W2018_Bis2A_Lecture07_reading
- Page ID
- 25319
\( \newcommand{\vecs}[1]{\overset { \scriptstyle \rightharpoonup} {\mathbf{#1}} } \)
\( \newcommand{\vecd}[1]{\overset{-\!-\!\rightharpoonup}{\vphantom{a}\smash {#1}}} \)
\( \newcommand{\dsum}{\displaystyle\sum\limits} \)
\( \newcommand{\dint}{\displaystyle\int\limits} \)
\( \newcommand{\dlim}{\displaystyle\lim\limits} \)
\( \newcommand{\id}{\mathrm{id}}\) \( \newcommand{\Span}{\mathrm{span}}\)
( \newcommand{\kernel}{\mathrm{null}\,}\) \( \newcommand{\range}{\mathrm{range}\,}\)
\( \newcommand{\RealPart}{\mathrm{Re}}\) \( \newcommand{\ImaginaryPart}{\mathrm{Im}}\)
\( \newcommand{\Argument}{\mathrm{Arg}}\) \( \newcommand{\norm}[1]{\| #1 \|}\)
\( \newcommand{\inner}[2]{\langle #1, #2 \rangle}\)
\( \newcommand{\Span}{\mathrm{span}}\)
\( \newcommand{\id}{\mathrm{id}}\)
\( \newcommand{\Span}{\mathrm{span}}\)
\( \newcommand{\kernel}{\mathrm{null}\,}\)
\( \newcommand{\range}{\mathrm{range}\,}\)
\( \newcommand{\RealPart}{\mathrm{Re}}\)
\( \newcommand{\ImaginaryPart}{\mathrm{Im}}\)
\( \newcommand{\Argument}{\mathrm{Arg}}\)
\( \newcommand{\norm}[1]{\| #1 \|}\)
\( \newcommand{\inner}[2]{\langle #1, #2 \rangle}\)
\( \newcommand{\Span}{\mathrm{span}}\) \( \newcommand{\AA}{\unicode[.8,0]{x212B}}\)
\( \newcommand{\vectorA}[1]{\vec{#1}} % arrow\)
\( \newcommand{\vectorAt}[1]{\vec{\text{#1}}} % arrow\)
\( \newcommand{\vectorB}[1]{\overset { \scriptstyle \rightharpoonup} {\mathbf{#1}} } \)
\( \newcommand{\vectorC}[1]{\textbf{#1}} \)
\( \newcommand{\vectorD}[1]{\overrightarrow{#1}} \)
\( \newcommand{\vectorDt}[1]{\overrightarrow{\text{#1}}} \)
\( \newcommand{\vectE}[1]{\overset{-\!-\!\rightharpoonup}{\vphantom{a}\smash{\mathbf {#1}}}} \)
\( \newcommand{\vecs}[1]{\overset { \scriptstyle \rightharpoonup} {\mathbf{#1}} } \)
\(\newcommand{\longvect}{\overrightarrow}\)
\( \newcommand{\vecd}[1]{\overset{-\!-\!\rightharpoonup}{\vphantom{a}\smash {#1}}} \)
\(\newcommand{\avec}{\mathbf a}\) \(\newcommand{\bvec}{\mathbf b}\) \(\newcommand{\cvec}{\mathbf c}\) \(\newcommand{\dvec}{\mathbf d}\) \(\newcommand{\dtil}{\widetilde{\mathbf d}}\) \(\newcommand{\evec}{\mathbf e}\) \(\newcommand{\fvec}{\mathbf f}\) \(\newcommand{\nvec}{\mathbf n}\) \(\newcommand{\pvec}{\mathbf p}\) \(\newcommand{\qvec}{\mathbf q}\) \(\newcommand{\svec}{\mathbf s}\) \(\newcommand{\tvec}{\mathbf t}\) \(\newcommand{\uvec}{\mathbf u}\) \(\newcommand{\vvec}{\mathbf v}\) \(\newcommand{\wvec}{\mathbf w}\) \(\newcommand{\xvec}{\mathbf x}\) \(\newcommand{\yvec}{\mathbf y}\) \(\newcommand{\zvec}{\mathbf z}\) \(\newcommand{\rvec}{\mathbf r}\) \(\newcommand{\mvec}{\mathbf m}\) \(\newcommand{\zerovec}{\mathbf 0}\) \(\newcommand{\onevec}{\mathbf 1}\) \(\newcommand{\real}{\mathbb R}\) \(\newcommand{\twovec}[2]{\left[\begin{array}{r}#1 \\ #2 \end{array}\right]}\) \(\newcommand{\ctwovec}[2]{\left[\begin{array}{c}#1 \\ #2 \end{array}\right]}\) \(\newcommand{\threevec}[3]{\left[\begin{array}{r}#1 \\ #2 \\ #3 \end{array}\right]}\) \(\newcommand{\cthreevec}[3]{\left[\begin{array}{c}#1 \\ #2 \\ #3 \end{array}\right]}\) \(\newcommand{\fourvec}[4]{\left[\begin{array}{r}#1 \\ #2 \\ #3 \\ #4 \end{array}\right]}\) \(\newcommand{\cfourvec}[4]{\left[\begin{array}{c}#1 \\ #2 \\ #3 \\ #4 \end{array}\right]}\) \(\newcommand{\fivevec}[5]{\left[\begin{array}{r}#1 \\ #2 \\ #3 \\ #4 \\ #5 \\ \end{array}\right]}\) \(\newcommand{\cfivevec}[5]{\left[\begin{array}{c}#1 \\ #2 \\ #3 \\ #4 \\ #5 \\ \end{array}\right]}\) \(\newcommand{\mattwo}[4]{\left[\begin{array}{rr}#1 \amp #2 \\ #3 \amp #4 \\ \end{array}\right]}\) \(\newcommand{\laspan}[1]{\text{Span}\{#1\}}\) \(\newcommand{\bcal}{\cal B}\) \(\newcommand{\ccal}{\cal C}\) \(\newcommand{\scal}{\cal S}\) \(\newcommand{\wcal}{\cal W}\) \(\newcommand{\ecal}{\cal E}\) \(\newcommand{\coords}[2]{\left\{#1\right\}_{#2}}\) \(\newcommand{\gray}[1]{\color{gray}{#1}}\) \(\newcommand{\lgray}[1]{\color{lightgray}{#1}}\) \(\newcommand{\rank}{\operatorname{rank}}\) \(\newcommand{\row}{\text{Row}}\) \(\newcommand{\col}{\text{Col}}\) \(\renewcommand{\row}{\text{Row}}\) \(\newcommand{\nul}{\text{Nul}}\) \(\newcommand{\var}{\text{Var}}\) \(\newcommand{\corr}{\text{corr}}\) \(\newcommand{\len}[1]{\left|#1\right|}\) \(\newcommand{\bbar}{\overline{\bvec}}\) \(\newcommand{\bhat}{\widehat{\bvec}}\) \(\newcommand{\bperp}{\bvec^\perp}\) \(\newcommand{\xhat}{\widehat{\xvec}}\) \(\newcommand{\vhat}{\widehat{\vvec}}\) \(\newcommand{\uhat}{\widehat{\uvec}}\) \(\newcommand{\what}{\widehat{\wvec}}\) \(\newcommand{\Sighat}{\widehat{\Sigma}}\) \(\newcommand{\lt}{<}\) \(\newcommand{\gt}{>}\) \(\newcommand{\amp}{&}\) \(\definecolor{fillinmathshade}{gray}{0.9}\)Matter and Energy in Biology
Matter and Energy
The concepts of matter and energy are essential to all scientific disciplines. Yet, while ubiquitous and fundamental, these concepts are often among the most confounding for students. Take the concept of energy. The term is used in a variety of contexts in everyday life:
- “Can we move the couch tomorrow? I don’t have the energy.”
- “Hey dude! Turn the light off. We need to conserve energy.”
- “This is a great energy drink.”
In many sciences classes, students are told that energy comes in different forms (i.e. kinetic, thermal, electrical, potential, etc.), making it difficult to understand exactly what energy “is”. In class, the concept of energy is also associated with a multitude of different equations, each with different variables, but that somehow all seem to end up having units of work. Hold on! Work? I thought we were talking about energy?!
Given all the different contexts and sometimes seemingly contradictory treatments and definitions, it’s not hard to understand why these topics seem challenging for many students and in some cases end up turning them off of the fields that make heavy use of these ideas. While the concepts of matter and energy are most often associated with chemistry and physics, they are nevertheless central ideas in biology and we don’t shy away from this in BIS2A. Our instructional goals, however, are to help students develop a conceptual framework that will help them use the concepts of matter and energy to:
- successfully describe biological reactions and transformations;
- create models and hypotheses for “how things work” in biology that explicitly include matter and energy and;
- be scientifically correct and transfer these ideas to new problems as well as other disciplines.
While there may be a couple of energy-related equations to learn and use in BIS2A, the main focus of the course will be on the robust development of the concepts of energy and matter and their use in the interpretation of biological phenomena.
