22.1: Overview of Nitrogen Metabolism - Nitrogenase
- Page ID
- 15178
\( \newcommand{\vecs}[1]{\overset { \scriptstyle \rightharpoonup} {\mathbf{#1}} } \)
\( \newcommand{\vecd}[1]{\overset{-\!-\!\rightharpoonup}{\vphantom{a}\smash {#1}}} \)
\( \newcommand{\dsum}{\displaystyle\sum\limits} \)
\( \newcommand{\dint}{\displaystyle\int\limits} \)
\( \newcommand{\dlim}{\displaystyle\lim\limits} \)
\( \newcommand{\id}{\mathrm{id}}\) \( \newcommand{\Span}{\mathrm{span}}\)
( \newcommand{\kernel}{\mathrm{null}\,}\) \( \newcommand{\range}{\mathrm{range}\,}\)
\( \newcommand{\RealPart}{\mathrm{Re}}\) \( \newcommand{\ImaginaryPart}{\mathrm{Im}}\)
\( \newcommand{\Argument}{\mathrm{Arg}}\) \( \newcommand{\norm}[1]{\| #1 \|}\)
\( \newcommand{\inner}[2]{\langle #1, #2 \rangle}\)
\( \newcommand{\Span}{\mathrm{span}}\)
\( \newcommand{\id}{\mathrm{id}}\)
\( \newcommand{\Span}{\mathrm{span}}\)
\( \newcommand{\kernel}{\mathrm{null}\,}\)
\( \newcommand{\range}{\mathrm{range}\,}\)
\( \newcommand{\RealPart}{\mathrm{Re}}\)
\( \newcommand{\ImaginaryPart}{\mathrm{Im}}\)
\( \newcommand{\Argument}{\mathrm{Arg}}\)
\( \newcommand{\norm}[1]{\| #1 \|}\)
\( \newcommand{\inner}[2]{\langle #1, #2 \rangle}\)
\( \newcommand{\Span}{\mathrm{span}}\) \( \newcommand{\AA}{\unicode[.8,0]{x212B}}\)
\( \newcommand{\vectorA}[1]{\vec{#1}} % arrow\)
\( \newcommand{\vectorAt}[1]{\vec{\text{#1}}} % arrow\)
\( \newcommand{\vectorB}[1]{\overset { \scriptstyle \rightharpoonup} {\mathbf{#1}} } \)
\( \newcommand{\vectorC}[1]{\textbf{#1}} \)
\( \newcommand{\vectorD}[1]{\overrightarrow{#1}} \)
\( \newcommand{\vectorDt}[1]{\overrightarrow{\text{#1}}} \)
\( \newcommand{\vectE}[1]{\overset{-\!-\!\rightharpoonup}{\vphantom{a}\smash{\mathbf {#1}}}} \)
\( \newcommand{\vecs}[1]{\overset { \scriptstyle \rightharpoonup} {\mathbf{#1}} } \)
\(\newcommand{\longvect}{\overrightarrow}\)
\( \newcommand{\vecd}[1]{\overset{-\!-\!\rightharpoonup}{\vphantom{a}\smash {#1}}} \)
\(\newcommand{\avec}{\mathbf a}\) \(\newcommand{\bvec}{\mathbf b}\) \(\newcommand{\cvec}{\mathbf c}\) \(\newcommand{\dvec}{\mathbf d}\) \(\newcommand{\dtil}{\widetilde{\mathbf d}}\) \(\newcommand{\evec}{\mathbf e}\) \(\newcommand{\fvec}{\mathbf f}\) \(\newcommand{\nvec}{\mathbf n}\) \(\newcommand{\pvec}{\mathbf p}\) \(\newcommand{\qvec}{\mathbf q}\) \(\newcommand{\svec}{\mathbf s}\) \(\newcommand{\tvec}{\mathbf t}\) \(\newcommand{\uvec}{\mathbf u}\) \(\newcommand{\vvec}{\mathbf v}\) \(\newcommand{\wvec}{\mathbf w}\) \(\newcommand{\xvec}{\mathbf x}\) \(\newcommand{\yvec}{\mathbf y}\) \(\newcommand{\zvec}{\mathbf z}\) \(\newcommand{\rvec}{\mathbf r}\) \(\newcommand{\mvec}{\mathbf m}\) \(\newcommand{\zerovec}{\mathbf 0}\) \(\newcommand{\onevec}{\mathbf 1}\) \(\newcommand{\real}{\mathbb R}\) \(\newcommand{\twovec}[2]{\left[\begin{array}{r}#1 \\ #2 \end{array}\right]}\) \(\newcommand{\ctwovec}[2]{\left[\begin{array}{c}#1 \\ #2 \end{array}\right]}\) \(\newcommand{\threevec}[3]{\left[\begin{array}{r}#1 \\ #2 \\ #3 \end{array}\right]}\) \(\newcommand{\cthreevec}[3]{\left[\begin{array}{c}#1 \\ #2 \\ #3 \end{array}\right]}\) \(\newcommand{\fourvec}[4]{\left[\begin{array}{r}#1 \\ #2 \\ #3 \\ #4 \end{array}\right]}\) \(\newcommand{\cfourvec}[4]{\left[\begin{array}{c}#1 \\ #2 \\ #3 \\ #4 \end{array}\right]}\) \(\newcommand{\fivevec}[5]{\left[\begin{array}{r}#1 \\ #2 \\ #3 \\ #4 \\ #5 \\ \end{array}\right]}\) \(\newcommand{\cfivevec}[5]{\left[\begin{array}{c}#1 \\ #2 \\ #3 \\ #4 \\ #5 \\ \end{array}\right]}\) \(\newcommand{\mattwo}[4]{\left[\begin{array}{rr}#1 \amp #2 \\ #3 \amp #4 \\ \end{array}\right]}\) \(\newcommand{\laspan}[1]{\text{Span}\{#1\}}\) \(\newcommand{\bcal}{\cal B}\) \(\newcommand{\ccal}{\cal C}\) \(\newcommand{\scal}{\cal S}\) \(\newcommand{\wcal}{\cal W}\) \(\newcommand{\ecal}{\cal E}\) \(\newcommand{\coords}[2]{\left\{#1\right\}_{#2}}\) \(\newcommand{\gray}[1]{\color{gray}{#1}}\) \(\newcommand{\lgray}[1]{\color{lightgray}{#1}}\) \(\newcommand{\rank}{\operatorname{rank}}\) \(\newcommand{\row}{\text{Row}}\) \(\newcommand{\col}{\text{Col}}\) \(\renewcommand{\row}{\text{Row}}\) \(\newcommand{\nul}{\text{Nul}}\) \(\newcommand{\var}{\text{Var}}\) \(\newcommand{\corr}{\text{corr}}\) \(\newcommand{\len}[1]{\left|#1\right|}\) \(\newcommand{\bbar}{\overline{\bvec}}\) \(\newcommand{\bhat}{\widehat{\bvec}}\) \(\newcommand{\bperp}{\bvec^\perp}\) \(\newcommand{\xhat}{\widehat{\xvec}}\) \(\newcommand{\vhat}{\widehat{\vvec}}\) \(\newcommand{\uhat}{\widehat{\uvec}}\) \(\newcommand{\what}{\widehat{\wvec}}\) \(\newcommand{\Sighat}{\widehat{\Sigma}}\) \(\newcommand{\lt}{<}\) \(\newcommand{\gt}{>}\) \(\newcommand{\amp}{&}\) \(\definecolor{fillinmathshade}{gray}{0.9}\)-
