) to form 3-hydroxy-3-methylglutaryl (HMG)–CoA, and requires the formation of a C-C bond. HMG-CoA synthase forms a C-C bond by activating the methyl group of acetyl-cysteine. The acetyl group originates from an acyl-CoA donor. The enzyme catalyzes the first committed step in the formation of complex isoprenoids, such as cholesterol and ketone bodies. The product, 3-hydroxy-3-methylglutaryl HMG–CoA, can either be reduced by HMG-CoA reductase to form mevalonate, which leads to cholesterol synthesis, or cleaved by the enzyme HMG-CoA lyase to produce acetoacetate, a ketone body.
HMG-CoA synthase catalyzes a bisubstrate reaction that displays ping-pong kinetics, characteristic of a covalent enzyme intermediate. The first substrate binds to the enzyme and transfers an acetyl group to the nucleophilic Cys 111 in the active site, forming an acetyl-Cys 111 intermediate. Free CoA departs. Next, the second substrate, acetoacetyl-CoA, binds and condenses with the acetyl group donated by acetyl-Cys 111. This condensation involves an enolate. A plausible reaction mechanism is shown in Figure \(\PageIndex{6}\).
Figure \(\PageIndex{6}\): Reaction mechanism for HMG-CoA synthase
Hence, there are three parts of the reaction: acetylation/deacetylation, condensation/cleavage with an enolate intermediate, and C-C formation and hydrolysis/dehydration. Figure \(\PageIndex{7}\) shows an interactive iCn3D model of the Staphylococcus aureus HMG-COA Synthase with bound HMG-CoA and acetoacetyl-CoA (1XPK).
Figure \(\PageIndex{7}\): Staphylococcus aureus HMG-COA Synthase with bound HMG-CoA and acetoacetyl-CoA (1XPK). (Copyright; author via source). Click the image for a popup or use this external link: https://structure.ncbi.nlm.nih.gov/i...deiFs7JceE5H76
The biologically active unit (homodimer) is shown, with each monomer shown in a different color. The A chain (light cyan) has bound HMG-CoA (HMG) while the B chain (light gold) has acetoacetyl-CoA (CAA) bound. The Glu 79, Cys 111, and His 233 in each subunit are shown in CPK sticks and labeled. Note that Cys 111 is covalently modified in each subunit.
Figure \(\PageIndex{8}\) shows an interactive iCn3D model of the human 3-hydroxy-3-methylglutaryl CoA synthase I with bound CoASH (2P8U)
Figure \(\PageIndex{8}\): Human 3-hydroxy-3-methylglutaryl CoA synthase I (monomer) with bound CoASH (2P8U). (Copyright; author via source). Click the image for a popup or use this external link: https://structure.ncbi.nlm.nih.gov/i...xxEoUfecTk3eL6
The active site side chains are numbered differently (Glu 95, Cys 129, and His 264) compared to the S. aureus protein. Only the monomer is shown in this model.
HMG-CoA Reductase
In this key rate-limiting step that commits HMG to the sterol and isoprenoid synthetic pathways, HMG is converted to mevalonate. A reducing agent, such as NADPH, is required for this biosynthetic reaction. A plausible mechanism is shown in Figure \(\PageIndex{9}\).
Figure \(\PageIndex{10}\) shows an interactive iCn3D model of the catalytic domain of human HMG-CoA reductase with bound HMG-CoA (1DQ9).
Figure \(\PageIndex{10}\): Catalytic domain of human HMG-CoA reductase with bound HMG-CoA (1DQ9). (Copyright; author via source). Click the image for a popup or use this external link: https://structure.ncbi.nlm.nih.gov/i...dZvWzTocKhUzt8
Two of the four identical monomers in the biological unit, the C (gray) and D (cyan) chains, are shown. HMG-CoA is shown in spacefill and CPK colors. Three key catalytic residues, His 866 in the C chain (b chain in the mechanism above) and Lys 691 and Asp 767 in the D chain (A chain in the mechanism above), are shown in sticks, CPK colors, and labeled.
Synthesis of 5-isopentenyl and 2-isopentenyl pyrophosphate
The next sequence of reactions involves first the phosphorylation of mevalonic acid by a mevalonate kinase to form the 5‑monophosphate ester, followed by further phosphorylation to yield an unstable pyrophosphate, which is rapidly decarboxylated to produce 5-isopentenyl pyrophosphoric acid, the universal isoprene unit. An isomerase converts part of the latter to 3,3-dimethylallyl pyrophosphoric acid. These reactions are shown in Figure \(\PageIndex{11}\).
Two phosphorylations are required, one by mevalonate kinase, which proceeds by an ordered sequential binding of mevalonate as the first reactant and its phosphomevalonate as the first product released. The enzyme is inhibited by two downstream products of the reaction pathway, farnesyl pyrophosphate and geranyl pyrophosphate. A mechanism for mevalonate kinase is shown in Figure \(\PageIndex{12}\).
A reaction mechanism for the second kinase, phosphomevalonate kinase, showing progression through the transition state, is shown in Figure \(\PageIndex{13}\). The reaction proceeds through direct phosphorylation through a dissociative mechanism.
Mevalonate diphosphate decarboxylase
This enzyme catalyzes the decarboxylation of 5-pyrophosphate mevalonate to 5-isopentenylpyrophosphate as shown in Figure \(\PageIndex{14}\).
The binding of the two substrates, pyrophosphatemevalonate (or mevalonate pyrophosphate - MVAPP) and ATP to the enzyme (A), and a reaction mechanism (B, are shown in Figure \(\PageIndex{15}\).
Panel (a) shows changes in the enzyme structure upon substrate binding. The apo-enzyme is shown on the left. The middle structure shows the enzyme after binding of the first substrate, MVAPP. A key loop (β10-α4) is shown as a cyan surface. The enzyme is then shown in an open conformation with the second substrate, ATP, also bound. An additional phosphate-binding loop is shown in magenta. At the far right, the enzyme is shown in a closed conformation, resulting from conformational changes in both loops, which trap substrates in the active site. These changes enable catalysis. Product release follows.
Panel (b) shows the dissociative phosphoryl transfer catalytic mechanism. At the top left, D282 is shown interacting with the 3′-OH group of MVAPP (red). The top right shows a dissociative phosphoryl (blue) transition state. In the bottom left, the phosphate attaches to the 3′ oxygen (red) of MVAPP. The bottom right shows the products after dephosphorylation and decarboxylation, which produce IPP, ADP, phosphate, and CO2. K187 from the β10-α4 loop and metal ions in the active site are involved in neutralizing the negatively charged environment and assist in catalysis.
Figure \(\PageIndex{16}\) shows an interactive iCn3D model of mevalonate diphosphate decarboxylase with mevalonate-5-diphosphate, AMPPCP, and Magnesium (6E2U).
Figure \(\PageIndex{16}\): Mevalonate diphosphate decarboxylase with mevalonate-5-diphosphate, AMPPCP and Magnesium (6E2U) . (Copyright; author via source). Click the image for a popup or use this external link: https://structure.ncbi.nlm.nih.gov/i...SukmP1BTyNhH26
The correctly positioned substrates interact with two Mg 2+ ions in the initial steps of the reaction. The conserved lysine facilitates the phosphoryl transfer.
Polymerization of isoprene
Isoprene, a small branched alkadiene, can polymerize into larger molecules containing isoprene monomer to form isoprenoids, often called terpenes. Instead of using isoprene as the polymerization monomer, either dimethylallyl pyrophosphate (DMAPP) or isopentenyl pyrophosphate (IPP) is used biologically.
Figure \(\PageIndex{x}\) below shows how DMAPP and IPP (both containing 5Cs) are used in a polymerization reaction to form geranyl-pyrophosphate (C10), farnesyl pyrophosphate (C15), and geranyl-geranyl pyrophosphate (C20). Figure \(\PageIndex{17}\):
The condensation of IPP and DMAPP is a head-to-tail condensation reaction. Another IPP reacts with geranylpyrophosphate using the same enzyme to produce farnesylpyrophosphate. DMAPP is first formed by the isomerization of IPP to DMAPP catalyzed by isopentenyl-diphosphate delta-isomerase. It catalyzes the 1,3-allylic rearrangement of the homoallylic substrate isopentenyl (IPP). 5-isopentenyl pyrophosphate is a nucleophile, but its isomer, DMAPP, is highly electrophilic, which promotes the condensation of the two molecules.
A mechanism for the next reaction, the first condensation of DMAPP and IPP to form geranylpyrophosphate, catalyzed by farnesyl pyrophosphate (diphosphate) synthase, is shown in Figure \(\PageIndex{18}\).
The reaction appears to proceed using a carbocation transition state, followed by the transfer of a hydrogen atom (not an ion) from IPP to pyrophosphate. Figure \(\PageIndex{19}\) shows an interactive iCn3D model of E. Coli farnesyl pyrophosphate synthase bound to isopentyl pyrophosphate and dimethylallyl S-thiolodiphosphate (1RQI).
Figure \(\PageIndex{19}\): Farnesyl pyrophosphate synthase Bound to isopentyl pyrophosphate (IPP) and dimethylallyl S-thiolodiphosphate (1RQI). (Copyright; author via source). Click the image for a popup or use this external link: https://structure.ncbi.nlm.nih.gov/i...kQBU5dNnKrCVt6
DST in the structure is dimethylallyl S-thiolodiphosphate, an analog of dimethylallyl diphosphate (DMAPP). Key conserved amino acids involved in substrate binding, transition state stabilization, and catalysis are shown as sticks, CPK colors. Thr203, Gln241, and Lys202 presumably stabilized the carbocation intermediate/transition state. Lys202 also forms a hydrogen bond with DMAPP. Two arginines (69, 116) form salt bridges with the pyrophosphates of IPP and DMAPP.
Many isoprenoid lipids are made from farnesyl pyrophosphate. For membrane purposes, the most important of these is cholesterol. Figure \(\PageIndex{20}\) shows an overview of the synthesis of cholesterol from two farnesyl pyrophosphates linking together in a "tail-to-tail" reaction to form squalene, a precursor of cholesterol. Each isoprene unit (5Cs) is shown in different colors to facilitate easier visualization.
Figure \(\PageIndex{20}\): Synthesis of squalene from isoprene units.
Figure \(\PageIndex{21}\) shows reactants (two farnesyl pyrophosphate), intermediate (presqualene diphosphate), and product (squalene) in the reaction catalyzed by squalene synthase.
In the squalene synthase reaction, two molecules of farnesyl pyrophosphate condense to yield presqualene pyrophosphate. In turn, this is reduced by NADPH to produce the key intermediate squalene. The enzyme squalene synthase, which regulates the flow of metabolites into either the sterol or non-sterol pathways, with farnesyl pyrophosphate as the branch point, is considered the first committed enzyme in cholesterol biosynthesis.
Given the importance of this reaction, we will explore the unique mechanism of squalene synthase in some detail.
Part 1: Formation of the cyclopropyl presqualene intermediate
Figure \(\PageIndex{22}\) illustrates the mechanism for the first part of the reaction, which leads to the formation of the cyclopropyl intermediate, presqualene. The reaction proceeds through a series of carbocation intermediates.
Part 2: Conversion of cyclopropyl intermediate to squalene - reduction
The next part of the reaction involves the reductive formation of squalene, as shown in Figure \(\PageIndex{23}\).
Figure \(\PageIndex{24}\) shows an interactive iCn3D model of human squalene synthase with bound inhibitor (1EZF).
Figure \(\PageIndex{24}\): Human squalene synthase with bound inhibitor (1EZF). (Copyright; author via source). Click the image for a popup or use this external link: https://structure.ncbi.nlm.nih.gov/i...jLreHkvbftoFPA
Tyr 171 acts as a general acid/base in the first half of the reaction. Arg 218 and 228 stabilize the diphosphate in the transition state as it leaves. Phe 288 stabilizes the reactive carbocation.
The enzyme has a single domain that contains a large channel on one face, which extends from a solvent-exposed site to the hydrophobic interior. Two FPPs bind at the beginning of the channel, where key side chains for the first reaction are located. The cyclopropyl intermediate then moves into the hydrophobic end of the channel, where it reacts in the second half of the reaction without exposure to water.
In the next crucial step, squalene is oxidized by squalene monooxygenase to form squalene 2,3-epoxide, a key control point in the cholesterol synthesis pathway. This introduces the oxygen atom to squalene, which becomes the signature oxygen of the hydroxyl group in cholesterol. The epoxide then undergoes cyclization catalyzed by the enzyme squalene epoxide lanosterol-cyclase to form the first steroidal intermediate, lanosterol (or cycloartenol en route to phytosterols in photosynthetic organisms). This is illustrated in Figure \(\PageIndex{26}\).
In this remarkable reaction, there is a series of concerted 1,2-methyl group and hydride shifts along the chain of the squalene molecule to bring about the formation of the four rings. No intermediate compounds have been found. This is believed to be one of the most complex single enzymatic reactions ever identified, despite the enzyme involved being only 90 kDa in size. Again, the reaction takes place in the endoplasmic reticulum. However, a cytosolic protein, sterol carrier protein 1, is required to bind squalene in an appropriate orientation in the presence of the cofactors NADPH, flavin adenine dinucleotide (FAD), and O2; the presence of phosphatidylserine promotes the reactions.
The ring closure reaction, initiated by the epoxide, involves a concerted flow of electrons from a source to the epoxide oxygen atom, which acts as an electron sink. This brings to mind a reaction familiar to all students who have studied chemistry: the reaction of the pH indicator phenolphthalein with a base, which produces a pink-colored solution. That reaction, which produces a more conjugated molecule that absorbs at 553 nm (green), results in a magenta solution color and is illustrated for comparison (and fun) in Figure \(\PageIndex{27}\).
In subsequent steps, lanosterol is converted to cholesterol by a series of demethylations, desaturations, isomerizations, and reductions, involving nineteen separate reactions as illustrated in Figure \(\PageIndex{28}\).
Thus, demethylation reactions produce zymosterol as an intermediate, and this is converted to cholesterol via a series of intermediates, all of which have been characterized, and by at least two pathways that utilize essentially the same enzymatic machinery but differ in the order of the various reactions, mainly at the point at which the Δ24 double bond is reduced. Desmosterol is the key intermediate in the so-called 'Bloch' pathway, while 7‑dehydrocholesterol is the immediate precursor in the 'Kandutsch-Russell' pathway. While some tissues, such as adrenal glands and testis, use the Bloch pathway mainly, the brain synthesizes much of its cholesterol by the 'Kandutsch-Russell' pathway. This may enable the production of a variety of other minor sterols for specific biological purposes in different cell types and locations.