Motivation for Learning About Matter and Energy
Discussions about matter and energy make many, but not all, BIS2A students a little apprehensive. After all, aren’t these topics that belong in chemistry or physics? However, the transformations of matter and energy transfer are not phenomena reserved for the chemists and physicists or even scientists and engineers more generally. Understanding, conceptualizing, and doing some basic accounting of transformations of matter and transfers of energy are fundamental skills regardless of occupation or academic training. The scientist may need more rigorous and systematic descriptions of these transformations than the artist but both make use of these skills at various points of their personal and or professional lives. Take the following examples:
Example 1: Matter and Energy Transformation in Global Warming
Let us for a moment consider a topic that affects us all, global warming. At its core lies a relatively simple model that is based on our understanding of energy in solar radiation, the transfer of this energy with matter on the Earth, and the role and cycling of key carbon containing gases in the Earth's atmosphere. In simple terms solar energy hits the earth and transfers energy to its surface, heating it. Some of this energy is transferred back into space. However, depending on the concentration of carbon dioxide (and other so-called greenhouse gases) different amounts of this energy may become “trapped” in the Earth’s atmosphere. Too little carbon dioxide and relatively little energy/heat is trapped - the Earth freezes and becomes inhospitable for life. Too much carbon dioxide and too much heat is trapped - the Earth overheats and becomes inhospitable for life. It stands to reason, therefore, that mechanisms (biological or other) that influence the levels of carbon dioxide in the atmosphere may be important to consider in the story of global warming and that developing a good understanding of global warming phenomena requires one to trace the flow of the carbon and oxygen (matter) through their different forms and the mechanisms by which energy is transferred to and from different components of the system.
Example 2: Muscle Contraction
Let us now consider a more personal example, the flexing of an arm starting from an extended position and ending in a flexed position. Like most processes, this one can be described and understood at various levels of detail: from the anatomical point of view where the system consists of muscles, skin, and bones to the molecular where the system is composed of individual interacting biomolecules. At whatever level of detail, if we want to create a story describing this process we know that: (a) the description must include an accounting for what happened to the matter in the system (this includes the change in position of the molecules making up the various parts of the arm and the fuel “burned” to move it) and (b) that some fuel was burned to initiate the movement and therefore, that any description of the process must also include an accounting change in the energy of the system. In simpler terms, this is really just saying that if you want to describe a process where something has happened, you need to describe what happened to the “stuff” in the system and what happened to the energy in the system to make the process happen.
We can't possibly cover all examples of matter and energy transfer in BIS2A. But, we will explore these issues often and practice describing transformations that happen in Nature with a structured and explicit attention to what is happening to the matter and energy in a system as it changes. We will do this exercise across different structural levels in biology, from the molecular level (like a single chemical reaction) to more large-scale and abstracted models like nutrient cycling in the environment. We will practice this skill by using a pedagogical tool we call “The Energy Story”. Be prepared to participate!
Energy
Energy is a central concept in all sciences. Energy is a property of a system. While it can be neither created nor destroyed, understanding the transfer of energy around physical systems is a key component of understanding how and why things change. In the following sections, we are going to explore some basic concepts associated with common transformations in biology and chemistry: the solubility of various biomolecules, the making and breaking of chemical bonds, the transfer of electrons, the transfer of energy to and from light, and the transfer of energy as heat. In class, many of the discussions will happen in the context of the Energy Story rubric, so when we consider a reaction of transformation, we will be interested in precisely defining the system in question and trying to account for all the various transfers of energy that occur within the system, making sure that we abide by the Law of Conservation of Energy.
There are plenty of examples where we use the concept of energy in our everyday lives to describe processes. A bicyclist can bike to get to campus for class. The act of moving herself and her bicycle from point A to point B can be explained to some degree by examining the transfers energy that take place. We can look at this example through a variety of lenses, but, as biologists, we more than likely want to understand the series of events that explain how energy is transferred from molecules of food, to the coordinated activity of biomolecules in a bicyclist's flexing muscle, and finally, to the motion of the bike from point A to point B. To do this, we need to be able to talk about various ways in which energy can be transferred between parts of a system and where it is stored or transferred out of the system. In the next section, we will also see the need to consider how that energy is distributed among the many microstates (molecular states) of the system and its surroundings.