Describe the Nitrogen Cycle:
- Explain the overall biological nitrogen cycle, including the roles of inorganic (e.g., ammonium, nitrate, nitrite) and organic (e.g., amino acids, nucleotides) nitrogen compounds.
- Differentiate between aerobic and anaerobic processes (e.g., nitrification, denitrification, anammox, and ammonification) and discuss their ecological significance.
-
Understand Nitrogen Fixation:
- Outline the process of nitrogen fixation by soil bacteria and archaea, including the conversion of atmospheric N₂ into biologically available ammonium (NH₄⁺).
- Compare biological nitrogen fixation with the industrial Haber–Bosch process, emphasizing the kinetic and thermodynamic challenges involved.
-
Analyze Thermodynamic and Kinetic Challenges:
- Explain why, despite favorable room-temperature thermodynamics, N₂ is kinetically inert due to its strong triple bond.
- Discuss how high pressure and the use of catalysts (in both industrial and biological systems) overcome these kinetic barriers.
-
Examine the Structure of Nitrogenase:
- Identify the key protein components of the nitrogenase complex, including the reductase (Fe-protein) and the catalytic (MoFe-protein) subunits.
- Describe the metal clusters (F-cluster, P-cluster, and the iron–molybdenum (M) cluster) and their proposed roles in electron transfer and N₂ reduction.
-
Detail the Role of ATP and Electron Transfer:
- Explain how ATP hydrolysis in the Fe-protein drives the electron transfer necessary for nitrogen fixation.
- Illustrate the coupling between ATP hydrolysis, conformational changes, and the sequential delivery of electrons from the reductase to the MoFe-protein.
-
Interpret the Lowe–Thorneley Model:
- Summarize the stepwise accumulation of electrons and protons in nitrogenase as described by the Lowe–Thorneley kinetic model.
- Define the E₄ “Janus” intermediate, and discuss its significance in binding N₂ and enabling reductive elimination of H₂.
-
Apply Organometallic Reaction Principles:
- Describe the concepts of oxidative addition, reductive elimination, and migratory insertion as they apply to the nitrogenase mechanism.
- Relate these reaction types to the biochemical transformations involved in the reduction of N₂ to NH₃.
-
Evaluate Experimental Evidence and Mechanistic Models:
- Critically assess experimental data (e.g., isotope labeling and deuterium incorporation studies) that support the proposed intermediates and mechanisms in nitrogenase catalysis.
- Discuss how mutations (e.g., at key amino acid residues) affect substrate access and the overall catalytic process.
-
Connect Nitrogen Metabolism to Ecosystem Function:
- Explain how microbial nitrogen fixation impacts plant nutrition and overall ecosystem productivity.
- Discuss the environmental implications of anthropogenic nitrogen inputs (e.g., fertilizer runoff) and how they disrupt the natural balance of the nitrogen cycle.
-
Integrate Knowledge Across Disciplines:
- Synthesize concepts from organic chemistry, enzymology, and inorganic/organometallic chemistry to explain the biochemical challenges and solutions inherent in N₂ fixation.
- Recognize the broader significance of studying nitrogen metabolism—not only in human biochemistry but also in understanding global ecological processes.
These learning goals aim to help students grasp both the biochemical mechanisms of nitrogen fixation and the broader environmental context in which these processes operate.
Introduction
Organic chemistry is usually described as the chemistry of carbon-containing molecules. But isn't that definition a bit carbon-centric, especially since the prevalence of oxygen-containing molecules is staggering? What about nitrogen? We live in a dinitrogen-rich atmosphere (80%), and all classes of biomolecules (lipids, carbohydrates, nucleic acids, and proteins) contain nitrogen. Dinitrogen is very stable due to its triple bond and nonpolar nature. We rely on a few organisms to fix N2 from the atmosphere to form ammonium (NH4+), which, through nitrification and denitrification, can form nitrite (NO2-), nitrate (NO3-), nitric oxide (NO), and nitrous oxide (N2O), the latter being a potent greenhouse gas. We'll concentrate on the metabolic fate of amino groups in amino acids and proteins in the next section. Before exploring their fates, look at Figure \(\PageIndex{1}\), which shows an overall view of the biological nitrogen cycle. The study of biochemistry should encompass more than homo sapiens and expand to the ecosystem in which we are such a small but damaging part.