The energy cost of the synthesis of one cholesterol molecule is roughly one hundred ATP equivalents, and eleven oxygen molecules are required. Synthesis occurs mainly in the liver, although the brain, peripheral nervous system, and skin also synthesize their own considerable supplies. Cholesterol is exported from the liver and transported to other tissues in the form of low-density lipoproteins (LDL) for uptake via specific receptors. In animals, cells can obtain the cholesterol they require either from the diet via circulating LDL or synthesize it themselves, as outlined above. Cholesterol biosynthesis is highly regulated, with rates of synthesis varying over hundreds of fold depending on the availability of any external sources of cholesterol, and cholesterol homeostasis requires the actions of a complex web of enzymes, transport proteins, and membrane-bound transcription factors, as discussed below.
Click the link below.
- Link
-
In humans, only about a third of the cholesterol is of dietary origin (mainly eggs and red meat), the remainder is produced by synthesis de novo in the endoplasmic reticulum. The latter must be tightly regulated as it is an energetically expensive process that requires appreciable amounts of acetyl-CoA, ATP, oxygen, and the reducing factors NADPH and NADH, especially since cholesterol cannot be catabolized for energy purposes (see below).
Many factors contribute to maintaining the significant differences in cholesterol concentrations among various membranes and organelles within cells, all within precise limits. To explain how cholesterol in the plasma membrane, where it is most abundant, can regulate cholesterol biosynthesis and uptake through enzymes in the endoplasmic reticulum, where it is least abundant, it has been suggested that a key to the process is that there are three pools of cholesterol in the plasma membrane with distinct functional roles. The first of these is “accessible” to receptor proteins for transport to the endoplasmic reticulum. In contrast, the second pool is sequestered by sphingomyelin and can be released by the action of sphingomyelinase if required. The third residual pool of cholesterol is essential for plasma membrane integrity. These correspond to about 16, 15, and 12 mol % of total plasma membrane lipids, respectively, in cholesterol-replete cells. Simplistically, when cholesterol in the plasma membrane is in excess for any reason, e.g., after LDL uptake by receptor-mediated endocytosis, there is a rise in accessible cholesterol, which is then transported to the endoplasmic reticulum to switch off cholesterol biosynthesis and expression of the LDL receptor. This process requires a host of regulatory proteins and mechanisms that can involve either vesicle formation or non-vesicular pathways, utilizing specific transport proteins, such as the ABC transporters.
Ultimately, post-translational control of the many different enzymes involved provides a rapid means for modifying flux through the biosynthetic pathway in the endoplasmic reticulum; some are rapidly degraded in response to tissue levels of cholesterol and its intermediates, while others have their activity altered through phosphorylation or acetylation mechanisms. For example, the second rate-limiting enzyme in cholesterol biosynthesis is squalene monooxygenase, which undergoes cholesterol-dependent proteasomal degradation when cholesterol is in excess. This degradation is guided by a 12-amino-acid hydrophobic sequence on the enzyme that can serve as a degradation signal. When the cholesterol concentration in the endoplasmic reticulum is high, the degradation sequence detaches from the membrane and is exposed, providing a signal for the enzyme to be degraded. Similarly, HMG-CoA reductase is recognized as the key enzyme in regulating cholesterol biosynthesis, and this process can be controlled by a feedback mechanism involving ubiquitin–proteasomal degradation. Further regulation of cholesterol biosynthesis is exerted by sterol intermediates in cholesterol biosynthesis, such as lanosterol and 24,25‑dehydrolanosterol (dimethyl-sterols), by accelerating the degradation of the biosynthetic enzymes, such as HMG-CoA reductase. It is noteworthy that ceramide downregulates cholesterol synthesis – another link between cholesterol and sphingolipid metabolism.
The regulatory element-binding proteins (mainly SREBP-1c and SREBP-2), which contain an N-terminal membrane domain and a C-terminal regulatory domain, play a crucial role in maintaining cholesterol levels. Each is synthesized as an inactive precursor that is inserted into the endoplasmic reticulum, where it can encounter an escort protein termed SREBP cleavage-activating protein (SCAP), which is the cellular cholesterol sensor. When the latter recognizes that cellular cholesterol levels are inadequate, it binds to the regulatory domain of SREBP. The SCAP-SREBP complex then moves to the Golgi, where two specific proteases (designated site-1 and site-2 proteases) cleave the SREBP, enabling the C-terminal regulatory domain to enter the nucleus. There, it activates transcription factors, such as the nuclear liver X receptor (LXR), which stimulate the expression of the genes coding for the LDL receptor in the plasma membrane and for the key enzyme in cholesterol biosynthesis, HMG-CoA reductase. This, in turn, stimulates the rate of cholesterol uptake and synthesis. Conversely, when cholesterol in the endoplasmic reticulum exceeds a threshold, it binds to SCAP in a manner that prevents the SCAP-SREBP complex from leaving the membrane for the nucleus. As a result, cholesterol synthesis and uptake are repressed, and cholesterol homeostasis is restored. In effect, cholesterol exerts feedback inhibition by suppressing its own production by preventing the proteolytic cleavage and maturation of SREBP-2. Oxysterols, especially 25-hydroxycholesterol, are also inhibitors of this process.
Cholesterol in the endoplasmic reticulum is transferred to the Golgi and eventually to the plasma membrane by vesicular and non-vesicular transport mechanisms involving, in part, soluble sterol transport proteins, including the so-called 'START' domain proteins, and partly by binding to those proteins that are intimately involved in the transport and metabolism of polyphosphoinositides such as phosphatidylinositol 4-phosphate (PI(4)P). In the latter mechanism, cholesterol is transported by binding to the ORD domain of oxysterol binding protein (OSBP) or Osh4 in yeast, before OSBP binds to PI(4)P in the plasma membrane to transfer its cargo. The key to this process is that cholesterol and PI(4)P are synthesized at two different locations: the endoplasmic reticulum for sterols and the trans-Golgi network and plasma membrane for PI(4)P. Therefore, the two lipids do not compete; instead, they can be exchanged. OSBP carries cholesterol in the forward direction to the trans-Golgi network and plasma membrane, and PI(4)P, which binds to a C-terminal PH domain in the protein, in the reverse direction. The subsequent hydrolysis of PI(4)P is the energy source for the reaction, and indeed PI(4)P has been termed "lipid ATP". As this reaction is irreversible, a gradient of cholesterol is established along the organelles of the secretory pathway. OSBP is thus a lipid transfer protein that enables two organelles to exchange cholesterol rapidly between them at membrane contact sites in a cycle of reactions involving membrane tethering, cholesterol transport, PI(4)P countertransport, and PI(4)P hydrolysis. A similar mechanism is involved in the transport of phosphatidylserine from the endoplasmic reticulum to the inner leaflet of the plasma membrane.
Subsequently, the ATP-binding cassette (ABC) transporters ABCA1 and ABCG1, located in the plasma membrane and containing a significant portion of the cellular cholesterol, are activated to export the excess. Nuclear factor erythroid 2-related factor-1 (NRF1) in the endoplasmic reticulum binds directly to cholesterol and senses when its level is high, thereby bringing about the derepression of genes involved in cholesterol removal, with mediation by the liver X receptor. Also, side-chain oxysterols, especially 25-hydroxycholesterol, can suppress the activation of SREBP by binding to an oxysterol-sensing protein in the endoplasmic reticulum.
Within cells, cholesterol derived initially from the lysosomal degradation of low-density lipoproteins is transferred first to the plasma membrane and thence to the endoplasmic reticulum, the latter step by a mechanism involving proteins known as GRAMD1s embedded in the endoplasmic reticulum membrane at sites in contact with the plasma membrane. These proteins have two functional domains: the START-like domain, which binds cholesterol, and the GRAM domain, which binds anionic lipids such as phosphatidylserine, thereby forming a link between the two membranes that enables the transfer of cholesterol.
In peripheral tissues, excess cholesterol is exported to high-density lipoproteins (HDL) in the circulation and returned to the liver, a process known as reverse cholesterol transport. The liver plays a crucial role in cholesterol synthesis, but it is equally essential for its elimination from the body through bile. Additionally, some lipoproteins containing cholesterol and cholesterol esters are delivered to lysosomes by endocytosis for degradation. Cholesterol is transported to the inner surface of the lysosomal membrane through the glycocalyx, via a transglycocalyx tunnel, with the aid of Niemann-Pick C1, C2, and other proteins, and then via contact sites between membranes to other organelles. Cholesterol in cellular membranes above the stoichiometric requirement can escape back into the cell, where it may serve as a feedback signal to downregulate cholesterol accumulation. Meanwhile, some is converted into the relatively inert storage form, i.e., cholesterol esters, while others are used for steroidogenesis.
The intestines play a crucial role in cholesterol homeostasis by absorbing dietary cholesterol and excreting it through feces, along with its metabolites. A specific transporter (Niemann-Pick C1-like 1 or NPC1L1)in the brush border membrane of enterocytes in the proximal jejunum of the small intestine is involved in the uptake of cholesterol from the intestinal contents. In contrast, the metabolism of sterols in the intestines is primarily controlled by an acetyl-CoA acetyltransferase (ACAT2), which facilitates intracellular cholesterol esterification, and the microsomal triglyceride transfer protein (MTTP), which is involved in the assembly of chylomicrons for export into the lymphatic system. Some cholesterol can be transferred in the opposite direction (transintestinal cholesterol excretion), but the quantitative importance of this process is unclear. There is evidence that dietary or synthesized cholesterol is necessary to maintain intestinal integrity, as cholesterol derived from circulating lipoproteins is not sufficient for the purpose.
In the intestines, especially the colon, the intestinal microflora can hydrogenate cholesterol from bile, diet, and desquamated cells to form coprostanol with an efficiency dependent on the composition of microbial species. Coprastanol is not significantly absorbed by intestinal tissue, and it may inhibit the uptake of residual cholesterol. There are two mechanisms for this conversion in bacteria: one involving direct reduction and another via cholestenone and coprostanone as intermediates. As the relevant genes have now been identified, the therapeutic potential is under investigation.
Brain: There are substantial differences in cholesterol synthesis and metabolism in the brain in comparison to the liver and peripheral tissues. Trace amounts of cholesterol can cross the blood-brain barrier via transport in low-density lipoproteins. Therefore, virtually all the cholesterol in the brain must be synthesized de novo, mainly in astrocytes (glial cells). During the perinatal and adolescent years, especially, cholesterol is synthesized in large amounts to form the myelin that surrounds the axons. Afterward, the rate begins to decline, eventually reaching about 10% of its earlier values.
Cholesterol is transported to neurons in the form of Apo E complexes in discoidal HDL-like particles, for which seven main receptors have been identified in brain cells that take up cholesterol from these lipoproteins. Apo E is synthesized in the brain, and 24-hydroxy-cholesterol concentrations regulate its transcription. Similarly, in the brain and central nervous system, cholesterol synthesis is regulated independently of that in peripheral tissues, mainly by forms of the liver X receptor (LXR). As cholesterol and oxysterols are involved in providing neuroprotective effects and lowering neuroinflammation, dysregulation of their concentrations has been noted in many neurodegenerative disorders. Most of the lipoproteins in cerebrospinal fluid differ from the nascent poorly-lipidated HDL secreted by astrocytes, suggesting that the latter are modified during maturation.
Cholesterol Catabolism
Cholesterol is not readily degraded in animal tissues, so it does not serve as a metabolic fuel to generate ATP. Only the liver possesses the enzymes to degrade significant amounts, and then via pathways that do not lead to energy production. Cholesterol and oxidized metabolites, known as oxysterols, are transferred back from peripheral tissues in lipoprotein complexes to the liver for catabolism by conversion to oxysterols and bile acids. The latter are exported into the intestines to aid in digestion, while leading to some loss that is essential for cholesterol homeostasis. Until recently, it was believed that approximately 90% of cholesterol elimination from the body in humans occurred via bile acids. However, experiments with animal models now suggest that a significant amount is secreted directly into the intestines by a process known as trans-intestinal cholesterol efflux. The mechanisms by which this occurs and its relevance to humans are under active investigation.
Gut bacteria reduce some of the cholesterol in the diet to highly insoluble 5β-cholestan-3β-ol (coprostanol), which is excreted and can be used as a biomarker for sewage in the environment. Certain bacterial species contain a 3β-hydroxysteroid:oxygen oxidoreductase (EC 1.1.3.6), commonly referred to as cholesterol oxidase, a flavoenzyme that catalyzes the oxidation of cholesterol to cholest-5-en-3-one, which is then rapidly isomerized to cholest-4-en-3-one, marking the first essential step in the catabolism of sterols. The enzyme is widespread in organisms that degrade organic wastes, but it is also present in pathogenic organisms where it influences the virulence of infections (see below). In biotechnology, it has been utilized for the production of various steroids, and it is employed in a clinical procedure for determining cholesterol levels in serum.
Cholesterol Esters
Cholesterol esters, i.e., with long-chain fatty acids linked to the hydroxyl group, are much less polar than free cholesterol and appear to be the preferred form for transport in plasma and as a biologically inert storage or detoxification form to buffer an excess. They do not contribute to membrane structures but are packed into intracellular lipid droplets. Cholesterol esters are major constituents of the adrenal glands, and they accumulate in the fatty lesions of atherosclerotic plaques. Similarly, esters of steroidal hormones are also present in the adrenal glands, where they are concentrated in cytosolic lipid droplets adjacent to the endoplasmic reticulum; 17β-estradiol, the principal estrogen in fertile women, is transported in lipoproteins in the form of a fatty acid ester.
