Spring_2024_Bis2A_Igo_Lecture_04
- Page ID
- 132977
\( \newcommand{\vecs}[1]{\overset { \scriptstyle \rightharpoonup} {\mathbf{#1}} } \)
\( \newcommand{\vecd}[1]{\overset{-\!-\!\rightharpoonup}{\vphantom{a}\smash {#1}}} \)
\( \newcommand{\id}{\mathrm{id}}\) \( \newcommand{\Span}{\mathrm{span}}\)
( \newcommand{\kernel}{\mathrm{null}\,}\) \( \newcommand{\range}{\mathrm{range}\,}\)
\( \newcommand{\RealPart}{\mathrm{Re}}\) \( \newcommand{\ImaginaryPart}{\mathrm{Im}}\)
\( \newcommand{\Argument}{\mathrm{Arg}}\) \( \newcommand{\norm}[1]{\| #1 \|}\)
\( \newcommand{\inner}[2]{\langle #1, #2 \rangle}\)
\( \newcommand{\Span}{\mathrm{span}}\)
\( \newcommand{\id}{\mathrm{id}}\)
\( \newcommand{\Span}{\mathrm{span}}\)
\( \newcommand{\kernel}{\mathrm{null}\,}\)
\( \newcommand{\range}{\mathrm{range}\,}\)
\( \newcommand{\RealPart}{\mathrm{Re}}\)
\( \newcommand{\ImaginaryPart}{\mathrm{Im}}\)
\( \newcommand{\Argument}{\mathrm{Arg}}\)
\( \newcommand{\norm}[1]{\| #1 \|}\)
\( \newcommand{\inner}[2]{\langle #1, #2 \rangle}\)
\( \newcommand{\Span}{\mathrm{span}}\) \( \newcommand{\AA}{\unicode[.8,0]{x212B}}\)
\( \newcommand{\vectorA}[1]{\vec{#1}} % arrow\)
\( \newcommand{\vectorAt}[1]{\vec{\text{#1}}} % arrow\)
\( \newcommand{\vectorB}[1]{\overset { \scriptstyle \rightharpoonup} {\mathbf{#1}} } \)
\( \newcommand{\vectorC}[1]{\textbf{#1}} \)
\( \newcommand{\vectorD}[1]{\overrightarrow{#1}} \)
\( \newcommand{\vectorDt}[1]{\overrightarrow{\text{#1}}} \)
\( \newcommand{\vectE}[1]{\overset{-\!-\!\rightharpoonup}{\vphantom{a}\smash{\mathbf {#1}}}} \)
\( \newcommand{\vecs}[1]{\overset { \scriptstyle \rightharpoonup} {\mathbf{#1}} } \)
\( \newcommand{\vecd}[1]{\overset{-\!-\!\rightharpoonup}{\vphantom{a}\smash {#1}}} \)
Learning Objectives Associated with Spring_2024_Bis2A_Igo_Lecture_04MS.6 Be able to classify common biomolecules as a lipid, protein, carbohydrate, or nucleic acid. MS.22 Create a "parts-level" abstract sketch of a generic glycerophospholipid that includes: (a) lipid tails, (b) glycerol, (c) phosphate, and (d) a "decoration" (e.g. ethanolamine). This need not be an atom-by-atom structure but rather a cartoon depicting the order and types of bonds between structural elements. MS.20 Draw how water molecules might interact with hydrophobic parts of molecules. MS.21 Describe how the chemical structures and properties of lipids contribute to their functions (e.g. formation of membrane structures, contribution to membrane fluidity, etc.). MS.23 Compare and contrast the influence of different phospholipid chain lengths and degree of saturation in membrane lipids on membrane fluidity. MS.24 Create simple illustrations of carbohydrates that include the major functional groups, the formation of glycosidic bonds, and the potential interactions of carbohydrates with water molecules or other biomolecules. MS.25 Diagram the pentose and hexose sugars, be able to number their carbon atoms, and identify the key functional groups on each molecule. MS.26 Identify a nucleotide from its molecular structure and be able to decompose the molecule into three main functional units: nitrogenous base, ribose, and phosphates. |
Lipids
Lipids are a diverse group of hydrophobic compounds that include molecules like fats, oils, waxes, phospholipids, and steroids. Most lipids are at their core hydrocarbons, molecules that include many nonpolar carbon-carbon or carbon-hydrogen bonds. The abundance of nonpolar functional groups
Fats and oils
A common fat molecule or triglyceride. These types of molecules are hydrophobic and, while they have many functions, are probably best known for their roles in body fat and plant oils. A triglyceride molecule derived from two types of molecular components—a polar "head" group and a nonpolar "tail" group. The "head" group of a triglyceride
Figure 2. Stearic acid is a common saturated fatty
Natural fats like butter, canola oil, etc.,
- The number of carbons in the hydrocarbon chains;
- The number of desaturations, or double bonds, in the hydrocarbon chains.