Figure \(\PageIndex{1}\): Nitrogen Cycle
Let's break down the diagram from a biochemical perspective. There are aerobic and anaerobic processes (conducted by bacteria). Nitrogen-containing substances include both inorganic (ammonium, nitrate, nitrite) and organic (amino acids, nucleotides, etc) molecules. The reactions shown are both oxidative and reductive (note: the oxidation numbers of the nitrogen atoms in the molecules are shown in red). Most of the reactions are carried out underground by bacterial and Archaeal microorganisms.
Here are some of the major reactions:
- N2 fixation (a reduction): N2 from the air is converted by bacteria to ammonium (NH4+) by the enzyme nitrogenase of soil prokaryotes. The energetically disfavored reaction requires lots of ATPs. Ammonium that has been made can then be taken up by primary producers, such as plants, and incorporated into biomolecules like amino acids, which animals consume. For those who may still believe that people have marginal effects on our biosphere, consider this. We may soon fix more N2 to NH3 through the industrial Haber-Bosch reaction (used for fertilizer and explosive production), which is all made by the biosphere. Much of the nitrogen in use comes from the Haber-Bosch reaction. The excess NH4+ (upwards of 50%) produced industrially and entering the soil through fertilizers (mostly as NH4NO3) has overwhelmed nature's ability to balance the nitrogen cycle and is not taken up by plants. It is metabolized by microorganisms to nitrite and nitrate.
- Nitrification: Ammonium is converted to nitrite by ammonia-oxidizing aerobic microorganisms and further to nitrate by a separate group of nitrite-oxidizing aerobic bacteria. Here are the reactions (Rx 1 and 2) to produce nitrate through a hydroxylamine intermediate, followed by the formation of nitrate (Rx 3).
NH3 + O2 + 2e- → NH2OH + H2O Rx 1
NH2OH + H2O → NO2- + 5H+ + 4e- Rx 2
NO2- + 1/2 O2 → NO3- Rx 3
These added ions exceed soil capacity and end up in runoff water, polluting our rivers and lakes.
- Denitrification: This anaerobic reaction pathway converts nitrate into N2. Here is the net reaction:
2NO3- + 10e- + 12H+ → 2N2 + 6H2O
- Anammox reaction: This recently discovered bacterial anaerobic reaction pathway converts ammonium and nitrate to nitrogen gas (N2). Here is the net reaction
NO2- + NH3+ → N2 + 2H2O
- Ammonification (not to be confused with mummification) occurs when plants and animals decompose, releasing ammonium back into the soil for reuse by plants and microbes.
These reactions are shown in the abbreviated Nitrogen Cycle shown in Figure \(\PageIndex{2}\).

Figure \(\PageIndex{2}\): Abbreviated Nitrogen Cycle
Nitrogen metabolites are essential nutrients for plants, playing a crucial role in regulating plant growth (primary productivity) and maintaining biodiversity in the biosphere. All living organisms require feedstocks to produce energy and as substrates for biosynthetic reactions. Which is used depends on the organism. Plants are primary producers, so they use their synthesized carbohydrates for both energy production and biosynthesis. For carnivores, proteins and their derived amino acids serve as an energy source through oxidation and as biosynthetic precursors for various cellular processes. For omnivorous organisms, the source of energy depends on the "fed" state. With abundant food resources, carbohydrates and lipids serve as the primary sources of energy. Unlike carbohydrates and lipids, which can be stored as glycogen and triacylglycerols for future use, excess protein and its associated amino acids can not be stored, so amino acids can be eliminated or used for oxidative energy.
In the fed state, carbohydrates are the main source, while in the unfed state, lipids take a predominant role. Under starving conditions, the organism's own proteins are broken down and utilized for oxidative energy production and any remaining biosynthesis. In diseased states like diabetes, which can be likened to a state of starvation in the presence of abundant carbohydrates, both lipids and amino acids become sources of energy.
How are amino acids in animals oxidatively metabolized? Several pathways could be employed to achieve this. Still, it would seem logical that NH4+ would be removed, and the carbons in the remaining molecule would eventually enter glycolysis or the TCA cycle in the form of ketoacids. NH4+ is toxic in high concentrations. Ammonium is not oxidized to nitrite or nitrates in humans, unlike what occurs in soil with the help of microorganisms. It can be recycled back into nucleotides or amino acids, and excess amounts are eliminated from the organism. Both processes must be highly controlled. We will turn our attention to the oxidation of amino acids in the next section.
Nitrogenase: An Introduction
Beauty is in the eye of the beholder.
As the domain of biochemistry covers the entire biological world, the extent of coverage of a given topic in textbooks can depend, in part, on the interest and experiences of the author(s) presenting the material. Is relevance a metric that should determine coverage? If so, books focused on human or medical biochemistry would surely omit the topic of photosynthesis. If topics are selected based on their importance for life, then photosynthesis must surely be covered. If so, then nitrogenase must also be included. Considering the degree of chemical difficulty in a chemical reaction and the remarkable eloquence of the evolved biochemistry solution, both photosynthesis and nitrogen fixation must be presented. Although nitrogen fixation is a reductive reaction, it shares strong similarities with the oxygen-evolving complex in photosynthesis. They catalyze enormously important redox reactions that involve an abundant atmospheric gas, utilizing a very complicated and unique inorganic metallic cofactor that evolution has selected as uniquely suited for the task.