Because of the mechanism of synthesis (see below), plasma cholesterol esters tend to contain relatively high proportions of the polyunsaturated components typical of phosphatidylcholine, as shown in Table \(\PageIndex{1}\) below. Arachidonic and “adrenic” (20:4(n-6)) acids can be especially abundant in cholesterol esters from the adrenal gland.
| Table \(\PageIndex{1}\). Fatty acid composition of cholesterol esters (wt% of the total) from various tissues. | |||||||
| Form | Fatty acids | ||||||
|---|---|---|---|---|---|---|---|
| 16:0 | 18:0 | 18:1 | 18:2 | 18:3 | 20:4 | 22:4 | |
| Human | |||||||
| plasma | 12 | 2 | 27 | 45 | 8 | ||
| liver | 23 | 10 | 28 | 22 | 6 | ||
| Sheep | |||||||
| plasma | 10 | 2 | 27 | 35 | 7 | - | - |
| liver | 17 | 9 | 29 | 7 | 4 | 3 | - |
| adrenals | 13 | 7 | 35 | 18 | 2 | 4 | 2 |
| Data from Christie, W.W. et al. Lipids, 10, 649-651 (1975); DOI. Nelson, G.J. Comp. Biochem. Physiol., 30, 715-725 (1969); Horgan, D.J. and Masters, C.J. Aust. J. Biol. Sci., 16, 905-915 (1963); Nestel, P.J. and Couzens, E.A. J. Clin. Invest., 45, 1234-1240 (1966); DOI. | |||||||
In plasma and the high-density lipoproteins (HDL) in particular, cholesterol esters are synthesized largely by the transfer of fatty acids to cholesterol from position sn-2 of phosphatidylcholine (‘lecithin’), catalyzed by the enzyme lecithin:cholesterol acyl transferase (LCAT); the other product is 1-acyl lysophosphatidylcholine. This is illustrated in Figure \(\PageIndex{29}\).
The reaction occurs in several steps. First, apoprotein A1 in HDL acts to concentrate lipid substrates near LCAT and present them in the optimal conformation; simultaneously, it opens a lid on the enzyme, activating it by exposing the site of transesterification. Then, cleavage of the sn-2 ester bond of phosphatidylcholine occurs via the phospholipase activity of LCAT, releasing a fatty acyl moiety. This is transacylated to the sulfur atom of a cysteine residue, forming a thioester, and ultimately it is donated to the 3β-hydroxyl group of cholesterol to form the cholesterol ester. Some LCAT activity has also been detected in apolipoprotein B100-containing particles (β-LCAT activity as opposed to α-LCAT with HDL).
It has been established that human LCAT is a relatively small glycoprotein with a polypeptide mass of approximately 49 kDa, which increases to about 60 kDa due to the addition of four N-glycosylation and two O-glycosylation moieties. Most of the enzyme is produced in the liver and circulates in the bloodstream bound reversibly to HDL, where the main protein component of HDL, apolipoprotein A1, activates it. As cholesterol esters accumulate in the lipoprotein core, cholesterol is removed from its surface, thus promoting the flow of cholesterol from cell membranes into HDL. This, in turn, leads to morphological changes in HDL, resulting in larger and more spherical particles. Subsequently, cholesterol esters are transferred to the other lipoprotein fractions, LDL and VLDL, a reaction catalyzed by cholesterol ester transfer protein. This process promotes the efflux of cholesterol from peripheral tissues, known as reverse cholesterol transport, especially from macrophages in the arterial wall, for subsequent delivery to the liver. LCAT is often cited as the primary driving force behind this process, and it is of great importance for cholesterol homeostasis, making it a suggested target for therapeutic intervention against cardiovascular disease.
The stereospecificity of LCAT changes with molecular species of phosphatidylcholine containing arachidonic or docosahexaenoic acid, when 2-acyl lysophosphatidylcholines are formed. This reaction may be significant for the supply of these essential fatty acids to the brain in that such lysophospholipids are believed to cross the blood-brain barrier more readily than the free acids.
In other animal tissues, a further enzyme acyl-CoA:cholesterol acyltransferase (ACAT) synthesizes cholesterol esters from CoA esters of fatty acids and cholesterol. ACAT exists in two forms, both of which are intracellular enzymes found in the endoplasmic reticulum and are characterized by multiple transmembrane domains and a catalytic histidine residue in a hydrophobic domain; they are members of the O-acyltransferase (MBOAT) superfamily. ACAT1 is present in many tissues, but is especially abundant in macrophages and the adrenal and sebaceous glands, which store cholesterol esters in the form of cytoplasmic lipid droplets. It is responsible for the synthesis of cholesterol esters in arterial foam cells within human atherosclerotic lesions. ACAT2 is found only in the liver and small intestine, and it is believed to be involved in the supply of cholesterol esters to the nascent lipoproteins. Analogous enzymes are found in yeast, where ergosterol is the primary sterol; however, a very different process occurs in plants.
Oxidized Cholesterol Esters: All lipid classes containing polyunsaturated fatty acids are susceptible to oxidation. Under normal circumstances, cholesterol esters are considered to be relatively inert. However, when they contain oxidized polyunsaturated fatty acids, their properties change, and they acquire biological activity. A reaction with 15-lipoxygenase may form such oxidized cholesterol esters, but they can also be produced through free radical-induced lipid peroxidation. They have been detected in lipoproteins, particularly LDL, in human blood and atherosclerotic lesions. Those oxidized cholesterol esters in plasma are trafficked into cells and metabolized by the same mechanisms as the corresponding unoxidized lipids.
Such "minimally oxidized LDL" does not bind to CD36 but rather to CD14, a receptor that recognizes bacterial lipopolysaccharides. The result is stimulation of toll-like receptor 4 (TLR4), although the response differs from that of lipopolysaccharides. In addition, oxidized metabolites of cholesteryl arachidonate of this kind stimulate macrophages to express inflammatory cytokines of relevance to atherosclerosis, among other effects. Oxidized cholesterol esters can be hydrolyzed to release their fatty acids, which can then be incorporated into phospholipids, resulting in a different repertoire of activities.
Hydrolysis of cholesterol esters: Cholesterol ester hydrolases in animals liberate cholesterol and free fatty acids when required for membrane and lipoprotein formation, and they also provide cholesterol for hormone synthesis in adrenal cells. Several cholesterol ester hydrolases have been identified, including carboxyl ester hydrolase, lysosomal acid cholesterol ester lipase, hormone-sensitive lipase, and hepatic cytosolic cholesterol ester hydrolase. These are located in various tissues and organelles, serving multiple functions. A neutral cholesterol ester hydrolase has received special attention, as it is involved in the removal of cholesterol esters from macrophages, thereby reducing the formation of foam cells and, consequently, the development of fatty streaks within the arterial wall —a key event in the progression of atherosclerosis.
Other Animal Sterols
Cholesterol will slowly oxidize in tissues or foods to form a range of different products with additional hydroperoxy, epoxy, hydroxy, or keto groups, and these can enter tissues through the diet. There is increasing interest in these compounds from the standpoint of human health and nutrition, as the accumulation of oxo-sterols in plasma is associated with the inhibition of cholesterol and bile acid biosynthesis, as well as other abnormalities in plasma lipid metabolism.
Some other sterols occur in small amounts in tissues, most of which are intermediates in the pathway from lanosterol to cholesterol. However, some of them have distinct functions in their own right. Lanosterol, the first sterol intermediate in cholesterol biosynthesis, was first identified in wool wax, both in its free and esterified forms, and remains the primary commercial source. It is found at low levels only in most other animal tissues (typically 0.1% of the cholesterol concentration). As oxygen is required, lanosterol cannot be produced by primitive organisms; hence, its absence from prokaryotes has led to some speculation on its evolutionary significance. When sterols became available to eukaryotes, numerous new possibilities emerged for their continued evolution. The production of cholesterol from lanosterol is then seen as ‘molecular streamlining’ by evolution, removing protruding methyl groups that hinder the interaction between sterols and phospholipids in membranes.
Desmosterol (5,24-cholestadien-3β-ol), the last intermediate in the biosynthesis of cholesterol by the Bloch pathway, may be involved in the process of myelination, as it is found in relative abundance in the brains of young animals but not in those of adults, other than astrocytes. It is also found in appreciable amounts in testes and spermatozoa, together with another cholesterol intermediate, testis meiosis-activating sterol. In addition, there is evidence that desmosterol activates certain genes involved in lipid biosynthesis in macrophages and may deactivate others associated with the inflammatory response. There is a rare genetic disorder in which there is an impairment in the conversion of desmosterol to cholesterol, desmosterolosis, with serious consequences in terms of mental capacity. These and related sterols appear to be essential for human reproduction.
In human serum, the levels of lathosterol (5α-cholest-7-en-3β-ol) were found to be inversely related to the size of the bile acid pool, and in general, the concentration of serum lathosterol is strongly correlated with the cholesterol balance under most dietary conditions. The isomeric saturated sterols, cholestanol and coprastanol, which differ in the stereochemistry of the hydrogen atom on carbon 5, are formed by microbial biohydrogenation of cholesterol in the intestines, and together with cholesterol are the main sterols in feces. Further examples of animal sterols include 7-dehydrocholesterol (cholesta-5,7-dien-3β-ol), found in the skin, which, upon irradiation with UV light, is converted to vitamin D3 (cholecalciferol). These sterols are shown in Figure \(\PageIndex{30}\).
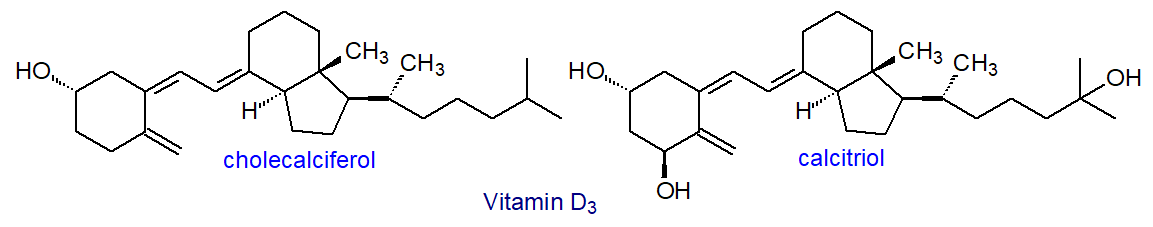
Figure \(\PageIndex{30}\): Other animal sterols
Marine invertebrates produce a large number of novel sterols, featuring both unusual nuclei and unconventional side chains, some derived from cholesterol and others from plant sterols or alternative biosynthetic intermediates. For example, at least 80 distinct sterols have been isolated from echinoderms and 100 from sponges.
Click the link for more information.
- Link
-
Elevated cholesterol and cholesterol ester levels are associated with the pathogenesis of cardiovascular disease, including atherosclerotic plaques, myocardial infarctions, and strokes, as is well known. The rate-limiting enzyme in cholesterol synthesis, HMG-CoA reductase, is the target of statins. However, drugs targeting other steps in the biosynthetic pathway, particularly squalene monooxygenase and lanosterol synthase, are also under investigation.
Cholelithiasis, or the presence of 'stones' in the gallbladder or bile ducts, which consist largely of cholesterol (~85%), is one of the most prevalent and costly digestive diseases in developed countries. The primary cause is the excessive excretion of cholesterol from the liver. Excess accumulation of cholesterol associated with the metabolism of bis(monoacylglycero)phosphate and causing disturbances in glycosphingolipid trafficking in cell membranes is involved in the pathogenesis of Niemann-Pick C disease, a lysosomal storage disease in which endocytosed cholesterol becomes sequestered in late endosomes/lysosomes because of gene mutations affecting two binding proteins (NPC1 and NPC2) thereby causing neuronal and visceral atrophy. In addition, deficiencies in cholesterol transport and metabolism are associated with many forms of neurodegeneration, including Alzheimer’s disease, Huntington’s disease, and related conditions associated with old age. These proteins are also key virulence factors for several viral and bacterial pathogens.
Several genetic disorders of cholesterol biosynthesis have been identified in recent years that can result in developmental malformations, including neurologic defects. As there is limited cholesterol transport across the placenta, the human fetus is highly dependent upon endogenous synthesis. While the molecular basis for the altered developmental pathways is not fully understood, impaired synthesis of the hedgehog family of signaling proteins, which require covalently linked cholesterol to function in membranes, is believed to be involved in many cases. In others, there are confirmed enzyme defects. For example, the recessive Smith-Lemli-Opitz syndrome in infants born with a decreased concentration of the enzyme 7-dihydrocholesterol reductase produces symptoms varying from mild autism to severe mental and often fatal physical problems. The effects are due to a lack of cholesterol and the accumulation of 7-dehydrocholesterol and its 27-hydroxy metabolite, as brain tissue cannot utilize dietary cholesterol or that produced peripherally. At least eight different inherited disorders of cholesterol biosynthesis result in congenital abnormalities in those affected. In animal models, deficiencies in SREBP-2 and genes encoding sterol biosynthetic enzymes display embryonic lethality. Deficiencies in the enzymes responsible for the hydrolysis of cholesterol esters, such as the lysosomal acid lipase, occur in Wolman disease and cholesterol ester storage disease.
Cholesterol and other sterols bind directly to several immune receptors, especially in macrophages and T cells. Dynamic changes in cholesterol biosynthesis directly impact innate and adaptive immune responses, such that functional coupling between sterol metabolism and immunity has implications for health and disease. For example, cholesterol binds directly to the αβ T cell antigen receptor (αβTCR) and has at least two opposing functions in its activation. By binding to the transmembrane domain of this receptor, it is maintained in an inactive, non-signaling conformation; however, when required, it can stimulate the formation of receptor nanoclusters to increase their avidity for the antigen. In cancer, there is a high demand for cholesterol to support the inherent nature of tumor cells to divide and proliferate, and perturbations of reverse cholesterol transport can have negative consequences. Drugs that lower cholesterol levels in cancer cells by inhibiting the mevalonate pathway are undergoing clinical trials.
When increased levels of sterols other than cholesterol are detected in plasma, they typically serve as markers for abnormalities in lipid metabolism associated with specific disease states. For example, premature atherosclerosis and xanthomatosis occur in two rare lipid storage diseases, cerebrotendinous xanthomatosis and sitosterolemia. In the former, cholestanol is present in all tissues, while in the latter, the dietary plant sterols campesterol and sitosterol accumulate in plasma and red blood cells. Inhibition of cholesterol biosynthesis may be associated with the appearance of some of the precursor sterols in the plasma.