How we will approach conceptualizing energy
In BIS2A we will think about energy with a "stuff" metaphor. Note, however, that energy is NOT a substance, it is rather a property of a system. But we will think of it, in some sense, as property that can be stored in a part of a physical system and transferred or "moved" from one storage place to another. The idea is to reinforce the idea that energy maintains its identity when transferred—it is not changing forms per se. This in turn also encourages us to make sure that energy always has a home and that we account for all of the energy in a system before and after a transformation; it does not just get "made" or get "lost" (both ideas contradict of the Law of Conservation of Energy). When energy is being transferred, we therefore must identify where it is coming from and where it is going—all of it! Again, we can't just have some getting lost. When energy is transferred, there must be some mechanism associated with that transfer. Let's think about that to help us explain some of the phenomena we're interested in. That mechanism is part of the "how" that we are often interested in understanding. Finally, if we talk about transfer, we must realize that both components, the part of the physical system that gave up energy and the part of the system that received that energy, are changed from their initial states. We should make sure that we are looking at all of the components of a system for changes in energy when examining a transformation.
Energy sources
Ultimately, the source of energy for many processes occurring on the Earth's surface comes from solar radiation. But as we will see, biology has been very clever at tapping a variety of forms of energy to construct and maintain living beings. As we move through this course, we will explore a variety of energy sources and the ways in which biology has devised to transfer energy from these fuels.
Energy in chemical reactions
Chemical reactions involve a redistribution of energy within the reacting chemicals and with their environment. So, like it or not, we need to develop some models that can help us to describe where energy is in a system (perhaps how it is "stored"/distributed) and how it can be moved around in a reaction. The models we develop will not be overly detailed in the sense that they would satisfy a hard-core chemist or physicist with their level of technical detail, but we expect that they should still be technically correct and not form incorrect mental models that will make it difficult to understand the "refinements" later.
In this respect, one of the key concepts to understand is that we are going to think about energy being transferred between parts of a system. We'll try not to think about it as being transformed. The distinction between "transfer" and "transform" is important. The latter gives the impression that energy is a property that exists in different forms, that it gets reshaped somehow. One problem with the "transform" language is that it is difficult to reconcile with the idea that energy is being conserved (according to the first law of thermodynamics) if it is constantly changing form. How can the entity be conserved if it is no longer the same thing? Moreover, the second law of thermodynamics tells us that no transformation conserves all energy in a system. If energy is getting "transformed," how can it be conserved?
So, instead, we are going to approach this issue by transferring and storing energy between different parts of a system and thus think about energy as a property that can get redistributed. That'll hopefully make the accounting of energy easier.
CAUTION:
If we are going to think about transferring energy from one part of a system to another, we also need to be careful about NOT treating energy like a substance that moves like a fluid or "thing." Rather, we need to appreciate energy simply as a property of a system that can be measured and reorganized but that is neither a "thing" nor something that is at one time in one form then later in another.
Since we will often be dealing with transformations of biomolecules, we can start by thinking about where energy can be found/stored in these systems. We'll start with a couple of ideas and add more to them later.
Let us propose that one place that energy can be stored is in the motion of matter. For brevity, we'll give the energy stored in motion a name: kinetic energy. Molecules in biology are in constant motion and therefore have a certain amount of kinetic energy (energy stored in motion) associated with them.
Let us also propose that there is a certain amount of energy stored in the biomolecules themselves and that the amount of energy stored in those molecules is associated with the types and numbers of atoms in the molecules and their organization (the number and types of bonds between them). The discussion of exactly where the energy is stored in the molecules is beyond the scope of this class, but we can approximate it by suggesting that a good proxy is in the bonds. Different types of bonds may be associated with storing different amounts of energy. In some contexts, this type of energy storage could be labeled potential energy or chemical energy. With this view, one of the things that happens during the making and breaking of bonds in a chemical reaction is that the energy is transferred about the system into different types of bonds. In the context of an Energy Story, one could theoretically count the amount of energy stored in the bonds and motion of the reactants and the energy stored in the bonds and energy of the products.
In some cases, you might find that when you add up the energy stored in the products and the energy stored in the reactants that these sums are not equal. If the energy in the reactants is greater than that in the products, where did this energy go? It had to get transferred to something else. Some will certainly have moved into other parts of the system, stored in the motion of other molecules (warming the environment) or perhaps in the energy associated with photons of light. One good, real-life example is the chemical reaction between wood and oxygen (reactants) and it's conversion to carbon dioxide and water (products). At the beginning, the energy in the system is largely in the molecular bonds of oxygen and the wood (reactants). There is still energy left in the carbon dioxide and water (products) but less than at the beginning. We all appreciate that some of that energy was transferred to the energy in light and heat. This reaction where energy is transferred to the environment is termed exothermic. By contrast, in some reactions, energy will transfer in from the environment. These reactions are endothermic.