Every first-year student of chemistry can draw the Lewis structure of dinitrogen, N2, which contains a triple bond and a lone pair on each nitrogen. If Lewis structures resonate with them, they should be able to state that the triple bond makes N2 extraordinarily stable, thus explaining why we can breathe an atmosphere containing 80% N2 without dying. If they have taken biology, they are also aware that very few biological organisms can utilize N2 as a substrate, as this requires breaking bonds between the nitrogen atoms, a chemical process reserved for nitrogen-fixing bacteria found in rhizomes of certain plants. Lastly, they likely memorized that high pressure and temperature are needed in the Haber-Bosch process to convert N2 and H2 to ammonia, NH3. As with any scientific advance, the Haber-Bosch process has brought both harm (it is used for explosive weapons) and benefit (in the form of fertilizers). This process now fixes enough N2 in the form of fertilizers to support half of the world’s population, with nitrogenase supporting the rest. Efforts are underway to genetically modify plants to produce nitrogenase, eliminating the need for fertilizers, but this may create unforeseen problems of its own.
You might be surprised to find out that at room temperature, the equilibrium constant favors ammonia formation, hence ΔG0 < 0. The reaction is favored enthalpically as it is exothermic at room temperature. It is disfavored entropically, as should be evident from the balanced equation:
\[\ce{N2(g) + 3H2(g) → 2 NH3(g)}. \nonumber \]
The thermodynamic parameters for the reaction (per mol) are ΔH° = –46.2 kJ, ΔS° = –389 J K–1, and ΔG° = –16.4 kJ at 298 K
The entropy is negative since the reaction proceeds from 4 to 2 molecules. From an enthalpy perspective, raising the temperature of an exothermic reaction drives it in the reverse direction. If you increase the pressure, you shift the equilibrium to the side that has the fewest number of molecules.
If the reaction is favored thermodynamically at room temperature, why doesn’t it proceed? This story sounds familiar, as this same descriptor applies to the oxidation of organic molecules with dioxygen. There, we showed using MO theory that the reaction is kinetically slow. Same with NH3 formation. A superficial way to view this is that we must break bonds in the stable N2 to initiate the reaction, resulting in a high activation energy and sluggish reaction kinetics.
One could jump-start the reaction by raising the temperature, but that would slow an exothermic reaction. The Keq (or KD) and ΔG0 are functions of temperature, and for this reaction, the reaction becomes disfavored at higher temperatures. The solution Bosch found was high pressure, forcing the reaction to the side that has fewer molecules of gas, and high temperature to overcome the activation energy barrier and make the reaction kinetically feasible. A complex metal catalyst (magnetite - Fe3O4 -with metal oxides like CaO and Al2O3, which prevent reduction of the Fe with H2) provides an absorptive surface to bring reagents together and facilitate bond breaking in H2 and N2.
In photosynthesis, the oxygen-evolving complex (OEC), which contains Mn, Fe, S, and Ca, is used to oxidize another highly stable and ubiquitous molecule, H2O. Now we explore the amazing mechanisms behind the nitrogenase complex, which fixes N2 to form NH3 in a reductive fashion.
What might be needed to drive this reaction biologically? You might surmise the list to include:
- a source of energy, most likely ATP, to facilitate this complex reaction;
- a source of electrons as the N atoms move from an oxidation state of 0 in elemental N to 3- in NH3; this source turns out to be a protein called flavodoxin or ferredoxin. Of course, these electrons also have an interesting origin before they are in the electron carriers of these proteins.
- some pretty amazing metal centers to accept and donate electrons in a controlled way; these centers are mostly FeS clusters with an additional cluster containing molybdenum (Mo). The clusters are named F, P, and M
- a source of hydrogen; you might have guessed correctly that it’s not H2 gas (from where would that come?), but H+ ions, which are pretty ubiquitously available.
- a net reaction that is different from the Haber-Bausch process (N2 + 3H2 → 2 NH3).
Here is the actual reaction catalyzed by nitrogenase:
\[\ce{N2 + 8e^{-} + 16ATP + 8H^{+} → 2NH3 + H2 + 16ADP + 16P_i}. \nonumber \]
Let’s think a bit about the reaction. As electrons are added, the attraction between the nitrogen atoms must decrease. Eventually, bonds between them must be broken. Protons could be easily added to maintain charge neutrality. A basic mechanism might involve intermediates as shown in Figure \(\PageIndex{3}\).
Figure \(\PageIndex{3}\): Possible intermediates in the conversion of dinitrogen to ammonia by nitrogenase
Nitrogenase can also interact with other small molecules with triple bonds, including C≡O: and H-C≡C-H.
The Structure of Nitrogenase
Nitrogenase is a multiprotein complex in which the functional biological unit is built from two sets of the following dimeric structures:
- a homodimer of subunits, E and F, which have binding sites for the mobile carrier of electrons (the protein ferredoxin or flavodoxin), ATP, and an FeS cofactor (4Fe-4S, called the F cluster) that accepts electrons. These subunits are hence referred to as the nitrogenase reductase subunits.
- a heterodimer of alpha and beta subunits. The a (alpha chain) binds the 8Fe-7S F cluster and the Fe-S-Mo M cluster. These subunits comprise the (di)nitrogenase catalytic subunits. The iron-molybdenum M cluster is in the α subunit and is where N2 reduction occurs. The P-cluster is between the α and β subunits and facilitates electron flow between the Fe-protein (F cluster) and FeMo-cofactor (M cluster)
For clarity, one-half of the overall structure of the protein complex with bound ATP and metal centers is shown in Figure \(\PageIndex{4}\).