In infections with Mycobacterium tuberculosis, the organism utilizes host cholesterol as its primary carbon and energy source, thereby promoting persistent infection with significant effects on pathogenicity. Similarly, Chlamydia trachomatis, a gram-negative obligate intracellular bacterium and a significant cause of sexually transmitted diseases, requires host cholesterol for growth. Many viruses utilize cholesterol as part of their life cycle, and a reduction in cellular cholesterol is sometimes employed as an anti-viral strategy. However, this approach may not always be beneficial. For example, an HIV protein has a binding site for cholesterol, which it utilizes to facilitate fusion with raft regions in the host cell's membranes.
Sterols: 2. Oxysterols and Other Cholesterol Derivatives
Oxysterols, as defined and discussed here, are oxygenated derivatives of cholesterol and its precursors, i.e., with additional hydroxyl, epoxyl, or keto groups, that are found in all animal tissues. Many of these have vital functions in animals. In contrast, others are important as short-lived intermediates or end products in the catabolism or excretion of cholesterol, as well as in the biosynthesis of steroid hormones, bile acids, and 1,25-dihydroxy-vitamin D3. They are usually present in biological membranes and lipoproteins at trace levels, although they can exert profound biological effects at these concentrations. However, they are always accompanied by a significant excess (as much as 106-fold) of cholesterol in itself.
A variety of different oxysterols are synthesized in cells through sequential reactions catalyzed by specific oxygenases. However, due to the presence of the double bond in the 5,6-position, oxysterols can also be formed rapidly by non-enzymatic oxidation, or autoxidation, of cholesterol and cholesterol esters within tissues, resulting in the formation of various oxygenated derivatives. Simplistically, non-enzymatic oxidation leads mainly to the generation of products in which the sterol ring system is oxidized. At the same time, enzymatic processes typically produce metabolites with an oxidized side chain, with 7-hydroxylation being an important exception. Oxidized cholesterol molecules can also be generated by the gut microflora and be taken up through the enterohepatic circulation. Once an oxygen function is introduced into cellular cholesterol, the product can act as a biologically active mediator by interacting with specific receptors before it is metabolized to bile acids or further degraded, a process assisted by the fact that oxysterols can diffuse much more rapidly through membranes than cholesterol itself. Cholesterol metabolites of this kind are significant in the brain, which is a major site for de novo cholesterol synthesis, and they are crucial elements of cholesterol homeostasis.
Enzymatic Oxidation of Cholesterol
Within animal cells, the oxidation of sterols is primarily an enzymatic process carried out by several enzymes from the cytochrome P450 family of oxygenases, named for their characteristic absorption at 450 nm. These comprise a disparate group of proteins that contain a single heme group and have a similar structural fold, though the amino acid sequences can differ appreciably. They are all mono-oxygenases. Oxysterol biosynthesis can be considered in terms of different pathways that depend on the position of the initial oxidation. These pathways tend to overlap and lead to a complex web of different oxysterols, eventually resulting in bile acids. As these enzymes, which include cytochrome P450, cholesterol hydroxylase, hydroxysteroid dehydrogenases, and squalene epoxidase, are specific to particular tissues and indeed animal species, there is considerable variation in oxysterol distributions between organs. A few examples only of the first steps in some of these pathways are illustrated in Figure \(\PageIndex{31}\).
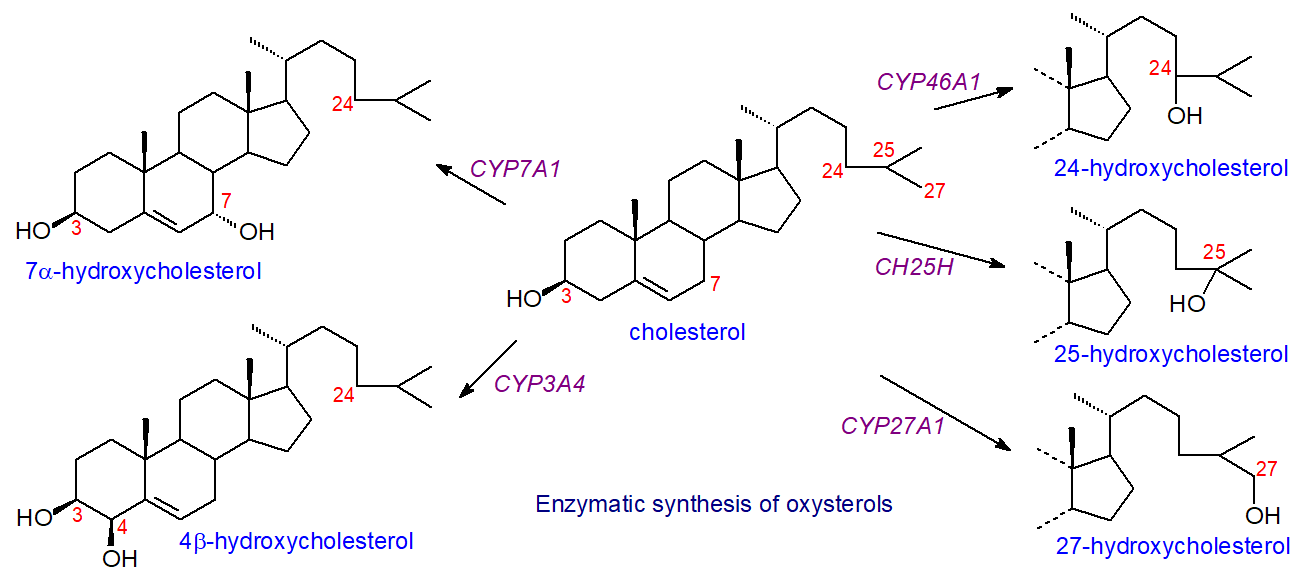
As an example, a primary product is 7α‑hydroxycholesterol, which is an important intermediate in the biosynthesis of bile acids by the 'neutral' pathway and of many other oxysterols, and it is produced in the liver by the action of cholesterol 7α-hydroxylase (CYP7A1). This enzyme plays a crucial role in maintaining cholesterol homeostasis. The reaction is under strict regulatory control, and the expression of CYP7A1 is controlled by the farnesoid X receptor (FXR) and is activated by cholic and chenodeoxycholic acids. Any circulating 7α‑hydroxycholesterol represents leakage from the liver. Further oxidation of 7α‑hydroxycholesterol can occur, and the action of CYP3A4 in humans generates 7α,25‑dihydroxycholesterol as an important metabolite, for example, while oxidation by CYP27A1 yields 7α,27‑dihydroxycholesterol; the latter is regarded as a key step in a further pathway to oxysterols and bile acids. On the other hand, the epimer 7β‑hydroxycholesterol is produced in the brain by the action of the toxic β-amyloid peptide and its precursor on cholesterol, but whether this is involved in the pathology of Alzheimer’s disease has yet to be determined.
The hydroxysteroid 11-β-dehydrogenase 1 (HSD11B1) is responsible for the conversion of 7β-hydroxy-cholesterol to the important metabolite 7-keto-cholesterol, while HS11B2 catalyzes the reverse reaction; 7-keto-cholesterol is also formed by autoxidation (see below). HSD11B1 is better known as the oxidoreductase that converts inactive cortisone to the active stress hormone cortisol in glucocorticoid target tissues.
An alternative ('acidic') pathway to bile acids starts with the synthesis of 27-hydroxycholesterol (or more systematically named (25R)26‑hydroxycholesterol), which is produced by the cytochrome P450 enzyme (CYP27A1) and introduces the hydroxyl group into the terminal methyl carbon (C27 or C26 - used interchangeably). While this enzyme is present in the liver, it is also found in many extrahepatic tissues, particularly the lung, which provides a steady flux of 27-oxygenated metabolites to the liver. As a multifunctional mitochondrial P450 enzyme in the liver, it generates both 27‑hydroxycholesterol and 3β‑hydroxy-5-cholestenoic acid, the bile acid precursor, which occurs in small but significant amounts in plasma. 27‑Hydroxycholesterol is the most abundant circulating oxysterol, and its concentration in plasma correlates with that of total cholesterol. It can be oxidized to 7α,27‑dihydroxycholesterol by the enzyme CYP7B1. 4β‑Hydroxycholesterol is also abundant in plasma and is relatively stable; it is produced in humans by the action of the cytochromes CYP3A4 and CYP3A5.
In humans, the specific cytochrome P450 that produces 24S-hydroxycholesterol (cholest-5-ene-3β,24-diol) is cholesterol 24S‑hydroxylase (CYP46A1) and is located almost entirely in the smooth endoplasmic reticulum of neurons in the brain, including those of the hippocampus and cortex, which are important for learning and memory. It is by far the most abundant oxysterol in the brain after parturition, but during development, many more oxysterols are produced. 24S‑hydroxycholesterol is responsible for 98-99% of the turnover of cholesterol in the central nervous system, which is the source of most of this oxylipin found in plasma. A small amount of it is converted in the brain directly into 7α,24S‑dihydroxycholesterol by the cytochrome CYP39A1 and thence via side-chain oxidation in peroxisomes to bile acids, such as cholestanoic acid. It is evident that constant passive fluxes of oxysterols cross the blood-brain barrier, but not cholesterol per se, due to their permissive chemical structures and concentration gradients. In plasma, it is transported via high-density lipoproteins, as discussed further below. In contrast to humans, CYP46A1 is present in the liver of rodents as well as the brain.
25-Hydroxycholesterol is a relatively minor but biologically important cholesterol metabolite that is produced rapidly by immune cells in response to inflammation resulting from bacterial or viral infections. The dioxygenase enzyme cholesterol 25-hydroxylase (CH25H in humans), which utilizes a diiron cofactor to catalyze hydroxylation, is the primary route to this metabolite in vivo. However, at least two cytochrome P450 enzymes, CYP27A1 and CYP3A4, can catalyze this conversion to a limited extent. Further oxidation by CYP7B1 is a second route to 7α,25‑dihydroxycholesterol, and thus to additional oxysterols.
24(S),25-Epoxycholesterol is not produced by the pathways described above but is synthesized in a shunt of the mevalonate pathway using the same enzymes that produce cholesterol, specifically squalene mono-oxygenase and lanosterol synthase, in which a second epoxy group is introduced on the other end of squalene from the initial epoxidation. A further mechanism in the brain is the action of CYP46A1 on desmosterol, another intermediate in cholesterol biosynthesis.
The oxysterols formed by both autoxidation and enzymatic routes can undergo further oxidation-reduction reactions, and they can be modified by many of the enzymes involved in the metabolism of cholesterol and steroidal hormones, such as esterification and sulfation of position 3, as illustrated for 7-keto-cholesterol as an example in Figure \(\PageIndex{32}\).
In most tissues, esterification of the 3β-hydroxyl group only occurs and requires the activity of sterol O-acyltransferases 1/2 (SOAT1/2 or ACAT1/2) with the participation of cytosolic phospholipase A2 (cPLA2α) to liberate the required fatty acids from phospholipids. In plasma, oxysterols can be esterified by lecithin–cholesterol acyltransferase (LCAT) for transport in lipoproteins; however, in this instance, a diester can be specifically produced from 27-hydroxycholesterol. Whether such esters serve as an inert storage form for oxysterols, which can be liberated on demand by esterases, remains to be determined.
It is noteworthy that the important human pathogen Mycobacterium tuberculosis utilizes a cytochrome P450 enzyme (CYP125) to catalyze the C26/C27 hydroxylation of cholesterol, an essential early step in its catabolism as part of the infective process.
Catabolism: Because of their increased polarity relative to cholesterol, oxysterols produced by both enzymatic and non-enzymatic means can exit cells relatively easily. A proportion is oxidized further and converted to bile acids, while some are converted to sulfate esters (especially at the 3-hydroxyl group) or glucuronides (see below) for elimination via the kidneys.
Non-Enzymatic Oxidation of Cholesterol
In biological systems in which both cholesterol and fatty acids are present, it would be expected that autoxidation of polyunsaturated fatty acids by free radical mechanisms would be favored thermodynamically, with the formation of isoprostanes from arachidonic acid in phospholipids. However, certain circumstances can favor cholesterol oxidation in vivo. For example, the concentration of cholesterol in low-density lipoprotein particles (LDL) is approximately three times higher than that of phospholipids, and the rate of cholesterol-hydroperoxide formation can be higher than that of phospholipid hydroperoxides. The rate and specificity of the reaction can also depend on whether it is initiated by free radical species, such as those arising from the superoxide/hydrogen peroxide/hydroxyl radical system (Type I autoxidation), or whether it occurs by non-radical but highly reactive oxygen species such as singlet oxygen, HOCl, or ozone (Type II autoxidation). As examples of the main types of products of non-enzymatic oxidation, the structures of a few of the more important of these oxysterols are illustrated in Figure \(\PageIndex{33}\).
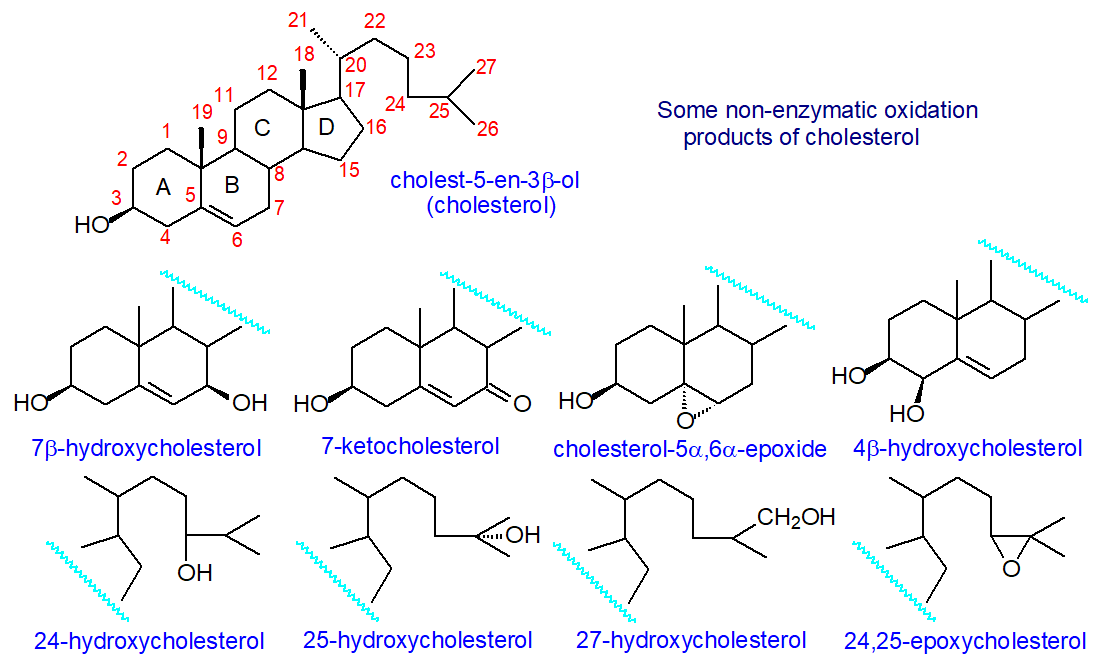
Oxysterols produced by this means can vary in type (hydroperoxy, hydroxy, keto, epoxy), number, and position of the oxygenated functions introduced, as well as the nature of their stereochemistry. Derivatives with the A and B rings and the iso-octyl side-chain oxidized are illustrated, but compounds with oxygen groups in position 15 (D ring) are also important biologically. Many are similar to those produced by enzymatic means, although the stereochemistry typically differs. Like the enzymic products, they are named according to their relationship to cholesterol, rather than by the strict systematic terminology.