The transfer of energy in or out of the reaction from the environment is NOT the only thing that determines whether a reaction will be spontaneous or not. We'll discuss that soon. For the moment, it is important to get comfortable with the idea that energy can be transferred among different components of a system during a reaction and that you should be able to envision tracking it.
Thermodynamics
Thermodynamics is concerned with describing the changes in systems before and after a change. This usually involves a discussion about the energy transfers and its dispersion within the system. In nearly all practical cases, these analyses require that the system and its surroundings be completely described. For instance, when discussing the heating of a pot of water on the stove, the system may includes the stove, the pot, and the water and the environment or surroundings may include everything else. Biological organisms are what are called open systems; energy is transferred between them and their surroundings.
The First Law of Thermodynamics
The first law of thermodynamics deals with the total amount of energy in the universe. It states that this total amount of energy is constant. In other words, there always has been, and always will be, exactly the same amount of energy in the universe.
According to the first law of thermodynamics, energy may be transferred from place to place, but it cannot be created or destroyed. Energy transfers take place around us all the time. Light bulbs transfer energy from electrical power stations into heat and photons of light. Gas stoves transfer energy stored in the bonds of chemical compounds into heat and light. (Heat, by the way, is the amount of energy transferred from one system to another because of a temperature difference.)
Plants perform one of the most biologically useful energy transfers on earth: they transfer energy in the photons of sunlight into the chemical bonds of organic molecules. In every one of these cases, energy is neither made nor destroyed, and we must try to account for all of the energy when we examine some of these reactions.
The First Law and the Energy Story
The first law of thermodynamics is deceptively simple. Students often understand that energy cannot be created or destroyed. Yet, when describing an energy story of a process they often make the mistake of saying things such as "energy is produced from the transfer of electrons from atom A to atom B." While most of us will understand the point the student is trying to make, the wrong words are being used. Energy is not made or produced; it is simply transferred. To be consistent with the first law, when telling an energy story, make sure that you try to explicitly track all of the places that ALL of the energy in the system at the start of a process goes by the end of a process.
The Second Law of Thermodynamics
An important concept in physical systems is that of entropy. Entropy is related to the ways in which energy can be distributed or dispersed within the particles of a system. The Second Law of Thermodynamics states that entropy is always increasing in a system and its surroundings (that is, everything outside the system).
This idea helps explain the directionality of natural phenomena. In general, the notion is that the directionality comes from the tendency for energy in a system to move towards a state of maximal dispersion. The Second Law, therefore, implies that in any transformation, we should look for an overall increase in entropy (or dispersion of energy), somewhere. As dispersion of energy in a system or its surroundings increases, the ability of the energy to be directed towards work decreases.
Keep in mind: you will find many examples in which the entropy of a system decreases locally. However, according to the Second Law, the entropy of the entire universe can never decrease. This must mean that there is an equal or greater increase in entropy somewhere else in the surroundings (most likely in a closely-connected system) that compensates for the local decrease.
The entropy of a system can increase when:
- the system gains energy;
- a change of state occurs from solid to liquid to gas;
- a mixing of substances occurs;
- the number of particles increases during a reaction.
Note: possible discussion
Does the second law say that entropy is conserved?
Note: possible discussion
Biological systems, on the surface, seem to defy the Second Law of Thermodynamics. They don't. Why?
Figure 1. An increase in disorder can happen in different ways. An ice cube melting on a hot sidewalk is one example. Here, ice is displayed as a snowflake, with organized, structured water molecules forming the snowflake. Over time, the snowflake will melt into a pool of disorganized, freely moving water molecules. (Source)
Note: possible discussion
A fine point. Figure 1 above discusses order and disorder and shows that this is somehow related to a change in entropy (ΔS). It is common to describe entropy as a measure of order as a way to simplify the more concrete description relating entropy to the number of states in which energy can be dispersed in a system. While the idea of measuring order to define entropy has some flaws, it is sometimes a useful, if imperfect, proxy. Consider the figure above. Here, order serves as a good proxy for approximating the number of ways to distribute energy in the system. Can you describe why this is the case?
If we consider the first and second laws together, we come to a useful conclusion. Whenever energy is transferred or redistributed within a system, entropy must increase. This increase in entropy is related to how "useful" the energy is to do work. Recall again that this energy generally becomes less and less available as entropy increases.
We conclude that while all of the energy must be conserved, if the required change increases entropy, it means that some of the energy will become distributed in a way that makes it less useful for work. In many cases, particularly in biology, some of the increase in entropy can be chalked up to a transfer of energy to heat in the environment.