Figure \(\PageIndex{4}\): Nitrogenase structure (4wzb)
This half-structure consists of a homodimer of the reductase monomers and a heterodimer of nitrogenase subunits.
Figure \(\PageIndex{5}\) shows an interactive iCn3D model of the half structure of nitrogenase complex from Azotobacter vinelandii (4WZB) (long load).
Figure \(\PageIndex{5}\): Nitrogenase complex from Azotobacter vinelandii (4WZB). (Copyright; author via source). Click the image for a popup or use this external link: https://structure.ncbi.nlm.nih.gov/i...zPNjfPRwq8MSeA (long load))
The structure is color-coded in a fashion similar to Figure 4. The F cluster is labeled as SF4, the P cluster as CLF (FE(8)-S(7) cluster), and the M cluster as ICS (iron-sulfur-molybdenum cluster with interstitial carbon).
The reductase subunits (called Av2 in Azotobacter vinelandii), accept electrons from ferredoxin and is where the ATP analog, ACP (phosphomethylphosphonic acid adenylate ester), and the 4Fe-4S F cluster is bound. The nitrogenase subunits (called Av1 in Azotobacter vinelandii) convert N2 to NH3, and it is where the 8Fe-7S P cluster, FeMo (C Fe7 Mo S9) M Cluster is bound.
An enhanced view of the bound cofactors and ATP is shown in the same spatial orientation in Figure \(\PageIndex{6}\)below.
Figure \(\PageIndex{6}\): Bound cofactor metal clusters and ATP in nitrogenase
You can easily imagine the direction of the flow of electrons from the F cluster to the P cluster to the M cluster.
The metal centers are shown in Figure \(\PageIndex{7}\) in more detail, both in line and space fill views.
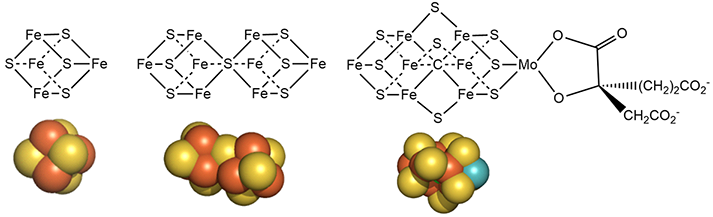
Figure \(\PageIndex{7}\): Detailed structures of the metal cofactors in nitrogenase
Mo is bound to 3 sulfur ions and oxygen from 3-hydroxy-3-carboxy-adipic acid as shown in Figure 7 above.
The M cluster contains an interstitial carbide ion derived from the -CH3 group attached to the sulfur of S-adenosyl-methionine (SAM), allowing the carbide to be labeled with either 13C or 14C for mechanistic studies. These labeled carbides are neither exchanged nor used as a substrate during the enzyme's catalytic turnover. Hence, it appears that the carbide likely stabilizes the M cluster. It will not be shown in the figures below, which provide more detailed mechanisms.
We will focus on two key features of the reaction mechanism: ATP hydrolysis and the flow of electrons and protons to N2 as it is reduced to NH3.
The answer is that O2 destroys the enzyme. Plants are aerobic. So how does it function? See Chapter 32.17: Part 4 - Fixing Nitrogen Fixation for details!
ATP hydrolysis
ATP binds in the reductase subunit (AV2) where ferredoxin brings in electrons, and where the F metal cluster is bound. Much as GTP hydrolysis controls conformational change and subunit dissociation in the heterotrimeric Gαβγ in signal transductions, ATP hydrolysis in reductase (AV2) subunits drives not only electron transfer but also dissociation/reassociation of the reductase and nitrogenase catalytic subunits. It appears that two ATPs are hydrolyzed per electron transferred from the F cluster to the M cluster. Since the oxidation numbers of N are 0 and -3 in N2 and NH3, respectively, sequential rounds of ATP hydrolysis and dissociation/reassociation occur. Note that the Fe-protein hydrolyzes ATP only when bound to the MoFe-protein.
The overall reaction involves the reduction of N2 to two molecules of NH3 at the FeMo cofactor. It involves the reductive elimination of hydrides bridged by Fe ions (Fe-H-Fe) in a reaction that also produces H2 as a by-product. We'll describe this complex reaction next.
Nitrogenase Reaction: Part 1 - Addition of Electrons and Protons
The sequential path of electrons from the reductase subunit containing the F cluster to the P and M clusters in the nitrogenase subunit should be apparent from the figures above. We will concentrate on the binding of N2 and how it receives electrons from the M cluster. Figure \(\PageIndex{8}\) shows the FeMo-cofactor and some adjacent amino acid residues. Mo is labeled but not shown in spacefill. HCA is a bound molecule of 3-hydroxy-3-carboxy-adipic acid, which interacts with Mo. The carbide is shown in the middle as the green sphere and labeled CX.
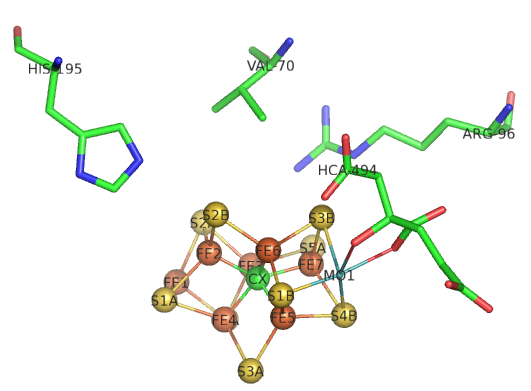
Figure \(\PageIndex{8}\): Side chains near the M cluster
If Val 70 is mutated to Ile, a substrate appears not to access the cluster, suggesting that N2 may interact with the top part of the structure, with the residues shown acting as gatekeepers. His side chains are often found at enzyme active sites, so you might expect His 195 to be a general acid/base. Mutations lead to drastic losses in the reduction of N2. His 195 is involved in hydrogen bonds to sulfur S2B and bridges Fe2 and Fe3 in Figure 8, where reduction of N2 likely occurs. If His 195 moves, it can form a short H bond between the imidazole N and an H bond to HFe2. If the ring is rotated 1800, no proton transfer occurs from the surface. It appears that His195 might be involved in the first N2 protonation event.