The first factor influences how these molecules interact with each other and with water, while the second factor dramatically influences their shape. Introducing a double bond causes a "kink" in the otherwise relatively "straight" hydrocarbon, depicted in a slightly exaggerated was in Figure 3.
Based on what you can understand from this brief description, propose a rationale—in your own words—to explain why butter is solid at room temperature while vegetable oil is liquid.
Here is an additional piece of information that could help you answer question: butter has a greater percentage of longer and saturated hydrocarbons in its triglycerides than does vegetable oil.
Figure 3. The straight saturated fatty acid versus the "bent"/"kinked" unsaturated fatty acid. Attribution:
Real-Life connection:
Ever thought about how lipids are important to vision? Read more here.
Sterols
Steroids are lipids with a fused ring structure. Although they do not resemble the other lipids discussed here,
| Cholesterol | Cortisol |
Phospholipids
Phospholipids are major constituents of the cell membrane, the outermost layer of cells. Like fats,
Note
Note in Figure 5 that the phosphate group has an R group linked to one of the oxygen atoms. R is a variable commonly used in these types of diagrams to show some other atom or molecule bound at that position. That part of the molecule can be different in different phospholipids—and will impart some different chemistry to the whole molecule. At the moment, however, you are
| 1,2-Dioleoyl-sn-glycero-3-phospho-L-serine |
In the presence of water, some phospholipids will spontaneously arrange themselves into a micelle (Figure 6). The lipids arrange such that their polar groups are on the outside of the micelle, and the nonpolar tails are on the inside. Under other conditions, a lipid bilayer can also form. This structure, only a few nanometers thick,
We discuss the phospholipid membrane in a later module. It is important to remember the chemical properties associated with the functional groups in the phospholipid to understand the function of the cell membrane.
Carbohydrates
Carbohydrates are one of the four main classes of macromolecules that make up all cells and are an essential part of our diet; grains, fruits, and vegetables are all natural sources. While we may be most familiar with the role carbohydrates play in nutrition, they also have a variety of other essential functions in humans, animals, plants, and bacteria. In this section, we will discuss and review basic concepts of carbohydrate structure and nomenclature, and a variety of functions they play in cells.
Molecular structures
In their simplest form, carbohydrates can be represented by the stoichiometric formula (CH2O
Nomenclature
One issue with carbohydrate chemistry is the nomenclature. Here are a few quick and simple rules:
- Simple carbohydrates, such as glucose, lactose, or dextrose, end with an "-ose."
Simple carbohydrates can be classified based on the number of carbon atoms in the molecule, as with triose (three carbons),pentose (five carbons), or hexose (six carbons).Simple carbohydrates can be classified based on the functional group found in the molecule,i.e ketose (contains a ketone) or aldose (contains an aldehyde).- Polysaccharides
are often organized by the number of sugar molecules in the chain, such as in a monosaccharide, disaccharide, or trisaccharide.
For a short video on carbohydrate classification, see the 10-minute Khan Academy video by clicking here.
Monosaccharides
Monosaccharides ("mono-" = one; "

Figure 1.
Glucose versus galactose
Galactose (part of
Fructose versus both glucose and galactose
In glucose and galactose, the carbonyl group is on the C1 carbon, forming an aldehyde group. In fructose, the carbonyl group is on the C2 carbon, forming a ketone group. The former sugars
Figure 2. Glucose, galactose, and fructose are all hexoses. They are structural isomers, meaning they have the same chemical formula (C6H12O6) but a different arrangement of atoms.