Free Energy
If we want to describe transformations, it is useful to have a measure of (a) how much energy is in a system, (b) the dispersal of that energy within the system and, of course, (c) how these change between the start and end of a process. The concept of free energy, often referred to as Gibbs free energy or free enthalpy (abbreviated with the letter G), in some sense, does just that. Gibbs free energy can be defined in several interconvertible ways, but a useful one in the context of biology is the enthalpy (internal energy) of a system minus the entropy of the system scaled by the temperature. The difference in free energy when a process takes place is often reported in terms of the change (Δ) of enthalpy (internal energy) denoted H, minus the temperature scaled change (Δ) in entropy, denoted S. See the equation below.
ΔG=ΔH−TΔS
The Gibbs energy is often interpreted as the amount of energy available to do useful work. With a bit of hand waving, we can interpret this by invoking the idea presented in the section on entropy, which states the dispersion of energy (required by the Second Law) associated with a positive change in entropy somehow renders some of the energy that is transferred less useful to do work. One can say that this is reflected in part in the T∆S term of the Gibbs equation.
To provide a basis for fair comparisons of changes in Gibbs free energy amongst different biological transformations or reactions, the free energy change of a reaction is measured under a set of common standard experimental conditions. The resulting standard free energy change of a chemical reaction is expressed as an amount of energy per mole of the reaction product (either in kilojoules or kilocalories, kJ/mol or kcal/mol; 1 kJ = 0.239 kcal), when measured at a standard pH, temperature, and pressure conditions. Standard pH, temperature, and pressure conditions are generally standardized at pH 7.0, 25 degrees Celsius, and 100 kilopascals (1 atm pressure), respectively. It is important to note that cellular conditions vary considerably from these standard conditions, and so actual ∆G inside a cell will differ considerably from those calculated under standard conditions.
Endergonic and exergonic reactions
For reactions with ∆G < 0, the products of the reaction have less free energy than the reactants. Since ∆G is the difference between the enthalpy and entropy changes in a reaction, a net negative ∆G can arise in different ways. The left panel of Figure 1 below shows a common graphical representation of an exergonic reaction. Free energy is plotted on the y-axis, and the x-axis in arbitrary units shows the progress of a reaction. This type of graph is called a reaction coordinate diagram. In the case of an exergonic reaction, the figure indicates two key things: (1) the difference between the free energy of the reactants and products is negative and (2) the progress of the reaction requires some input of free energy (shown as an energy hill). This graph does not tell us how the energy in the system was redistributed, only that the difference between enthalpy and entropy is negative. Reactions that have a negative ∆G are termed exergonic reactions. These reactions are said to occur spontaneously. Understanding which chemical reactions are spontaneous is extremely useful for biologists who are trying to understand whether a reaction is likely to "go" or not.
It is important to note that the term "spontaneous"—in the context of thermodynamics—does NOT imply anything about how fast the reaction proceeds. The change in free energy only describes the difference between beginning and end states, NOT how fast that transition takes place. This is somewhat contrary to the everyday use of the term, which usually carries the implicit understanding that something happens quickly. As an example, the oxidation/rusting of iron is a spontaneous reaction. However, an iron nail exposed to air does not rust instantly—it may take years.
A chemical reaction with a positive ∆G means that the products of the reaction have a higher free energy than the reactants (see the right panel of Figure 1). These chemical reactions are called endergonic reactions, and they are NOT spontaneous. An endergonic reaction will not take place on its own without the transfer of energy into the reaction or increase of entropy somewhere else.
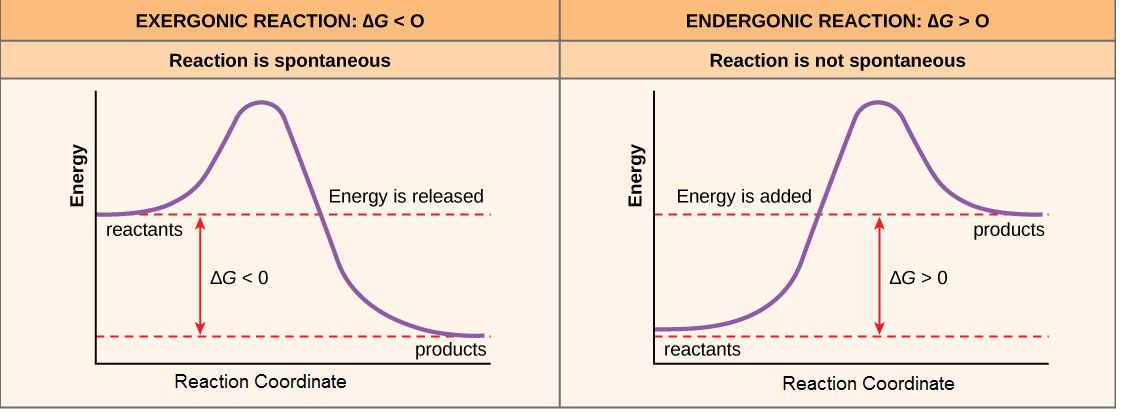
Figure 1. Exergonic and endergonic reactions result in changes in Gibbs free energy. In an exergonic reaction, the free energy of the products is lower than that of the reactants; meanwhile, in an endergonic reaction, the free energy of the products is higher than that of the reactants. Attribution: Marc T. Facciotti (own work)
The building of complex molecules, such as sugars, from simpler ones is an anabolic process and is endergonic. On the other hand, the catabolic process, such as the breaking down of sugar into simpler molecules, is generally exergonic. Like the example of rust above, while the breakdown of biomolecules is generally spontaneous, these reactions don’t necessarily occur instantaneously (quickly). Remember, the terms endergonic and exergonic only refer to the difference in free energy between the products and reactants; they don't tell you about the rate of the reaction (how fast it happens). The issue of rate will be discussed in later sections.