The Lowe and Thorneley (LT) model has been proposed as a mechanism for the reduction of dinitrogen. In this model, an electron and a proton are added to the oxidized form of the enzyme (Eo) to produce E1. This is repeated three more times to form sequentially, E2, E3, and E4. Only then does N2 bind, and the reduction of N2 occurs. Two of the added electrons are accepted by H+ ions, which form H2, which is liberated on N2 binding. Hence, only 6 electrons are added to the actual N2 molecule, in agreement with the change in oxidation numbers discussed above. The Lowe and Throneley model is shown in Figure \(\PageIndex{9}\).
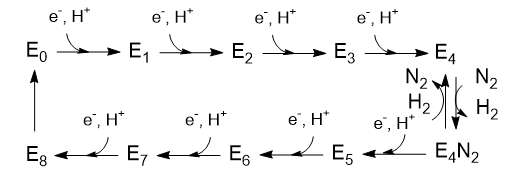
Figure \(\PageIndex{9}\): Lowe and Throneley model for electron and proton additions in nitrogenase
The crystal structure shows 2 ATP analogs bound to the reductase subunit. The stoichiometry of the reaction indicates that 16 ATP are used. Simple math suggests that 2 ATP are cleaved to support the entry of one electron into the complex, assuming 8 electrons are transferred (6 to N2 and 2 to protons to form H2).
Part 1 - E1-E4: A potential structure for the E4 intermediate is shown in Figure \(\PageIndex{10}\).
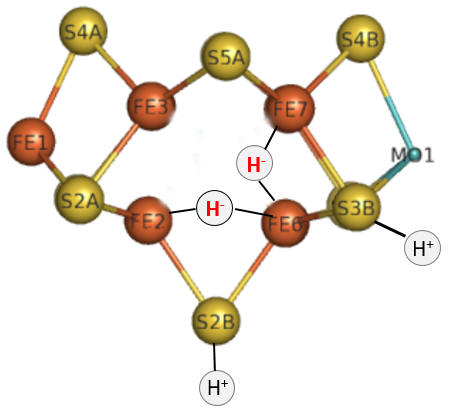
Figure \(\PageIndex{10}\): The E4 Janus intermediate in the reduction of N2.
Note that the central carbide is not shown. This is often referred to as the Janus intermediate, as it is situated halfway through the catalytic cycle. It is named after Janus, the Roman god of beginnings and transitions, and has been associated with gates, doors, doorways, and passages. Janus is typically shown with two faces, one looking to the future and one to the past (image below: DOI:10.1590/2177-6709.21.1.018-023.oin. License CC BY 4.0 Creative Commons Attribution 4.0 International)

The hydrides bridge 2 Fe ions, so these are examples of three-center, two-electron bonds. The H+ ions in Figure 10 balance the charge from the hydrides.
How does this reaction occur? We must look to organometallic chemistry to help us understand the mechanism of this and subsequent steps. Hydride equivalents have been added to the metals, associated with the oxidation of metal ions in the center. This particular reaction is called an oxidative addition. Presumably, the sulfur ions act as Lewis bases as they gain protons from a Lewis acid, probably His 195.
Oxidative addition reactions
Figure \(\PageIndex{11}\) shows oxidative additions to metal centers for three different types of reactants. L is the ligand(s) and M is the metal.
Figure \(\PageIndex{11}\): Three different types of oxidative addition reactions (after Schaller, http://employees.csbsju.edu/cschaller/ROBI1.htm)
In oxidative insertion, the oxidation state of the metal ion increases, hence the name oxidative. The hydrogens are now hydrides. Note the example of H2 insertion, which is similar to the proposed hydride additions to the M cluster. Oxidative reduction occurs most readily when the two oxidation states of the metal ion are stable. It is likewise favored for metal centers that are not sterically hindered (makes sense if A-B is to be added) and if A-B has a low bond dissociation energy.
One way to study reaction intermediates is to trap them in place. If N2 can't access the binding site and the temperature is reduced, the accumulated hydrides (and for charge balance, the H+s) in E4 might leave in the opposite reaction, reductive elimination. We will discuss this below, as the reaction goes back to E1. In the elimination, they can form H2 as the metal gains back electrons in a reduction. The Val70Ile discussed above would allow an intermediate to be trapped.
Nitrogenase Reaction: Part 2 - Reduction of N2
Step E4 to E5 seems a bit bizarre as H2 gas is released. This would seem to waste ATP, but we should trust that evolution has led to this mechanism for a reason. This mechanism, the reverse of oxidative addition, is another classic organometallic reaction, reductive elimination.
Reductive elimination reactions
In this reaction, a molecule is eliminated or expelled from the complex as the metal ion is reduced, gaining two electrons. Figure \(\PageIndex{12}\) shows reductive elimination. Reductive elimination occurs most readily in higher oxidation state metal centers, which can be stabilized on reduction. It occurs most readily from electron-rich ligands, and if the other surrounding ligands are bulky. The dissociating species must also be cis to each other in the transition metal complex so they can form a bond with each other when they leave.
