Using Claim, Evidence, Reasoning (CER)
- Page ID
- 26995
This page is a draft and is under active development.
\( \newcommand{\vecs}[1]{\overset { \scriptstyle \rightharpoonup} {\mathbf{#1}} } \)
\( \newcommand{\vecd}[1]{\overset{-\!-\!\rightharpoonup}{\vphantom{a}\smash {#1}}} \)
\( \newcommand{\dsum}{\displaystyle\sum\limits} \)
\( \newcommand{\dint}{\displaystyle\int\limits} \)
\( \newcommand{\dlim}{\displaystyle\lim\limits} \)
\( \newcommand{\id}{\mathrm{id}}\) \( \newcommand{\Span}{\mathrm{span}}\)
( \newcommand{\kernel}{\mathrm{null}\,}\) \( \newcommand{\range}{\mathrm{range}\,}\)
\( \newcommand{\RealPart}{\mathrm{Re}}\) \( \newcommand{\ImaginaryPart}{\mathrm{Im}}\)
\( \newcommand{\Argument}{\mathrm{Arg}}\) \( \newcommand{\norm}[1]{\| #1 \|}\)
\( \newcommand{\inner}[2]{\langle #1, #2 \rangle}\)
\( \newcommand{\Span}{\mathrm{span}}\)
\( \newcommand{\id}{\mathrm{id}}\)
\( \newcommand{\Span}{\mathrm{span}}\)
\( \newcommand{\kernel}{\mathrm{null}\,}\)
\( \newcommand{\range}{\mathrm{range}\,}\)
\( \newcommand{\RealPart}{\mathrm{Re}}\)
\( \newcommand{\ImaginaryPart}{\mathrm{Im}}\)
\( \newcommand{\Argument}{\mathrm{Arg}}\)
\( \newcommand{\norm}[1]{\| #1 \|}\)
\( \newcommand{\inner}[2]{\langle #1, #2 \rangle}\)
\( \newcommand{\Span}{\mathrm{span}}\) \( \newcommand{\AA}{\unicode[.8,0]{x212B}}\)
\( \newcommand{\vectorA}[1]{\vec{#1}} % arrow\)
\( \newcommand{\vectorAt}[1]{\vec{\text{#1}}} % arrow\)
\( \newcommand{\vectorB}[1]{\overset { \scriptstyle \rightharpoonup} {\mathbf{#1}} } \)
\( \newcommand{\vectorC}[1]{\textbf{#1}} \)
\( \newcommand{\vectorD}[1]{\overrightarrow{#1}} \)
\( \newcommand{\vectorDt}[1]{\overrightarrow{\text{#1}}} \)
\( \newcommand{\vectE}[1]{\overset{-\!-\!\rightharpoonup}{\vphantom{a}\smash{\mathbf {#1}}}} \)
\( \newcommand{\vecs}[1]{\overset { \scriptstyle \rightharpoonup} {\mathbf{#1}} } \)
\(\newcommand{\longvect}{\overrightarrow}\)
\( \newcommand{\vecd}[1]{\overset{-\!-\!\rightharpoonup}{\vphantom{a}\smash {#1}}} \)
\(\newcommand{\avec}{\mathbf a}\) \(\newcommand{\bvec}{\mathbf b}\) \(\newcommand{\cvec}{\mathbf c}\) \(\newcommand{\dvec}{\mathbf d}\) \(\newcommand{\dtil}{\widetilde{\mathbf d}}\) \(\newcommand{\evec}{\mathbf e}\) \(\newcommand{\fvec}{\mathbf f}\) \(\newcommand{\nvec}{\mathbf n}\) \(\newcommand{\pvec}{\mathbf p}\) \(\newcommand{\qvec}{\mathbf q}\) \(\newcommand{\svec}{\mathbf s}\) \(\newcommand{\tvec}{\mathbf t}\) \(\newcommand{\uvec}{\mathbf u}\) \(\newcommand{\vvec}{\mathbf v}\) \(\newcommand{\wvec}{\mathbf w}\) \(\newcommand{\xvec}{\mathbf x}\) \(\newcommand{\yvec}{\mathbf y}\) \(\newcommand{\zvec}{\mathbf z}\) \(\newcommand{\rvec}{\mathbf r}\) \(\newcommand{\mvec}{\mathbf m}\) \(\newcommand{\zerovec}{\mathbf 0}\) \(\newcommand{\onevec}{\mathbf 1}\) \(\newcommand{\real}{\mathbb R}\) \(\newcommand{\twovec}[2]{\left[\begin{array}{r}#1 \\ #2 \end{array}\right]}\) \(\newcommand{\ctwovec}[2]{\left[\begin{array}{c}#1 \\ #2 \end{array}\right]}\) \(\newcommand{\threevec}[3]{\left[\begin{array}{r}#1 \\ #2 \\ #3 \end{array}\right]}\) \(\newcommand{\cthreevec}[3]{\left[\begin{array}{c}#1 \\ #2 \\ #3 \end{array}\right]}\) \(\newcommand{\fourvec}[4]{\left[\begin{array}{r}#1 \\ #2 \\ #3 \\ #4 \end{array}\right]}\) \(\newcommand{\cfourvec}[4]{\left[\begin{array}{c}#1 \\ #2 \\ #3 \\ #4 \end{array}\right]}\) \(\newcommand{\fivevec}[5]{\left[\begin{array}{r}#1 \\ #2 \\ #3 \\ #4 \\ #5 \\ \end{array}\right]}\) \(\newcommand{\cfivevec}[5]{\left[\begin{array}{c}#1 \\ #2 \\ #3 \\ #4 \\ #5 \\ \end{array}\right]}\) \(\newcommand{\mattwo}[4]{\left[\begin{array}{rr}#1 \amp #2 \\ #3 \amp #4 \\ \end{array}\right]}\) \(\newcommand{\laspan}[1]{\text{Span}\{#1\}}\) \(\newcommand{\bcal}{\cal B}\) \(\newcommand{\ccal}{\cal C}\) \(\newcommand{\scal}{\cal S}\) \(\newcommand{\wcal}{\cal W}\) \(\newcommand{\ecal}{\cal E}\) \(\newcommand{\coords}[2]{\left\{#1\right\}_{#2}}\) \(\newcommand{\gray}[1]{\color{gray}{#1}}\) \(\newcommand{\lgray}[1]{\color{lightgray}{#1}}\) \(\newcommand{\rank}{\operatorname{rank}}\) \(\newcommand{\row}{\text{Row}}\) \(\newcommand{\col}{\text{Col}}\) \(\renewcommand{\row}{\text{Row}}\) \(\newcommand{\nul}{\text{Nul}}\) \(\newcommand{\var}{\text{Var}}\) \(\newcommand{\corr}{\text{corr}}\) \(\newcommand{\len}[1]{\left|#1\right|}\) \(\newcommand{\bbar}{\overline{\bvec}}\) \(\newcommand{\bhat}{\widehat{\bvec}}\) \(\newcommand{\bperp}{\bvec^\perp}\) \(\newcommand{\xhat}{\widehat{\xvec}}\) \(\newcommand{\vhat}{\widehat{\vvec}}\) \(\newcommand{\uhat}{\widehat{\uvec}}\) \(\newcommand{\what}{\widehat{\wvec}}\) \(\newcommand{\Sighat}{\widehat{\Sigma}}\) \(\newcommand{\lt}{<}\) \(\newcommand{\gt}{>}\) \(\newcommand{\amp}{&}\) \(\definecolor{fillinmathshade}{gray}{0.9}\)Cell Membrane Proteins: To understand the origins of pain, ask a flatworm
https://www.sciencenews.org/article/understand-origins-pain-ask-flatworm
Hydrogen peroxide, a molecule produced by cells under duress, may be a common danger signal, helping to alert animals to potential harm and send them scurrying. New details from planarian flatworms of how this process works may deepen scientists’ understanding of how people detect pain, and may ultimately point to better ways to curb it.
And by finding cellular similarities among planarians, fruit flies and people, the new study, published online October 16 in Nature Neuroscience, provides hints about how this threat-detecting system might have operated hundreds of millions of years ago.
The results center on a protein called TRPA1, a well-known pain detector in people. Embedded in the outside of cells, TRPA1 helps many different animals detect (and ultimately escape) harmful chemicals, physical injuries and extreme temperatures. In humans, mutations in the TRPA1 gene can cause syndromes marked by intense pain.
But scientists have puzzled over TRPA1’s seemingly inconsistent behavior in different animals. In Caenorhabditis elegans worms, for instance, the protein is activated by cold. But in other animals such as mosquitoes, TRPA1 is activated by heat. “The more people started looking at activation of TRPA1 in different species, the more the puzzle became complicated,” says study coauthor Marco Gallio of Northwestern University.