Mechanisms of autoxidation have been studied intensively in relation to unsaturated fatty acids, and it appears that similar mechanisms also operate with sterols. The first event in lipid peroxidation by a radical mechanism is an initiation reaction, in which a carbon with a labile hydrogen undergoes hydrogen abstraction by reaction with a free radical, such as a non-lipid species, including transition metals, hydroxyl radicals, or peroxynitrite radicals. This is followed by oxygen capture. The resulting reactive species recruits further non-oxidized lipids, initiating a chain reaction known as the propagation phase. Finally, the reaction is terminated by the conversion of hydroperoxy intermediates into more stable hydroxy products through reaction with endogenous antioxidants, such as tocopherols.
As an example, the reaction mechanism leading to the production of 7-oxygenated cholesterol derivatives is illustrated in Figure \(\PageIndex{34}\).
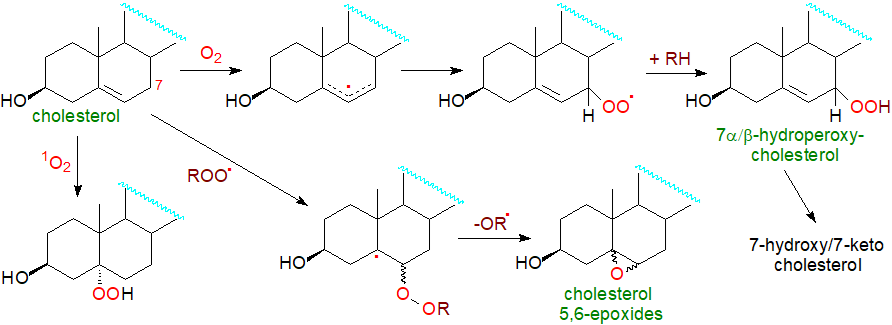
Figure \(\PageIndex{34}\): Cholesterol non-enzymatic oxidation mechanisms
In aqueous dispersions, oxidation is initiated by a radical attack from a reactive-oxygen species such as a hydroxyl radical with the abstraction of hydrogen from the C-7 position to form a delocalized three-carbon allylic radical, which reacts with oxygen to produce 7α‑hydroperoxycholesterol, which gradually isomerizes to the more thermodynamically stable 7β-hydroperoxycholesterol. Subsequent enzymic and non-enzymic reactions lead to the 7-hydroxy and 7-keto analogs, which tend to be the most abundant non-enzymatically generated oxysterols in tissues, often accompanied by epoxy-ene and ketodienoic secondary products. Reaction with singlet oxygen (1O2) produces 5α‑hydroperoxycholesterol mainly together with some 6α- or 6β-hydroperoxycholesterol. The reaction does not occur readily at the other allylic carbon, 4, presumably due to steric hindrance. When cholesterol is in the solid state, the reaction occurs primarily at the tertiary carbon-25, though some products oxygenated at C-20 may also be produced.
Cholesterol hydroperoxides can be converted to stable diols only by the phospholipid hydroperoxide glutathione peroxidase-type 4 (GPx4) and then relatively slowly, but not by the type 1 glutathione peroxidase (GPx1) when in a membrane-bound state. However, in mammalian cells, monomeric GPx4 (~20 kDa), although present in several cellular compartments, including mitochondria, is much less abundant than tetrameric GPx1. Phospholipid-hydroperoxides are reduced most rapidly, followed by cholesterol 6β-OOH > 7α/β-OOH >> 5α-OOH. The result is that cholesterol hydroperoxides are expected to have a relatively long half-life and can therefore be potentially quite hazardous in biological systems. Of these, 5α-OOH, with the lowest reduction rate, is the most cytotoxic of the hydroperoxides, unfortunately.
Epimeric 5,6-epoxy-cholesterols may be formed by a non-radical reaction involving the non-enzymatic interaction of a hydroperoxide with the double bond, a process believed to occur primarily in macrophages and low-density lipoproteins (LDL). In this instance, the initial peroxidation product is a polyunsaturated fatty acid. The hydroperoxide transfers an oxygen atom to cholesterol, producing the epoxide, and in doing so, is reduced to a hydroxyl. Other non-radical oxidation processes include reactions with singlet oxygen, which is especially important in the presence of light and photosensitizers, and can generate 5-hydroxy as well as 6- and 7-hydroxy products. Additionally, the reaction with ozone in the lungs can create a family of distinctive oxygenated cholesterol metabolites.
Similarly, a diverse range of oxidation products is generated by the peroxidation of the cholesterol and vitamin D precursor 7-dehydrocholesterol, which has the highest propagation rate constant known for any lipid in free-radical chain oxidation. These metabolites possess important biological properties.
Oxysterols occur in tissues both in the free state and esterified with long-chain fatty acids. For example, in human atherosclerotic lesions, 80–95% of all oxysterols are esterified. Appreciable amounts of oxysterols can be present in foods, especially those rich in cholesterol, such as meat, eggs, and dairy products, where they are most probably generated non-enzymically during cooking or processing when such factors as temperature, oxygen, light exposure, the associated lipid matrix, and the presence of antioxidants and water all play a part. Those present in foods can be absorbed from the intestines and transported into the circulation in chylomicrons. However, the extent to which dietary sources contribute to tissue levels of total oxysterols or individual isomers is not known and is likely to be highly variable, but relatively lower than that of cholesterol itself.
Oxysterols – Biological Activity
General Functions: In vivo tissues, the very low oxysterol:cholesterol ratio means that oxysterols have a minimal impact on the primary role of cholesterol in cell membrane structure and function. However, it has been claimed that oxysterols could cause packing defects and, consequently, atheroma formation in vascular endothelial cells. It is often argued that there are few reliable measurements of cellular or subcellular oxysterol concentrations, because of the technical difficulties in the analysis of the very low concentrations of oxysterols in the presence of a vast excess of native cholesterol; the average levels of 26-, 24- and 7α-hydroxy-cholesterol in human plasma that are often quoted are 0.36, 0.16 and 0.14 μM, respectively. Autoxidation products of cholesterol, particularly 7-keto- and 7-hydroxy-cholesterol, are cytotoxic and may serve as useful markers of oxidative stress or for monitoring the progression of various diseases. However, experts in the field caution that it can be challenging to extrapolate from experiments in vitro to the in vivo situation, due to the rapidity with which cholesterol can autoxidize in experimental systems and the difficulty of conducting experiments with physiological levels of oxysterols.
Nonetheless, aside from their role as precursors of bile acids and some steroidal hormones, it is evident that oxysterols have a variety of roles in maintaining cholesterol homeostasis and perhaps in signaling, where those formed enzymatically are most significant. They can exert potent biological effects at physiologically relevant concentrations by binding to various receptors, thereby eliciting transcriptional programs that regulate gene and, consequently, protein expression. Among many cell membrane receptors for oxysterols that have been identified, nuclear receptors are especially important and include the liver X receptors (LXRs), retinoic acid receptor-related orphan receptors (RORs), estrogen receptors (ERs), and glucocorticoid receptors (GRs). In addition, N-methyl-D-aspartate receptors (NMDARs) are expressed in nerve cells and work over a short time scale to regulate excitatory synaptic function. In contrast, G protein-coupled receptors operate at the cell membrane and are activated by molecules outside the cell to initiate signaling pathways within the cell. As various isoforms of these receptors exist in different tissues and can interact with several oxysterols, only a summary of this complex topic is possible here.
A family of oxysterol-binding proteins (OSBP) transports and regulates the metabolism of sterols and targets oxysterols to specific membranes, especially to contact sites between organelles, with the transport of phosphatidylinositol 4-phosphate in the reverse direction (see our web page on the latter). In this way, they can enable oxysterols to regulate membrane composition and function, as well as mediate intracellular lipid transport. As with cholesterol, oxysterols can be eliminated from cells by transporters such as the ATP-binding cassette proteins ABCA1 and ABCG1. They are transported in the bloodstream within lipoproteins, especially in association with HDL and LDL, and are mainly present in the esterified form.
Cholesterol homeostasis: While cholesterol plays a key role in its own feedback regulation, there is some evidence that oxysterols are regulators of cholesterol concentration in cell membranes, and that 25‑hydroxycholesterol and 24(S),25‑epoxycholesterol may be especially effective, although the other side-chain oxysterols 22-, 24- and 27‑hydroxycholesterol have been implicated. Several mechanisms appear to be involved, and it is suggested that 24(S),25‑epoxycholesterol especially acts as a ligand for the liver X receptor, which forms a heterodimer with the retinoic X receptor, to inhibit the transcription of key genes in cholesterol biosynthesis, as well as directly inhibiting or accelerating the degradation of such important enzymes in the process as HMG-CoA reductase and squalene synthase. Similarly, both 26-hydroxylanosterol and 25-hydroxycholesterol inhibit HMG-CoA reductase. 25‑Hydroxycholesterol inhibits the transfer of the 'sterol regulatory element binding protein' (SREBP-2) to the Golgi for processing to its active form as a transcription factor for the genes of the cholesterol biosynthesis pathway, and it stimulates the enzyme acyl-CoA:cholesterol acyl transferase in the endoplasmic reticulum to esterify cholesterol. Through these mechanisms, oxysterols fine-tune cholesterol homeostasis, ensuring smooth regulation rather than substantial fluctuations in tissue concentration.
Oxysterols and the immune system: Oxysterols are known to have vital and diverse roles in immunity by regulating both the adaptive and innate immune responses to infection. For example, infection with viruses leads to the production of type I interferon. In macrophages, this induces the synthesis of 25-hydroxycholesterol, which is generally regarded as anti-inflammatory and exerts broad antiviral activity by activating integrated stress response genes and reprogramming protein translation again via its interaction with LXR receptors. It is a potent inhibitor of SARS-CoV-2 replication, for example, possibly by a mechanism involving the blocking of cholesterol export from the late endosome/lysosome compartment and depletion of membrane cholesterol levels. However, the formation of 25-hydroxycholesterol may be detrimental in cases of influenza infections, as it can lead to the overproduction of inflammatory metabolites. Similarly, the biosynthesis of 25-hydroxycholesterol in macrophages is stimulated by the endotoxin Kdo2-lipid A, the active component of the lipopolysaccharide present on the outer membrane of Gram-negative bacteria, which acts as an agonist for Toll-like receptor 4 (TLR4). There is enhanced expression of the oxygenase CH25H in immune cells in response to bacterial and viral infection.
Many oxysterol species are active in a range of immune cell subsets, mediated through the control of LXR and SREBP signaling, as well as by acting as ligands for RORs and for the cell surface receptors G protein-coupled receptor 183 (GPR183) or CXCR2. Activation of LXR tends to dampen the immune response. In response to various stimuli, they can operate through ion channels to effect rapid changes in intracellular ion concentrations, especially of Ca2+, which brings about changes in membrane potential, cell volume, cell death (including apoptosis, autophagy, and necrosis), gene expression, secretion, endocytosis, or motility. For example, 27-hydroxycholesterol in human milk is reported to be active against the pathogenic human rotavirus and rhinovirus, which are important in pediatrics. Additionally, 7-dehydrocholesterol also exhibits antiviral properties. While they can exert their immune functions within the cell in which they are generated, oxysterols can also operate in a paracrine fashion towards other immune cells.
25-Hydroxycholesterol, in particular, can have either pro- or anti-inflammatory effects, depending on the conditions; however, the enzyme CH25H, responsible for its biosynthesis, is induced markedly in macrophages activated by inflammatory agents. It is reported to have a regulatory effect on the biosynthesis of sphingomyelin, which is required in conjunction with cholesterol for the formation of raft subdomains in membranes, where signaling molecules are concentrated. Together with other oxysterols, such as 24S,25-epoxycholesterol, it regulates the activities of the hedgehog proteins involved in embryonic development. Metabolites of 25-hydroxycholesterol, such as 7α,25-dihydroxycholesterol, and further oxidation products, are pro-inflammatory and act as chemoattractants for lymphocytes, playing a role in the regulation of immunity in secondary lymphoid organs through interactions with the GPR183 receptor.
Oxysterols in the brain: Oxysterols are especially important for cholesterol homeostasis in the brain, which contains 25% of the total body cholesterol, virtually all of it in unesterified form, in only about 2% of the body volume. Cholesterol is a major component of the plasma membrane, particularly in its role of regulating fluidity and permeability. This membrane is produced in large quantities in the brain. It forms the basis of compacted myelin, which is essential for the conduction of electrical stimuli and contains approximately 70% of the brain's cholesterol. While this pool is relatively stable, the remaining 30% is present in the membranes of neurons and glial cells in the gray matter and is more metabolically active. Even in the fetus and the newborn infant, all the cholesterol required for growth is produced by synthesis de novo in the brain, not by transfer from the circulation across the blood-brain barrier, which consists of tightly opposed endothelial cells lining the extensive vasculature of the tissue. The fact that this pool of cholesterol in the brain is independent of circulating levels must reflect a requirement for constancy in the content of this lipid in membranes and myelin. In adults, although the membrane undergoes continuous turnover, cholesterol is efficiently recycled and has a remarkably high half-life, up to 5 years. The rate of cholesterol synthesis is a little greater than the actual requirement, so net movement of cholesterol out of the central nervous system must occur. A crucial component of this system is apolipoprotein E (Apo E), a 39-kDa protein highly expressed in the brain, which facilitates the cellular transport of cholesterol and contributes to cholesterol homeostasis. Apo E complexes with cholesterol are required for transport from the site of synthesis in astrocytes to neurons.