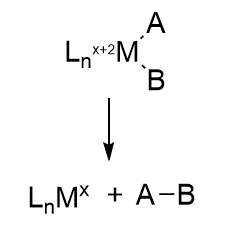
Figure \(\PageIndex{12}\): Reductive Elimination reaction (after Schaller, ibid)
Oxidative addition and reductive elimination (OA/RE) reactions at metal centers are often coupled together in organometallic catalytic cycles, much like a histidine can act as a general acid and then accept a proton back as a general base to complete the catalytic cycle. In the OA/RE reactions at metal centers, some rearrangements or other modifications can also occur. Think about it. The FeS clusters must return to their original oxidation state after the complete LT cycle. We will encounter another organometallic reaction after the addition of N2, migratory insertion, in the second half of the reaction. Another advantage of coupling OA/RE is that the positive charge or oxidation state on the transition metal complex does not become too high, which would be unstable. Making a cation with a positive charge more positive becomes more difficult, much as removing a second proton from a polyprotic acid is more difficult than removing the first (as reflected in the higher pKa for removal of the second proton).
Is H2(g) released? To study this, investigators have used alternative substrates such as acetylene, HC≡CH (similar to N≡N), in the presence of D2 and N2 in an aqueous system. It is helpful to review Figure \(\PageIndex{9}\) again.
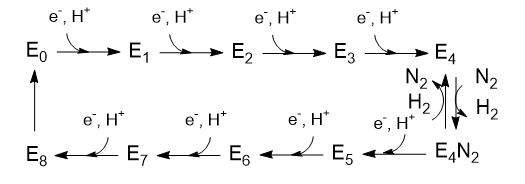
Figure \(\PageIndex{9}\): Lowe and Throneley model for electron and proton additions in nitrogenase
The acetylene was reduced and formed C2H2D2 and C2H3D. Hence, E4 must have had two Ds in it, and E2 probably had one. These results support the reversible reductive elimination mechanism for the E4 to E4:N2 reaction above. Previously, it had been shown that H+ are reduced by D2 in the presence of D2 and N2 in an aqueous system, so these results are consistent. In addition, deuterium from D2 is not incorporated into products (C2H2D2, C2H3D, or HD) in the absence of N2.
Let's return for a moment to the bridging hydrides as shown again Figure \(\PageIndex{10}\).
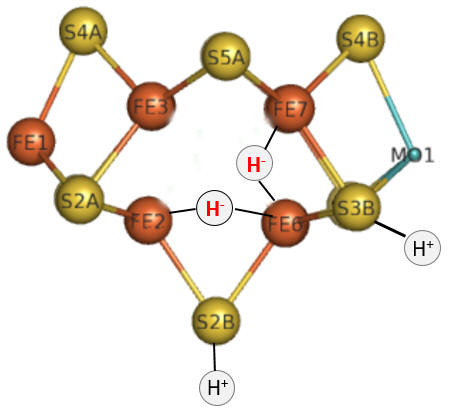
Figure \(\PageIndex{10}\): The E4 Janus intermediate in the reduction of N2
It appears likely that the reductive elimination of the two proximal bridging hydrides is the mechanism for the formation of H2. The bridging hydrides, which are strong bases, are much less likely to be protonated than if the hydrides were terminal. A simple and competing protonation reaction could also form H2, and if that occurred, the reducing equivalents of the bridging hydrides would be lost. Hence, the bridging hydrides (shared by two metal centers) are more stable and can "wait" for the incoming N2 reactants before their reducing equivalents are lost. They may convert to terminal hydrides eventually to facilitate substrate reduction (hydrogenation).
The Janus intermediate E4 is now positioned to bind N2 and release H2. For each H- that binds to the M cluster, two H+s bind to the M cluster sulfides for electrostatic stabilization.
Migratory Insertions
We need to consider one last common type of reaction at a metal center, migratory insertions (MI). In an MI reaction, a group attached to a metal ion center is transferred to another group attached to the same metal. Figure \(\PageIndex{13}\) shows four examples of MI reactions.
Figure \(\PageIndex{13}\): Migratory insertion reactions at metal centers
Panel (a) shows a generic MI reaction. Panel (b) shows the interaction of carbon monoxide, :C≡O: (isoelectronic and analogous to :N≡N:), with a metal center. Panel (c) shows how a hydride could engage in 1:1 insertion as it shifts and covalently bonds to the first atom of another ligand bound to the metal center. Note that metal does not have to have a negative charge. This could theoretically be important for the reduction of N2 bound as a ligand through a coordinate covalent (dative) bond to the metal. Panel (d) shows the migration of an alkyl group.
We will see that the MI reaction is involved in adding Hs to N2, starting not with N2 but with the N2H2 stage as the intermediates insert into an Fe-H bond. N2 is not reactive to the insertion of a hydride, as is carbon monoxide, CO, which provides a positive oxygen to facilitate electron flow during the insertion. In addition, the oxygen becomes neutral after the reaction.
This provides a compelling explanation for how nature selected the FeMo cluster for nitrogen fixation. The interaction of two hydrides requires a 4 Fe face (coordinated with the carbon) that allows for the storage of reducing equivalents for the initial reduction of N2. The large M cluster is more stable and effectively held together by a central carbide anion. This also allows the metal centers to never change their oxidation state by more than one charge unit.
The source of the protons to form N2H2 comes directly from the two H2 attached to the two sulfurs. They can't come from the hydrides, which are released as H2. The seemingly wasteful reductive elimination of H2 and energy is required to allow the kinetically unreactive N2 molecule to bind to the reduced and activate 4Fe face, which is also electrostatically facilitated by the two bound protons in what has been called a push (reductive)-pull (protonation) reaction. This first step in N2 reduction to N2H2 is the hardest.