Linear versus ring form of the monosaccharides
Monosaccharides can exist as a linear chain or as ring-shaped molecules. In aqueous solutions, monosaccharides are usually found in ring form (Figure 3). Glucose in a ring form can have two different arrangements of the hydroxyl group (OH) around the anomeric carbon (C1 that becomes asymmetric in the process of ring formation). If the hydroxyl group is below C1 in the sugar,
Figure 3. Five- and six-carbon monosaccharides exist in equilibrium between linear and ring form. When the ring forms, the side chain it closes on
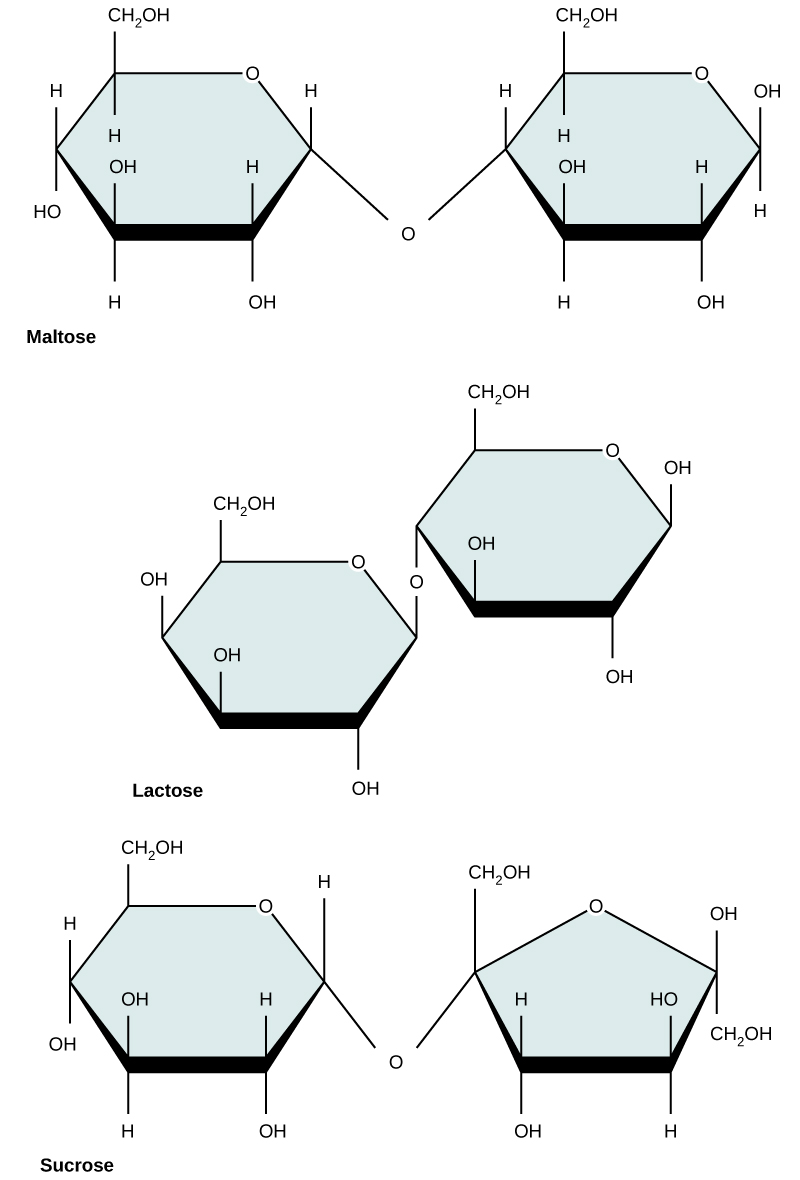
Disaccharides
Disaccharides ("di-" = two) form when two monosaccharides undergo a dehydration reaction (also known as a condensation reaction or dehydration synthesis). During this process, the hydroxyl group of one monosaccharide combines with the hydrogen of another monosaccharide, releasing a molecule of water and forming a covalent bond. A covalent bond formed between a carbohydrate molecule and another molecule (in this case, between two monosaccharides) is known as a glycosidic bond. Glycosidic bonds (also called glycosidic linkages) can be of the alpha or the beta type.
Figure 4. Sucrose
Common disaccharides include lactose, maltose, and sucrose (Figure 5). Lactose is a disaccharide
Figure 5. Common disaccharides include maltose (grain sugar), lactose (milk sugar), and sucrose (table sugar).
| Sucrose | Lactose | Maltose |
Polysaccharides
A long chain of monosaccharides linked by glycosidic bonds is known as a polysaccharide ("poly-" = many). The chain may
Starch is the stored form of
Figure 6. Amylose and amylopectin are two different
Glycogen
Glycogen is a common stored form of glucose in humans and other vertebrates. Glycogen is the animal equivalent of starch and is a highly branched molecule usually stored in liver and muscle cells. Whenever blood glucose levels decrease,
| Glycogen |
Cellulose
Cellulose is the most abundant natural biopolymer.