Hydroxylation by CYP46A1 of cholesterol to 24(S)‑hydroxycholesterol (cerebrosterol) is responsible for 50–60% of all cholesterol metabolism in the adult brain. If cholesterol itself cannot cross the blood-brain barrier, this metabolite can do so with relative ease. When the hydroxyl group is introduced into the side chain, this oxysterol causes a local reordering of membrane phospholipids, making it more favorable energetically to expel it at a rate that is orders of magnitude greater than that of cholesterol alone, although still only 3-7 mg per day. There is a continuous flow of metabolites from the brain into the circulation, much of which is in the form of inactive sterol esters, which are transported by lipoprotein particles to the liver for further catabolism. Specifically, it undergoes hydroxylation at position 7 and is then converted into bile acids. This is illustrated in Figure \(\PageIndex{35}\).
Both 24(S)-hydroxycholesterol and 24(S),25-epoxycholesterol are believed to play a role in regulating cholesterol homeostasis in the brain. They interact with the specific nuclear receptors involved in the expression and synthesis of proteins involved in sterol transport. For example, 24-hydroxycholesterol regulates the transcription of apolipoprotein E (ApoE). In particular, it is an agonist of the nuclear liver X receptors (LXRs), influencing the expression of those LXR target genes involved in cholesterol homeostasis and inflammatory responses. It is also a high-affinity ligand for the retinoic acid receptor-related orphan receptors α and γ (RORα and RORγ). In this way, it can act locally to affect the functioning of neurons, astrocytes, oligodendrocytes, and vascular cells.
24(S)-Hydroxycholesterol downregulates the trafficking of the amyloid precursor protein and may be a factor in preventing neurodegenerative diseases. Especially high levels of 24(S)‑hydroxycholesterol are observed in the plasma of human infants and patients with brain trauma. In contrast, reduced levels are found in the plasma of patients with neurodegenerative diseases, including Parkinson’s disease, multiple sclerosis, and Alzheimer's disease. In contrast, there are elevated levels in the brain and especially cerebrospinal fluid in patients with these conditions, where it may be a marker of neurodegeneration. Increased expression of cholesterol 24-hydroxylase (CYP46A1) is believed to improve cognition. At the same time, a reduction leads to poor cognitive performance, as occurs at advanced stages of the disease, likely reflecting a selective loss of neuronal cells. This may be a factor in age-related macular degeneration. An excess of 24(S)‑hydroxycholesterol and especially of its ester form can lead to neuronal cell death, and elevated levels in plasma are reported to be a potential marker for Autism Spectrum Disorders in children. On the other hand, it may be protective against glioblastoma, the most common primary malignant brain tumour in adults, via activation of LXRs.
27‑Hydroxy-cholesterol diffuses across the blood-brain barrier from the bloodstream into the brain (in the reverse direction to 24‑hydroxycholesterol), where it does not accumulate but is further oxidized and then exported as steroidal acids. This flux may regulate certain key enzymes within the brain, and there are suggestions that the balance between the levels of 24- and 27-hydroxy-cholesterol, especially an excess of the latter, may be relevant to the generation of β-amyloid peptides in Alzheimer's disease by reducing insulin-mediated glucose uptake by neurons. While 7β-hydroxycholesterol is pro-apoptotic, any links with Alzheimer's disease are unproven. However, there is a school of thought that other oxidized cholesterol metabolites may be major factors behind the development of this disease. For example, seco-sterols such as 3β‑hydroxy-5-oxo-5,6-secocholestan-6-al and its stable aldolization product, the main ozonolysis metabolites derived from cholesterol, have been detected in brain samples of patients who have died from Alzheimer's disease and Lewy body dementia; they are also found in atherosclerotic lesions. Oxidation products of the cholesterol precursor 7‑dehydrocholesterol and especially those of 3β,5‑dihydroxycholest-7-ene-6-one are involved in the pathophysiology of the human disease Smith-Lemli-Opitz syndrome.
Cell differentiation: Oxysterols can influence the differentiation of various cell types, a phenomenon first studied in the skin, where 22(R)- and 25(R)-hydroxycholesterol were shown to induce differentiation in human keratinocytes. Subsequently, by stimulating nuclear binding receptors, oxysterols were found to have similar effects on mesenchymal stem cells. There have been numerous reports of the involvement of oxysterols in various disease processes, including atherosclerosis and the formation of human atherosclerotic plaques, as well as cytotoxicity, necrosis, inflammation, immunosuppression, phospholipidosis, and gallstone formation. They have been implicated in the development of cancers, especially those of the breast, prostate, colon, and bile duct. For example, 27-hydroxycholesterol is a component in the elimination of cholesterol from macrophages and arterial endothelial cells. Still, it is also an endogenous ligand for the human nuclear estrogen receptor (ERα) and the liver X receptor. It modulates their activities, with effects on various human disease states, including cardiovascular dysfunction and the progression of breast and prostate cancer, as well as involvement in the regulation of bone mineralization (osteoporosis). It has been linked to cancer metastasis through effects on immune cells, and there is hope that pharmacological inhibition of CYP27A1 and thence the formation of 27‑hydroxycholesterol may be a useful strategy in the treatment of breast cancer; CYP7A1 gene polymorphism has been associated with colorectal cancer. In contrast, oxysterols can interfere with the proliferation of several types of cancer cells (glioblastoma, leukemia, colon, breast, and prostate cancer).
Cholesterol 5,6-epoxide (with either 5α or 5β stereochemistry) is formed non-enzymatically in tissues, but it is also believed to be produced by an unidentified cytochrome P450 enzyme in the adrenal glands. While it was for some time believed to be a causative agent in cancer, it is now recognized that downstream metabolites are responsible. Thus, cholesterol epoxide hydrolase converts cholesterol 5,6-epoxide into cholestane-3β,5α,6β-triol, which is transformed by 11β‑hydroxysteroid-dehydrogenase-type-2 into the oncometabolite 3β,5α-dihydroxycholestan-6-one (oncosterone). By binding to the glucocorticoid receptor, this oncosterone stimulates the growth of breast cancer cells, and it also acts as a ligand to the LXR receptors, which may mediate its pro-invasive effects. In contrast, in normal breast tissue, cholesterol 5,6‑epoxide is metabolized to the tumor suppressor metabolite, a steroidal alkaloid designated dendrogenin A, which is a conjugation product with histamine and controls a nuclear receptor to trigger lethal autophagy in cancers; its synthesis is greatly reduced in cancer cells. Tamoxifen, a widely used drug against breast cancer, binds to the cholesterol 5,6-epoxide hydrolase, which is also a microsomal anti-estrogen binding site (AEBS), thereby inhibiting its activity.
7-Ketocholesterol is a major oxysterol produced during the oxidation of low-density lipoproteins and is one of the most abundant in plasma and atherosclerotic lesions; it accumulates in erythrocytes of heart failure patients. It exhibits a high pro-apoptotic potential and preferentially associates with membrane lipid raft domains. As it is not readily exported from macrophages, it impairs cholesterol efflux and promotes the development of the foam cell phenotype. In cardiomyocytes, this accumulation can lead to cell hypertrophy and death, and it has been suggested that oxysterols constitute a significant factor precipitating morbidity in atherosclerosis-induced cardiac diseases and inflammation-induced heart complications. Photooxidation in the retina, mediated by the action of free radicals or singlet oxygen, generates unstable cholesterol hydroperoxides, which may contribute to the development of age-related macular degeneration. For example, these compounds can be quickly converted to highly toxic 7α- and 7β-hydroxycholesterols and 7-ketocholesterol, depending on the status of tissue oxidases and reductases. Three separate enzymatic pathways have developed in the eye to neutralize their activities. Novel branches of the acidic pathway of bile acid biosynthesis metabolize these sterols.
Those oxysterols formed non-enzymatically can be particularly troublesome in disease. For example, they are enriched in pathologic cells and tissues, such as macrophage foam cells, atherosclerotic lesions, and cataracts. They may regulate some of the metabolic effects of cholesterol, but as cautioned above, effects observed in vitro may not necessarily be of physiological importance in vivo. Various oxysterols have been implicated in the differentiation of mesenchymal stem cells, as well as the signaling pathways involved in this process. High levels of 7‑hydroxycholesterol and cholestane-3β,5α,6β-triol are characteristic of the lysosomal storage diseases Niemann-Pick types B and C, as well as lysosomal acid lipase deficiency.
Cholesterol hydroperoxides: With the aid of START domain proteins, cholesterol hydroperoxides can translocate from a membrane of origin to another membrane, such as mitochondria. Such a transfer of free radical-generated 7-hydroperoxycholesterol, for example, has adverse consequences, as it impairs cholesterol utilization in steroidogenic cells and the anti-atherogenic reverse-cholesterol transport in vascular macrophages. The antioxidant activity of GPx4 may be crucial for maintaining mitochondrial integrity and functionality in these cells.
Vitamin D
Vitamin D encompasses two primary sterol metabolites that are essential for the regulation of calcium and phosphorus levels and hence for bone formation in animals. However, these have many other functions, including induction of cell differentiation, inhibition of cell growth, immunomodulation, and control of other hormonal systems. Vitamin D deficiency, often accompanied by calcium deficiency, is responsible for the disease rickets in children, characterized by weak and deformed bones. It is also associated with various types of cancer and autoimmune diseases. Ultraviolet light mediates the cleavage of 7-dehydrocholesterol, an important intermediate in cholesterol biosynthesis, by opening the second (B) ring in the skin to produce pre-vitamin D. This pre-vitamin D rearranges spontaneously to form the secosteroid vitamin D3, also known as cholecalciferol. Its structure is shown in Figure \(\PageIndex{36}\).

The newly generated vitamin D3 is transported to the liver, where it undergoes 25-hydroxylation and is subsequently transported to the kidney for 1α-hydroxylation to produce the active form, 1α,25-dihydroxyvitamin D3 (calcitriol). This is an actual hormone and serves as a high-affinity ligand for the vitamin D receptor in distant tissues. For transportation in plasma, it is bound to a specific glycoprotein, vitamin D-binding protein (BDP). Vitamin D2 or ergocalciferol is derived from ergosterol, which is obtained from plant and fungal sources in the diet.
Vitamin D3 functions by activating a cellular receptor, known as the vitamin D receptor (VDR), a transcription factor that binds to sites in the DNA called vitamin D response elements. There are thousands of such binding sites, which, together with co-modulators, regulate innumerable genes in a cell-specific manner. In this way, it enhances bone mineralization by promoting the absorption of dietary calcium and phosphate, as well as having direct effects on bone cells. In addition, it functions as a general development hormone in many different tissues, while together with Vitamin D2 it has profound effects on immune responses in the defense against microbes.
Steroidal Hormones and Their Esters
Steroidal hormones cannot be discussed in depth here as their structures, biosynthesis, and functions comprise a rather substantial and specialized topic. In brief, animal tissues produce small amounts of vital steroidal hormones from cholesterol, the primary precursor, with 22R-hydroxycholesterol, produced by hydroxylation via the cholesterol side-chain cleavage enzyme (P450scc), as the first metabolite in the pathway. This step involves the 'STAR' protein, which enables the transport of cholesterol into mitochondria, where conversion to pregnenolone is rate-limiting and involves first hydroxylation and then cleavage of the side-chain. After export from the mitochondria, it can be converted directly to progesterone or, in several steps, to testosterone. 17β-Estradiol, for example, is the most potent and important of the endogenous estrogens; it is made mainly in the follicles of the ovaries and regulates menstrual cycles and reproduction, but it is also present in testicles, adrenal glands, fat, liver, breasts, and brain. Testosterone is the primary male sex hormone and an anabolic steroid, and it is produced mainly in the testes; it has a key function in the development of male reproductive tissues such as the testes and prostate, in addition to promoting secondary sexual characteristics. Pregnane neurosteroids are synthesized in the central nervous system. Cholesterol homeostasis is therefore vital to fertility and numerous bodily functions. The structures of key steroidal hormones are shown in Figure \(\PageIndex{37}\).
Steroidal esters accumulate in tissues, such as the adrenal glands, which synthesize corticosteroids like cortisol and aldosterone, and are responsible for releasing hormones in response to stress and other factors. It is also apparent that fatty acyl esters of estradiols, such as dehydroepiandrosterone, accumulate in adipose tissue in post-menopausal women. Small amounts of estrogens acylated with fatty acids at the C-17 hydroxyl are present in the plasma lipoproteins. In each instance, they appear to be biologically inert storage or transport forms of the steroid. Eventually, esterified steroids in low-density lipoproteins (LDL) particles are taken up by cells via lipoprotein receptors and then are hydrolyzed to release the active steroid.
Sterols 3. Sterols and Their Conjugates from Plants and Lower Organisms
Plant Sterols - Structures and Occurrence
Plants, algae, and fungi contain a somewhat different range of sterols from those in animals. Like cholesterol, to which they are structurally and biosynthetically related, plant sterols form a group of triterpenes with a tetracyclic cyclopenta[a]phenanthrene structure and a side chain at carbon 17, sometimes referred to as the C30H50O triterpenome. The four rings (A, B, C, D) have trans ring junctions, and the side chain and two methyl groups (C-18 and C-19) are oriented at an angle to the rings above the plane, exhibiting β stereochemistry (similar to a hydroxyl group commonly located on C-3). The basic sterol from which other sterol structures are defined is 5α-cholestan-3β-ol with the numbering scheme recommended by IUPAC as shown in Figure \(\PageIndex{38}\).
The phytosterols (as opposed to zoosterols) include campesterol, β-sitosterol, stigmasterol, and Δ5‑avenasterol, some of which are illustrated in Figure \(\PageIndex{39}\).

These more common plant sterols have a double bond in position 5, and a definitive feature – a one- or two-carbon substituent with variable stereochemistry in the side chain at C-24, which is preserved during subsequent metabolism. For example, campesterol is a 24-methylsterol, while β-sitosterol and stigmasterol are 24‑ethylsterols. Occasionally, there is a double bond in this chain that can be of the cis or trans configuration, as in stigmasterol (at C22) or fucosterol (C24), the principal sterol in green algae.