An important concept in the study of metabolism and energy is that of chemical equilibrium. Most chemical reactions are reversible. They can proceed in both directions, often transferring energy into their environment in one direction and transferring energy in from the environment in the other direction. The same is true for the chemical reactions involved in cell metabolism, such as the breaking down and building up of proteins into and from individual amino acids, respectively. Reactants within a closed system will undergo chemical reactions in both directions until a state of equilibrium is reached. This state of equilibrium is one of the lowest possible free energy states and is a state of maximal entropy. Equilibrium in a chemical reaction is the state in which both reactants and products are present in concentrations that have no further tendency to change with time. Usually, this state results when the forward reaction proceeds at the same rate as the reverse reaction. NOTE THIS LAST STATEMENT! Equilibrium means that the relative concentrations of reactants and products are not changing in time, BUT it does NOT mean that there is no interconversion between substrates and products—it just means that when the reactant(s) are converted to product(s) that product(s) are converted to reactant(s) at an equal rate (see Figure 2).
Either a rebalancing of substrate or product concentrations (by adding or removing substrate or product) or a positive change in free energy, typically by the transfer of energy from outside the reaction, is required to move a reaction out of a state of equilibrium. In a living cell, most chemical reactions do not reach a state of equilibrium—this would require that they reach their lowest free energy state. Energy is therefore required to keep biological reactions out of their equilibrium state. In this way, living organisms are in a constant, energy-requiring, uphill battle against equilibrium and entropy.

Figure 2. At equilibrium, do not think of a static, unchanging system. Instead, picture molecules moving in equal amounts from one area to another. Here, at equilibrium, molecules are still moving from left to right and right to left. The net movement however, is equal. There will still be about 15 molecules in each side of this flask once equilibrium is reached. Source: https://courses.candelalearning.com/...apter/entropy/
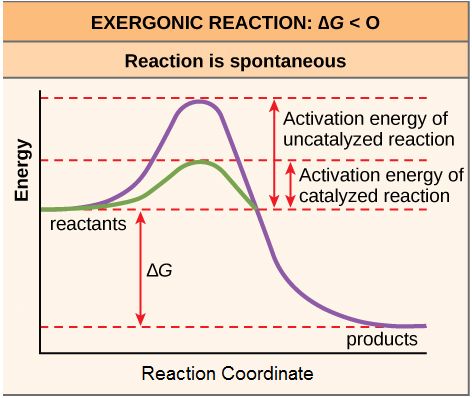
Let's start to think a little about the rate of a reaction. Even exergonic (spontaneous) reactions typically require a small increase in free energy before they can begin converting reactants to products. This initial positive change in free energy is called the activation energy (or free energy of activation) and is sometimes abbreviated EA.
Note: possible discussion
The oxidation of gasoline is highly exergonic. Despite this, why do cars not spontaneously explode in parking lots?
Why do nearly all chemical reactions—even those with a very large negative ∆G—first require some free energy increase to proceed? The reason lies in the steps that take place during a chemical reaction. Chemical reactions, almost by definition, require that some chemical bonds be broken and/or formed. For example, when a glucose molecule is broken down, the glycosidic bonds are broken, bonds within water are broken and new bonds are made between the "disassembled" water and the atoms that were involved in the glycosidic bond. While the overall reaction (the combination of energy cost of breaking bonds, energy gained by making bonds, and the change of entropy between reactants and products) may be negative, the breaking of the bonds requires some energy input which increases the free energy of the system. The state of the reaction at the maximum free energy of a reaction is often termed the transition state. This state is considered to be relatively unstable in which the reaction may either relax back to the reactant state or transition to the products. The height of the activation energy "barrier" has a direct relationship to the rate of a reaction. The higher/larger the barrier, the slower the reaction.