Oxidation States of Nitrogenase Fe Centers
First Half: It would be difficult to assign specific oxidation states to each Fe ion in the M complex. Instead, we can assign relative changes in the oxidation states as the reaction proceeds from E0 to E4. In each step of the LT model, 1 electron is added. We will first assign this to an average Fe ion, M0, with an arbitrarily assigned oxidation state of 0. Upon the addition of 1 electron, the oxidation state would go from M0 to M-1 as the metal is reduced. The M-1 state is then oxidized as an electron is transferred to H+, and when 2 electrons are transferred, a single H- is made. The diagram in Figure \(\PageIndex{14}\) illustrates the change in oxidation state as the system transitions from E0 to E4.
Figure \(\PageIndex{14}\): Change in oxidation state in going from E0 to E4
The red boxes highlight thermodynamic cycle-like steps, which show how changes in the redox state of the Fe ions (M) could be visualized. Note that in going from E0 to E4, the actual oxidation state of M changes back and forth from 0 to +1 and finally back to 0. That is quite amazing given that 4 electrons have been added. Note also that in the red box going from step E0 to E1, M goes from -1 to +1, which corresponds to our description of an oxidative addition when the metal center loses two electrons. This mechanism suggests that nitrogenase can be considered a "hydride storage device."
Second Half (facing forward to production NH3):
How does N2 initially interact with E4? It must depend on how the hydrides are released as H2, which evidence shows occurs by reductive elimination (re) and not hydride protonation (hp). On addition, the N2 is very quickly converted to diazene, HN=NH, with the departing H2 taking with it 2 H+s and 2 electrons (or reducing equivalents). These events could occur as shown in Figure \(\PageIndex{15}\).
Figure \(\PageIndex{15}\): Reaction mechanism for the formation of N2H2
Now, with N2 bound as diazene (N2H2) and H2 released, the rest of the reaction could occur as shown below. One new step, a migratory insertion, is shown in Figure \(\PageIndex{16}\).
Figure \(\PageIndex{16}\): Reaction mechanism for the conversion N2H2 to NH3.
The two halves of the reaction are similar, with bridging hydrides utilized - the E4 Janus intermediate links the two halves together.
Summary
This chapter explores the central role of nitrogen in biochemistry and ecology by examining the biological nitrogen cycle and, in depth, the mechanism of biological nitrogen fixation via nitrogenase.
Key Points:
-
The Nitrogen Cycle in Nature:
The chapter begins by challenging the carbon-centric view of organic chemistry, emphasizing that nitrogen—present in both inorganic forms (ammonium, nitrate, nitrite) and organic biomolecules (amino acids, nucleotides)—is equally essential. It provides an overview of the nitrogen cycle, highlighting how aerobic and anaerobic bacterial processes (nitrification, denitrification, anammox, and ammonification) transform atmospheric N₂ into bioavailable forms and back again, thereby supporting ecosystem productivity. -
Biological Nitrogen Fixation:
Despite the thermodynamic favorability at room temperature (exothermic with a negative ΔG°), the reduction of the highly stable N₂ molecule is kinetically hindered by its strong triple bond. Nitrogen fixation is accomplished by specialized prokaryotes that use the enzyme nitrogenase to convert atmospheric N₂ into ammonium (NH₄⁺) under ambient conditions, powered by the hydrolysis of ATP. -
Structure and Function of Nitrogenase:
Nitrogenase is a complex, multi-protein enzyme composed of two main components:- The Reductase (Fe-protein): A homodimer containing a 4Fe–4S cluster that accepts electrons from electron carriers (e.g., ferredoxin or flavodoxin) and hydrolyzes ATP to drive electron transfer.
- The Catalytic Component (MoFe-protein): A heterodimer housing two key metal clusters—the P-cluster and the iron–molybdenum (M) cofactor. The M cluster, which includes molybdenum, iron, sulfur, and an interstitial carbide, is the site of N₂ binding and reduction.
-
Kinetic Mechanism and the Lowe–Thorneley Model:
The chapter details the stepwise accumulation of electrons and protons in nitrogenase, as described by the Lowe–Thorneley kinetic model. In this model, the enzyme cycles through intermediate states (E₀ to E₄), culminating in the “Janus” intermediate (E₄). At this stage, bridging hydrides accumulate and, upon N₂ binding, undergo reductive elimination to release H₂. This critical step “activates” N₂, allowing its reduction to ammonia. -
Organometallic Reaction Principles in Nitrogenase:
The enzyme’s mechanism is illuminated by concepts from organometallic chemistry. Key reactions such as oxidative addition (where H₂ is “added” to the metal center, raising its oxidation state) and reductive elimination (where H₂ is released as the metal center is reduced) are central to the enzyme’s function. Additionally, migratory insertion may play a role during later steps of N₂ reduction. Together, these processes illustrate how nature has evolved a sophisticated strategy to overcome the kinetic barriers associated with breaking the strong N≡N bond. -
Ecological and Biochemical Significance:
Finally, the chapter connects these biochemical processes to broader ecological implications. Nitrogen fixation by microorganisms supplies essential nitrogen to plants, underpinning global primary productivity. This understanding also offers insight into the impact of human activities (such as industrial fertilizer use) on the natural nitrogen cycle.
In Summary:
This chapter integrates organic, inorganic, and organometallic chemistry principles to explain how nature fixes atmospheric nitrogen—a process essential for life on Earth. By dissecting the structure and mechanism of nitrogenase, the text illustrates how energy (from ATP), electrons, and sophisticated metal cofactors work together to overcome kinetic barriers and drive the reduction of N₂ to ammonia. Moreover, it places these biochemical insights within the context of global nutrient cycles and environmental sustainability, underscoring the interconnectedness of molecular-level processes and ecosystem dynamics.
This summary provides a comprehensive overview designed to help advanced undergraduate biochemistry students integrate concepts from thermodynamics, enzymology, and inorganic chemistry with ecological and environmental perspectives.




.png?revision=1&size=bestfit&width=426&height=362)