Gallio and colleagues studied TRPA1 behavior in the planarian Schmidtea mediterranea, a type of simple flatworm that is evolutionarily far from humans and other animals typically studied in labs. Normally, planarians avoid hot areas. In an experiment, flatworms with the TRPA1 protein kept away from the parts of a chamber that were heated to 32° Celsius. But planarians without the protein glided around the entire chamber, seemingly unbothered by the heat. Likewise, the flatworms without TRPA1 seemed content to stay in a chamber laced with the chemical that gives mustard and wasabi their kick. Those results suggest that TRPA1 is involved, somehow, in planarians’ responses to cellular danger.
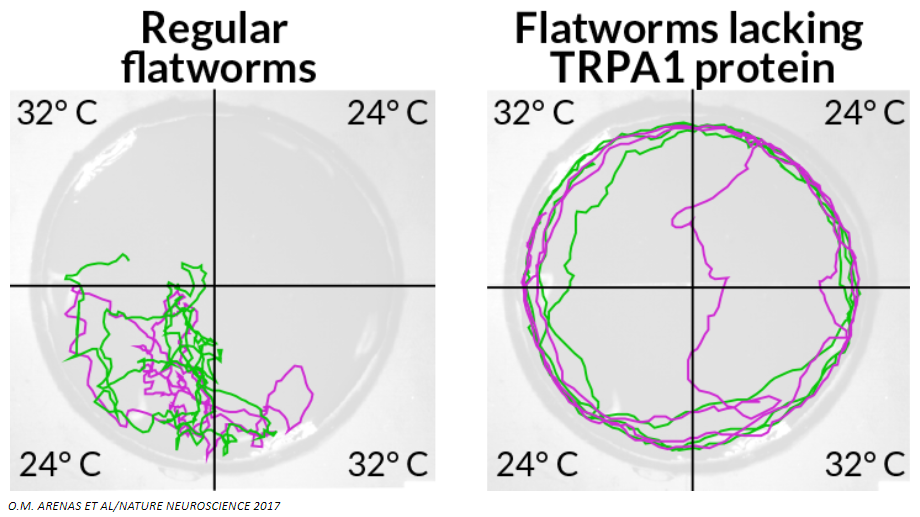
Hot spot
Two regular planarians (track marks of each shown in green and purple) avoid hot areas of a chamber (left), but two planarians lacking TRPA1 protein, which is linked to pain detection, don’t seem to mind the heat, gliding all around the chamber (right).
In further tests, the researchers found that as the planarians were heated to harmful temperatures, their bodies produced more reactive oxygen species, chemicals that include hydrogen peroxide. And experiments with cells in dishes revealed that hydrogen peroxide directly activates the planarians’ TRPA1 protein.
Hydrogen peroxide, or other reactive oxygen species, may serve as universal danger signals that TRPA1 detects, Gallio and his colleagues suspect . This general signal may explain a somewhat paradoxical property of TRPA1 that the researchers observed: Human TRPA1, which is activated by cold temperatures, allowed fruit flies to avoid hot areas, presumably by detecting hydrogen peroxide. The same was true for planarian TRPA1, which doesn’t directly respond to heat. That overlap in duty is possible because the protein isn’t responding to specific temperatures, but instead, to hydrogen peroxide, the researchers suspect.
The use of a chemical intermediate “really solves the mystery” of TRPA1’s seemingly variable behavior, Gallio says. With hydrogen peroxide as a chemical middleman, TRPA1 can detect a variety of temperatures and other danger signals, allowing TRPA1 to detect threats that are particular to different animals.
Studying TRPA1 in planarians offers insights about the evolutionary history of human pain sensing. Because the planarian lineage split from those of flies, mice and humans so long ago, it’s possible that an ancient common ancestor possessed a hydrogen peroxide‒sensing TRPA1 protein similar to that found in planarians. “Getting that perspective is terrific because you can get a very high-altitude perspective view of how evolution might be working,” Garrity says.
Citations
O.M. Arenas et al. Activation of planarian TRPA1 by reactive oxygen species reveals a conserved mechanism for animal nociception. Nature Neuroscience. Published online October 16, 2017. doi: 10.1038/s41593-017-0005-0.
CER Chart