Note: possible discussion
Can you propose a physical analog (or model) that can help explain why the activation energy barrier is related to the rate of the reaction, whereas the free energy difference between substrate and product is not.
Figure 1. Activation energy is the energy required for a reaction to proceed, and it is lower if the reaction is catalyzed. The horizontal axis of this diagram describes the sequence of events in time.
Where does the free energy required required to overcome the activation energy barrier come from? The sources vary. One source is the energy transferred as heat from the surroundings. This transfer changes the kinetic energy of molecules in the system, increasing the frequency and force with which they collide and thus the frequency that they will react. In other cases, energy may be transferred from other reactions.
As noted, the activation energy of a particular reaction determines the rate at which it will proceed. The higher the activation energy, the slower the chemical reaction will be. The example of iron rusting illustrates an inherently slow reaction. The conversion of diamond into graphite is another spontaneous reaction that takes a LONG time. These reactions occur slowly over time because of high activation energy barriers. The burning (oxidation) of many fossil fuels, which is an exergonic process, will take place at a negligible rate unless their activation energy is overcome by sufficient heat from a spark. Once these fuels begin to burn, however, the chemical reactions release enough heat to help overcome the activation energy barrier for the combustion of the rest of the fuel. Like these reactions outside of cells, the activation energy for most cellular reactions is too high for heat energy to overcome at efficient rates. By the way, this is a very good thing as far as living cells are concerned. Important macromolecules, such as proteins, DNA, and RNA, store considerable energy, and their breakdown is exergonic. If cellular temperatures alone provided enough heat energy for these exergonic reactions to overcome their activation barriers, the essential components of a cell would disintegrate. Therefore, in order for important cellular reactions to occur at appreciable rates (number of reactions per unit time), their activation energies must be lowered (see figure 4). Something that helps lower the activation energy barrier is referred to as catalysis.
Note: possible discussion
If no activation energy were required to break down sucrose (table sugar), would you be able to store it in a sugar bowl?
Exercise 1: activation energy
Lowering the activation energy:
- Makes the reaction happen faster.
- Lowers the energy level of the transition state.
- Is accomplished by adding a catalyst to the reaction.
- Always causes more product to be produced.
- Only reduces the transition state energy level in one direction (from reactants to products).
- a, b, and c
- b and c
- All of the above are true.
Exercise 2
Which of the following comparisons or contrasts between endergonic and exergonic reactions are false?
- Endergonic reactions have a +∆G and exergonic reactions have a -∆G.
- Endergonic reactions consume energy and exergonic reactions release energy.
- Both endergonic and exergonic reactions require a small amount of energy to overcome an activation barrier.
- Endergonic reactions take place slowly and exergonic reactions take place quickly.
Exercise 3
Which of the following is the best way to judge the relative activation energies between two given chemical reactions?
- Compare the ∆G values between the two reactions.
- Compare their reaction rates.
- Compare their ideal environmental conditions.
- Compare the spontaneity between the two reactions.
Appendix: energy units
In the International System of Units (SI), the unit of work or energy is the Joule (J). For very small amounts of energy, the erg (erg) is sometimes used. An erg is one ten-millionth of a Joule:
1 Joule = 10,000,000 ergs (1)
Power is the rate at which energy is used. The unit of power is the Watt (W), named after James Watt, who perfected the steam engine:
1 Watt = 1 Joule/second (2)
Power is sometimes measured in horsepower (hp):
1 horsepower = 746 Watts (3)
Electrical energy is generally expressed in kilowatt-hours (kWh):
1 kilowatt-hour = 3,600,000 Joules (4)
It is important to realize that a kilowatt-hour is a unit of energy not power. For example, an iron rated at 2000 Watts would consume 2x3.6x106 J of energy in one hour.
Heat energy is often measured in calories. One calorie (cal) is defined as the heat required to raise the temperature of one gram of water from 14.5 to 15.5 ºC:
1 calorie = 4.189 Joules (5)
An old but still used unit of heat is the British Thermal Unit (BTU). It is defined as the heat energy required to raise the energy temperature of one pound of water from 63 to 64°F.
1 BTU = 1055 Joules (6)
| Physical quantity | Name | Symbol | SI unit |
|---|---|---|---|
| Force | Newton | N | kg*m/s2 |
| Energy | Joule | J | kg*m/s2 |
| Power | Watt | W | kg*m/s3 |