Phytosterols can be further classified on a structural or biosynthetic basis as 4‑desmethyl sterols (i.e., with no substituent on carbon‑4), 4α‑monomethyl sterols, and 4,4‑dimethyl sterols. The most abundant group is the 4‑desmethyl sterols, which may be subdivided into Δ5-sterols (illustrated above), Δ7‑sterols (e.g., α-spinasterol), and Δ5,7-sterols depending on the position of the double bonds in the B ring. As the name suggests, brassicasterols (24‑methyl-cholesta-5,22-dien-3β-ol and related sterols) are best known from the brassica family of plants, but they are also common constituents of marine algae (phytoplankton). Phytostanols (fully saturated) are normally present at trace levels only in plants, but they are relatively abundant in cereal grains.
Many different sterols may be present in photosynthetic organisms, and their amounts and relative proportions depend on the species. Over 250 different phytosterols have been recorded, with 60 in corn (maize) alone, for example. As a rough generalization, a typical plant sterol mixture would be 70% sitosterol, 20% stigmasterol, and 5% campesterol (or >70% 24-ethyl-sterols and <30% 24-methyl-sterols). However, this composition may vary with the stage of development and in response to stress. Table 1 contains data on the main components from some representative commercial seed oils.
| Table \(\PageIndex{2}\). Sterol composition in some seed oils of commercial importance (mg/Kg). | ||||||||
| corn | cottonseed | olive | palm | rapeseed | safflower | soybean | sunflower | |
|---|---|---|---|---|---|---|---|---|
| cholesterol | - | - | - | 26 | - | - | - | - |
| campesterol | 2691 | 170 | 28 | 358 | 1530 | 452 | 720 | 313 |
| stigmasterol | 702 | 42 | 14 | 204 | - | 313 | 720 | 313 |
| β‑sitosterol | 7722 | 3961 | 1310 | 1894 | 3549 | 1809 | 1908 | 2352 |
| Δ5‑avenasterol | 468 | 85 | 29 | 51 | 122 | 35 | 108 | 156 |
| Δ7‑stigmasterol | 117 | - | 58 | 25 | 306 | 696 | 108 | 588 |
| Δ7‑avenasterol | - | - | - | - | - | 104 | 36 | 156 |
| brassicasterol | - | - | - | - | 612 | - | - | - |
| other | - | - | - | - | - | 69 | - | 39 |
| Data adapted from Gunstone, F.D. et al. The Lipid Handbook (Second Edition) (Chapman & Hall, London) (1994). | ||||||||
Cholesterol is usually a minor component only of plant sterols (<1%). Still, it is unwise to generalize too much, as it can be the main sterol component of red algae and some families of higher plants, such as in the Solanaceae, Liliaceae, and Scrophulariaceae. It can also be a significant constituent sterol of chloroplasts, shoots, pollen, and leaf surface lipids in other plant families; wheat roots contain 10% and Arabidopsis cells 19% of the sterols as cholesterol. Yeasts and fungi typically contain ergosterol as their primary sterol (see below). Ecdysteroids (phytoecdysteroids) are polyhydroxylated plant sterols that can occur in appreciable amounts in some species. Sterols are also found in some bacterial groups, but not in archaea. Hopanoids in bacteria are considered functional counterparts of triterpenes.
Sterols can occur in plants in the 'free' state, i.e., in which the sterol hydroxyl group is not linked to any other moiety. Still, they are usually present as conjugates with the hydroxyl group covalently bound via an ester bond to a fatty acid, for example, as sterol esters, or via a glycosidic linkage to glucose (and occasionally other sugars), as steryl glycosides.
Plant Sterols - Biosynthesis
The biosynthetic route to plant sterols resembles that to cholesterol in many aspects in that it follows an isoprenoid biosynthetic pathway with isopentenyl pyrophosphate, derived primarily from mevalonate, as the key building block in the cytoplasm (but not plastids) at least. The main pathway for the biosynthesis of isopentenyl pyrophosphate and dimethylallyl pyrophosphate, the isoprene units, has been described previously and therefore need not be repeated here. It is known as the 'mevalonic acid (MVA) pathway' and functions in the cytosol, endoplasmic reticulum, and mitochondria.
However, an alternative pathway that does not use mevalonic acid as a precursor was first established for bacterial hopanoids, but has since been found in plant chloroplasts, algae, cyanobacteria, eubacteria, and some parasites (although not in animals). This route is variously termed the ‘non-mevalonate’, ‘1‑deoxy-D-xylulose-5-phosphate’ (DOXP), or better, the 2C-methyl-D-erythritol 4-phosphate (MEP) pathway, as the last compound is presumed to be the first committed intermediate in sterol biosynthesis by this route. In the first step, pyruvate and glyceraldehyde phosphate are combined to form deoxyxylose phosphate, which is in turn converted to 2C-methyl-D-erythritol 4-phosphate. The pathway then proceeds via various erythritol intermediates until isopentenyl pyrophosphate and dimethylallyl pyrophosphate are formed. This pathway is illustrated in Figure \(\PageIndex{40}\).
There is evidence that some of the isoprene units are exchanged between the cytoplasm and plastids. In much of the plant kingdom, both the MVA and MEP pathways operate in parallel, but green algae use the MEP pathway only. Thereafter, sterol biosynthesis continues via squalene and (3S)-2,3-oxidosqualene.
In photosynthetic organisms (as opposed to yeast and fungi), the subsequent steps in the biosynthesis of plant sterols differ from those for cholesterol in that the important intermediate in the route from squalene via 2,3-oxidosqualene is cycloartenol, rather than lanosterol, and this is produced by the action of a 2,3(S)‑oxidosqualene-cycloartenol cyclase (cycloartenol synthase). Then, the enzyme sterol methyltransferase 1 is of special importance because it converts cycloartenol to 24-methylene cycloartenol, the first step in introducing the methyl group onto C-24. The enzyme cyclopropyl sterol isomerase is required to open the cyclopropane ring. Animals lack the sterol C24-methyltransferase gene. While this pathway is essentially linear up to the synthesis of 24-methylene lophenol, a bifurcation then occurs, resulting in two alternative pathways. One of these pathways leads to the synthesis of sitosterol and stigmasterol, while the other leads to the synthesis of campesterol. This pathway is shown in Figure \(\PageIndex{41}\).
There are more than thirty enzyme-catalyzed steps in the overall process of plant sterol biosynthesis, each of which is associated with membranes. We will describe a few here. The 4,4-dimethyl- and 4α-methylsterols are part of the biosynthetic pathway, but are only minor, if ubiquitous, sterol components of plants. New biosynthetic pathways are being discovered through genome analysis, revealing the complexity of sterol biosynthesis in various plant species.
Dinoflagellates produce a characteristic 4-methylsterol, termed dinosterol, as well as others, such as gorgosterol, via lanosterol as a precursor. Protozoans synthesize many different sterols related to those in plants. For example, some species of Acanthamoeba and Naegleria produce both lanosterol and cycloartenol. Still, only the latter is used for the synthesis of other sterols, especially ergosterol, but in other protozoan species, sterol biosynthesis occurs via lanosterol. The best-studied bacterial pathway is that of the methylotroph Methylococcus capsulatus, which produces several unique Δ18(14)-sterols and is known to possess a squalene epoxidase and a lanosterol 14-demethylase.
Cholesterol in plants is produced from cycloartenol as the key intermediate, with Sterol Side Chain Reductase 2 (SSR2) as the key enzyme. It is now established that the cholesterol biosynthetic pathway in tomato plants comprises 12 enzymes acting in 10 steps. Of these, half evolved through gene duplication and divergence from phytosterol biosynthetic enzymes, whereas others act reciprocally in both cholesterol and phytosterol metabolism. Algae can also produce cholesterol through a multi-step process from cycloartenol, as well as many other sterols, via 24-methylene lophenol as the key intermediate. It is hoped that genetic manipulation of these enzymes will lead to plants that synthesize high-value steroidal products.
Oxidation: Phytosterols can undergo non-enzymatic oxidation, resulting in the formation of oxysterols similar to those found in animals. This process results in ring products, including hydroxy, keto-, epoxy-, and triol-derivatives. Further enzymatic reactions can oxidize the side chain. However, photosensitized oxidation is more common in plants and is much faster (>1500 times); it starts with the ene-addition of singlet oxygen (1O2) on either side of the double bond in the B ring to generate 5α-/6α-/6β-hydroperoxysterols, of which 5α-OOH is the most abundant and rearranges to form the more stable 7α‑OOH isomer. This is the main reaction in foods stored under LED lighting in food retailers.
Plant Sterols - Function
Like cholesterol, plant sterols are amphiphilic and are vital constituents of all membranes, and especially of the plasma membrane, the mitochondrial outer membrane, and the endoplasmic reticulum. The three-dimensional structure of the plant sterols is such that there are planar surfaces at both the top and the bottom of the molecules, which permit multiple hydrophobic interactions between the rigid sterol and the other components of membranes. Indeed, they must determine the physical properties of membranes to an appreciable extent. It is believed that campesterol, β-sitosterol, and 24-methylcholesterol, in this order, can regulate membrane fluidity and permeability in plant membranes by restricting the mobility of fatty acyl chains, similarly to cholesterol in mammalian cells. However, stigmasterol has a much less significant effect on lipid ordering and no effect on membrane permeability. In the plasma membrane, plant sterols associate with glycosphingolipids, such as glucosylceramide and glycosylinositolphosphoceramides, in raft-like subdomains, analogous to those in animal cells. These associations support the membrane localization and activities of many proteins with important functions in plant cells. The sterol glycosides are especially important in this context (see below).
Sterols (and their conjugates) are involved in plant membrane adaptations to changes in temperature and other biotic and abiotic stresses. For example, β‑sitosterol is a precursor of stigmasterol via the action of a C22-sterol desaturase, and the ratio of these two sterols is important to the resistance of A. thaliana plants to low and high temperatures. In addition, plant sterols can modulate the activity of membrane-bound enzymes. Thus, stigmasterol and cholesterol regulate the activity of the Na+/K+-ATPase in plant cells, probably in a manner analogous to that of cholesterol in animal cells. Stigmasterol may be explicitly required for cell differentiation and proliferation. In addition to being the precursor of plant steroidal hormones, campesterol serves as a signaling molecule that regulates growth, development, and stress adaptation.
Perhaps surprisingly, cholesterol is a precursor for the biosynthesis of some steroidal saponins and alkaloids in plants, such as the well-known steroidal glycoalkaloid α-solanine in potatoes, as well as other steroids, including phytoecdysteroids, which are derived from lathosterol in some species. While the physiological roles of ecdysteroids in plants have yet to be confirmed, they are believed to enhance stress resistance by promoting health and vitality. Withanolides are complex oxysterols believed to serve as defense compounds against insect herbivores.
Steroidal Plant Hormones

They play a crucial role in plant growth processes, including cell elongation, division, differentiation, immunity, and the development of reproductive organs. They are involved in regulating numerous aspects of metabolism. Via signal transduction pathways, they interact with transcription factors through phosphorylation cascades to regulate the expression of target genes. Brassinosteroids are also signaling molecules in responses to abiotic stresses, including drought, salinity, high temperatures, low temperatures, and heavy metal stresses. They may have biomedical applications as anticancer drugs for endocrine-responsive cancers, inducing apoptosis and inhibiting growth. Some plant species produce small amounts of steroid hormones that are often considered to be of animal origin only, including progesterone and testosterone, and these may have physiological roles in plants.
Sterol Esters in Higher Plants
Sterol esters are present in all plant tissues, but they are most abundant in tapetal cells of anthers, pollen grains, seeds, and senescent leaves. In general, they are minor components relative to the free sterols, other than in waxes. Typically, the sterol components of sterol esters are similar to those of free sterols, although there may be a relatively lower concentration of stigmasterol. The fatty acid components tend to resemble those of the other plant tissue lipids, but there can be significant differences on occasion. Sterol esters are presumed to serve as inert storage forms of sterols, as they are often enriched in the intermediates of sterol biosynthesis and can accumulate in lipid droplets within the cells. However, they have been found in some membranes, especially in microsomes and mitochondrial preparations, although their function in these locations is uncertain. They may also play a role in transport within cells and between tissues, as they can exist in the form of soluble lipoprotein complexes.
Biosynthesis of sterol esters in A. thaliana is known to occur in the endoplasmic reticulum by the action of a phospholipid:sterol acyltransferase, which catalyzes the transfer of a fatty acyl group to the sterol from position sn-2 of phospholipids - mainly phosphatidylethanolamine; the enzyme is very different from those in animals and yeasts. However, an acyl CoA:sterol acyltransferase closer in structure to the animal enzyme has also been characterized; it prefers saturated fatty acyl-CoAs as acyl donors and cycloartenol as the acceptor molecule. The enzymes responsible for the hydrolysis of sterol esters in A. thaliana are not yet known. Certain distinctive phytosterol esters occur in the aleurone cells of cereal grains, including trans-hydroxycinnamate, ferulate (4-hydroxy-3-methoxycinnamate), and p-coumarate esters. Similarly, rice bran oil is a rich source of esters of ferulic acid and a mixture of sterols and triterpenols, termed 'γ-orizanol'’, and an example of one of these compounds is illustrated in Figure \(\PageIndex{43}\).
This is sold as a health food supplement due to its claimed beneficial effects, including cholesterol-lowering and antioxidant activities, as well as enhancements to muscle growth and sports performance. However, none of these effects have been confirmed by rigorous clinical testing.
Sterol Glycosides
Leaf and other tissues in plants contain a range of sterol glycosides and sterol acyl-glycosides in which the hydroxyl group at C3 on the sterol is linked to the sugar by a glycosidic bond. Other than in the genus Solanum, where they can represent up to 85% of the sterol fraction in tomato fruit, as an example, they tend to be minor components relative to other lipids. Typical examples (glucosides of β-sitosterol) are illustrated in Figure \(\PageIndex{44}\).

Most of the common plant sterols occur in this form, but Δ5 sterols are preferred (Δ7 in some genera). Glucose is the most common carbohydrate moiety, but galactose, mannose, xylose, and arabinose can also be present, depending on plant species; occasionally, complex carbohydrates with up to five hexose units linked linearly are present. Algae also contain sterol glycosides with a wide range of sterol and carbohydrate components. Plant, animal, fungal, and most bacterial steryl glycosides have a β‑glycosidic linkage, but in a few bacterial species, there is an α-linkage.
Similarly, the nature of the fatty acid component in the acyl sterol glycosides can vary, as well as the hydroxyl group to which they are linked. However, it is usually position 6 of the glucose moiety. In potato tubers, for example, the 6'-palmitoyl-β-D-glucoside of β-sitosterol is the major species, while the corresponding linoleate derivative predominates in soybeans. Usually, the sterol acyl-glycosides are present at concentrations that are two- to tenfold greater than those of the non-acylated forms. They are known to be located in the plasma membrane, tonoplasts, and endoplasmic reticulum.
Biosynthesis involves the reaction of free sterols with a glucose unit catalyzed by a sterol glycosyltransferase, or by the reaction of the sterol with uridine diphosphoglucose (UDP-glucose) and UDP-glucose:sterol glucosyltransferase on the cytosolic side of the plasma membrane. The acyl donor for acyl sterol glycoside synthesis is not acyl-coenzyme A but is believed to be a glycerolipid. Steryl β-D-glycoside hydrolases have been characterized from plants that reverse this reaction, but no fatty acyl hydrolase activity for sterol acyl-glycosides is yet known. One route to the biosynthesis of glucosylceramides in plants involves the transfer of the glucose moiety of sterol glycosides to ceramide.
The functions of sterol glycosides and sterol acyl-glycosides are slowly being revealed, and they are believed to be significant components of the plasma membrane that associate with sphingolipids in raft-like domains; the esterified form, especially, may be involved in the adaptation of plant membranes to low temperatures and other stresses. It is possible that they have a role in signal transmission through membranes, and they are reported to be beneficial in the response to pathogens. It seems probable that sterol glycosides are oriented with the sterol moiety buried in the hydrophobic core of the lipid bilayer, with the sugar located in the plane of the polar head groups of the membrane, while with sterol acyl-glycosides, both the sterol moiety and the fatty acid chain are embedded in the hydrophobic core of the membrane. Sitosterol-β-D-glucoside in the plasma membrane is believed to be the primer molecule for cellulose synthesis in plants, as in cotton (Gossypium arboreum) fiber, where it may be required for the initiation of glucan polymerization. The sterol is eventually removed from the polymer by a specific cellulase enzyme; the multimeric cellulase synthase is believed to be stabilized by sterols in the plasma membrane.
Sterol glycosides appear to be essential for the pathogenicity of certain fungi and some bacteria, and ergosterol glycosides are especially troublesome components of plant fungal pathogens. Sterol glycosides have only rarely been reported from organisms other than plants and fungi. However, some bacteria, such as the gram-negative bacterium Helicobacter pylori and Borrelia burgdorferi, the causative agent of Lyme disease, produce cholesterol glucoside from host cholesterol. On the other hand, cholesteryl glucoside has been identified as a natural component of a few animal tissues. Acting as immunoadjuvants, sterol glycosides are reported to be efficacious in protecting animal hosts against lethal Cryptococcal infections. In the human diet, sterol glycosides offer potential benefits similar to those of free sterols, as they inhibit the absorption of cholesterol from the gut and reduce plasma cholesterol levels. The fatty acids are removed from sterol acyl-glycosides by enzymes in the intestine.
Some species of monocotyledons contain complex steroidal saponins, which consist of an aglycone based on a triterpenoid furostanol or spirostanol skeleton (derived from cholesterol) and an oligosaccharide chain of two to five hexose or pentose moieties attached to the 3β-hydroxyl group of the sterol. These can interact with cholesterol in plant membranes to form insoluble complexes, which increase membrane permeability.
Ergosterol and Other Sterols in Yeasts and Fungi
Yeasts and fungi, together with microalgae and protozoa, can contain a wide range of different sterols. However, ergosterol ((22E)‑ergosta-5,7,22-trien-3β-ol) is the most common sterol in fungi and yeast. It is accompanied by other sterols not normally abundant in higher plants, including cholesterol, 24-methyl cholesterol, 24-ethyl cholesterol, and brassicasterol, depending on the species. In Saccharomyces cerevisiae, which is widely studied as a model species of yeasts, ergosterol is the most abundant sterol (ca. 12% of all lipids), with the highest levels in the plasma membrane (up to 40% of the lipids or 90% of the total cell sterols). Its structure is shown in Figure \(\PageIndex{45}\).
Like cholesterol, and in contrast to plant sterols, it is synthesized in the endoplasmic reticulum via lanosterol as the key intermediate, followed by zymosterol. However, the pathway diverges at this stage to produce fecosterol en route to ergosterol (see the reading list below for further details). Ergosterol is transported to other organelles within the cell in a non-vesicular manner by two families of evolutionarily conserved sterol-binding proteins - 'Osh' and 'Lam', which can optimize the sterol composition of cell membranes rapidly under conditions of stress. As in humans, a Niemann-Pick protein, NCR1, integrates sterols into the lysosomal membrane before further distribution as part of the mechanism of sterol homeostasis. Some antifungal drugs target ergosterol, either by binding to it to cause damaging cellular leakage or by preventing its synthesis from lanosterol.
Many mutants defective in ergosterol biosynthesis have been isolated, yielding a wealth of information on the features of the sterol molecule required for its structural role in the membranes of yeast and fungi. Ergosterol stabilizes the liquid-ordered phase in the same manner as cholesterol and also forms raft microdomains with sphingolipids in membranes, whereas lanosterol does not. It is also evident that ergosterol has multiple functions in regulating yeast growth.
Under some conditions, especially those that retard growth, a high proportion of the sterols in yeasts can be in esterified form, where they are stored in lipid droplets. Ergosterol esters are synthesized in yeast by enzymes (ARE1 and ARE2), which are related to ACAT-1 and ACAT-2 that perform this function in animals, and both transfer an activated fatty acid to the hydroxyl group at the C3-position of a sterol molecule. In addition, specific sterol ester hydrolases that catalyze the reverse reaction have been characterized in yeasts, with two enzymes located in lipid droplets and one at the plasma membrane. Many fungal species and slime molds contain steryl glycosides (ergosteryl β-monoglucopyranosides in the former). Still, they are present at very low levels only in the widely studied yeast Saccharomyces cerevisiae.
Most fungi conjugate the 3β-hydroxyl group of ergosterol with aspartate in an RNA-dependent reaction catalyzed by an ergosteryl-3β-O-L-aspartate synthase, with the reverse reaction using a dedicated hydrolase. A phylogenomic study has shown that this pathway is conserved across higher fungi (except S. cerevisiae), including pathogens. It has been suggested that these reactions constitute a homeostasis system with potential impacts on membrane remodeling, trafficking, antimicrobial resistance, and pathogenicity.
Bacterial Sterols
Hopanoids replace sterols in many species of bacteria. It has long been recognized that some bacteria take up cholesterol and other sterols from host animals for use as membrane constituents. Indeed, an external source of sterols is required for growth in species of Mycoplasma. Additionally, numerous reports have been published regarding the biosynthesis of sterols by various bacterial species. However, a significant proportion of these appear to have been discounted due to fungal contamination. In particular, the possibility of sterol biosynthesis in cyanobacteria has been a subject of controversy, and molecular biology studies have yet to detect the presence of the required enzyme, squalene epoxide cyclase.
That said, there is good evidence that at least a few species of prokaryotes can synthesize sterols de novo. Among the eubacteria, certain methylotrophs, such as Methylobacterium and Methylosphaera species, produce mono- and dimethyl sterols, including lanosterol. Similarly, some soil bacteria produce 4‑desmethylsterols. It has now been established through gene sequence studies that a few bacteria contain enzymes of the sterol biosynthesis pathway, such as oxidosqualene cyclase. However, as these enzymes have no obvious evolutionary link, it seems probable that they were acquired via lateral transfer from eukaryotes.
Plant Sterols in the Human Diet
The absorption of dietary plant sterols and stanols in humans is low, ranging from 0.02% to 3.5%, compared to cholesterol, which is absorbed at rates of 35-70%. Although there are similar amounts of plant sterols and stanols in an average Western diet, their absorption rates are significantly lower. The explanation is believed to be that the Niemann-Pick C1-like protein 1 (NPC1L1), which is responsible for cholesterol absorption in enterocytes, does not efficiently take up plant sterols. Two transporters, ABCG5 and ABCG8, redirect any that are absorbed back into the intestinal lumen. In rare cases, elevated levels of plant sterols in plasma serve as markers for a hereditary lipid storage disease (phytosterolemia) caused by mutations in the enterocyte transporters. Among many symptoms, accelerated atherosclerosis is often reported, although the reasons for this are not clear. There is evidence that while plant sterols can substitute for cholesterol in maintaining membrane function in mammalian cells, they can exert harmful effects by disrupting cholesterol homeostasis. This may be particularly relevant to the brain, as phytosterols can cross the blood-brain barrier; however, they cannot be enzymatically oxidized due to the alkyl moiety on carbon 24. In contrast, dietary supplements of plant sterols have been reported to have anti-cancer effects.
Substantial amounts of phytosterols are available as by-products of the refining of vegetable oils and of tall oil from the wood pulp industry. As it appears that they can inhibit the uptake of cholesterol from the diet and thereby reduce the levels of this in the plasma low-density lipoproteins, there is an increasing interest in such commercial sources of plant sterols to be added as "nutraceuticals" to margarines and other foods. Hydrogenated phytosterols or "stanols" are also used for this purpose. Studies suggest they are as effective as sterols in reducing LDL cholesterol. The consensus among experts in the field, including the FDA in the USA, is that such dietary supplements indeed have the effects claimed. Such claims can be used in the advertising of commercial products, with the important caveat that there are no randomized, controlled clinical trial data to establish the ensuing health benefits, especially concerning cardiovascular disease. Other pharmacological effects are under investigation, and there may be beneficial effects for the development of the human fetus and newborn, as well as for the treatment of non-alcoholic steatohepatitis, inflammatory bowel diseases, and allergic asthma.
It is unclear whether oxy-phytosterols are generated in animal tissues; however, those produced enzymatically or non-enzymatically can enter the food chain, particularly when they are formed during cooking. Although they are not efficiently absorbed, 7-keto-sitosterol and 7-keto-campesterol have been detected in human plasma and have the potential to exert various biological effects. For example, they have pro-atherogenic and pro-inflammatory properties in animal models.
Summary
This chapter explores the central role of cholesterol—a key sterol in animal tissues—and its esterified forms in cellular structure, metabolism, and signaling. We begin by examining cholesterol’s unique polycyclic structure, characterized by its rigid, planar tetracyclic nucleus, a flexible iso‑octyl side chain, and a 3β‑hydroxyl group. These structural features not only determine its orientation within lipid bilayers but also facilitate its interaction with other membrane lipids, influencing membrane fluidity, thickness, and the formation of specialized microdomains known as lipid rafts.
The biosynthesis of cholesterol occurs via the mevalonate pathway, starting from acetyl‑CoA. Key intermediates include 3‑hydroxy-3‑methylglutaryl (HMG)‑CoA, which is reduced by HMG‑CoA reductase—the rate-limiting enzyme and target of statins—to form mevalonate. Subsequent reactions generate isopentenyl pyrophosphate, which polymerizes to form squalene. Squalene is then converted to lanosterol and ultimately to cholesterol through a series of demethylation, isomerization, and reduction reactions. This process is highly energy‐demanding and tightly regulated to balance endogenous synthesis with dietary uptake.
Cholesterol is differentially distributed among cellular membranes: it is most abundant in the plasma membrane, where it can comprise 30–50% of the lipid content, and less so in the endoplasmic reticulum and mitochondria. This uneven distribution is critical for maintaining membrane properties and for regulating the lateral organization of proteins, including those involved in cell signaling. Cholesterol also modulates membrane permeability and interacts with sphingolipids to form liquid‑ordered domains that serve as platforms for signaling and protein sorting.
To prevent the deleterious effects of cholesterol overaccumulation, cells employ sophisticated regulatory mechanisms. Cholesterol homeostasis is maintained via feedback regulation—mediated by sterol regulatory element-binding proteins (SREBPs) and their escort protein SCAP—as well as through the action of oxysterols, which can act as ligands for nuclear receptors such as the liver X receptor (LXR). In addition, excess cholesterol is esterified by enzymes such as lecithin:cholesterol acyltransferase (LCAT) in the plasma and acyl-CoA:cholesterol acyltransferase (ACAT) within cells, forming cholesterol esters that are stored in lipid droplets or packaged into lipoproteins for transport throughout the body.
Cholesterol serves as the precursor for several vital biomolecules, including bile acids, vitamin D, and steroid hormones. For instance, in the skin, 7‑dehydrocholesterol is converted into vitamin D3 under ultraviolet light, initiating a cascade that produces the active hormone calcitriol, essential for calcium and phosphorus homeostasis.
Overall, this chapter emphasizes the intricate balance of cholesterol synthesis, transport, and catabolism that is essential for cellular function and organismal health. Dysregulation of these processes is linked to a range of diseases—from cardiovascular disorders and neurodegeneration to metabolic and developmental abnormalities—highlighting the clinical significance of sterol metabolism and the potential of targeting these pathways for therapeutic intervention.




.png?revision=1&size=bestfit&width=494&height=366)
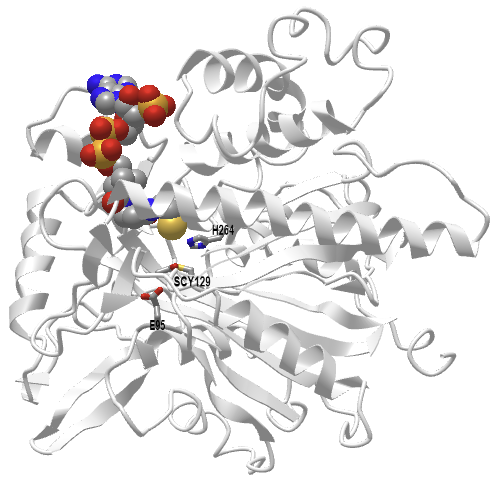
.png?revision=1&size=bestfit&width=328&height=344)
.png?revision=1&size=bestfit&width=377&height=282)
.png?revision=1&size=bestfit&width=360&height=326)
.png?revision=1&size=bestfit&width=340&height=382)