Figure 7. In cellulose,
As shown in the figure above, every other glucose monomer in cellulose
Interactions with carbohydrates
We have just discussed the various types and structures of carbohydrates found in biology. The next thing to address is how these compounds interact with other compounds. The answer to that is that it depends on the final structure of the carbohydrate. Because carbohydrates have many hydroxyl groups associated with the molecule, they are therefore excellent H-bond donors and acceptors. Monosaccharides can quickly and easily form H-bonds with water and are readily soluble. All of those H-bonds also make them quite "sticky". This is also true for many disaccharides and many short-chain polymers. Longer polymers may not be readily soluble.
Finally, the ability to form a variety of H-bonds allows polymers of carbohydrates or polysaccharides to form strong intramolecular and
Possible NB Discussion  Point
Point
Lipids and carbohydrates are not just classes of macromolecules that we discuss in BIS 2A but are also two of the essential macronutrients that we can obtain from eating various foods. Some popularized diet programs (e.g., Atkins, ketogenic) suggest limiting carbohydrates and/or fats. As you learn more about biomolecules and their roles in living systems, are you refining your perspective on foods and diets? What have you learned so far? Do you think there is anything missing in your understanding? Are you able to better understand and evaluate certain diets, such as the aforementioned?
Nucleic acids
There are two types of nucleic acids in biology: DNA and RNA. DNA carries the heritable genetic information of the cell and
Nucleotide structure
The two main types of nucleic acids are deoxyribonucleic acid (DNA) and ribonucleic acid (RNA).
The nitrogenous base
The nitrogenous bases of nucleotides are organic molecules and are so named because they contain carbon and nitrogen. They are bases because they contain an amino group that has the potential of binding an extra
Possible NB Discussion  Point
Point
Take a moment to review the five nitrogenous bases in Figure 1 above. Identify functional groups as described in class. For each functional group identified, describe what type
The pentose sugar
The pentose sugar contains five carbon atoms. Each carbon atom of the sugar molecule
The pentose sugar in DNA
The phosphate group
There can be anywhere between one and three phosphate groups bound to the 5' carbon of the sugar. When one phosphate
Note: "high-energy" bonds
The term "high-energy bond"

| Adenosine triphosphate |
Double helix structure of DNA
DNA has a double helix structure (shown below) created by two strands of covalently linked nucleotide subunits.
In a double helix, certain combinations of base pairing are chemically more favored than others based on the types and locations of functional groups on the nitrogenous bases of each nucleotide. In
Adenine (A) is chemically complementary with thymidine (T) (
and
Guanine (G) is chemically complementary with cytosine (C) (G pairs with C).
We often refer to this pattern as "base complementarity" and say that the antiparallel strands are complementary to each other. For example, if the sequence of one strand is of DNA is 5'-AATTGGCC-3', the complementary strand would have the sequence 5'-GGCCAATT-3'.
We sometimes
5' - GGCCAATTCCATACTAGGT - 3'
3' - CCGGTTAAGGTATGATCCA - 5'
Note that each strand has its 5' and 3' ends labeled and that if one were to walk along each strand starting from the 5' end to the 3' end that the direction of travel would be opposite the other for each strand; the strands are antiparallel. We commonly say things like "running 5-prime to 3-prime" or "synthesized 5-prime to 3-prime" to refer to the direction we are reading a sequence or the direction of synthesis. Start getting yourself accustomed to this nomenclature.


Functions and roles of nucleotides and nucleic acids to look out for in General Biology
Besides their structural roles in DNA and RNA, nucleotides such as ATP and GTP also serve as mobile energy carriers for the cell. It surprises some students when they learn to appreciate that the ATP and GTP molecules we discuss in bioenergetics are the same as those involved in the formation of nucleic acids. We will cover this in more detail when we discuss DNA and RNA synthesis reactions. Nucleotides also play important roles as
Nucleic acids, RNA in particular, play a variety of roles in in
PRACTICE POST-GUIDE
General Practice
3. Why: The lecture slides are usually full of examples and sample questions. These were not created by accident and are directly linked to working on the learning objectives. We don’t want to miss the opportunity to go back and study the examples and use them to better understand expectations.
How to practice: Revisit the lecture slide questions and make sure you can answer the questions and/or do the exercises. If you can’t, work with your study group and/or go visit the TA or instructor at office hours for clarification.
4. Why: You are responsible for the learning objectives associated with the sections in the text and lecture on lipids, carbohydrates, and nucleic acids. One of the key objectives is to “Be able to classify common biomolecules as a lipid, protein, carbohydrate, or nucleic acid.” You should be able to recognize carbohydrates, nucleic acids, proteins, and lipids when they appear and be able to distinguish their basic chemical structures from one another, paying attention to functional group chemistry as well. I did some examples in class and you’ll do more in discussion.
How to practice: Go to Google or your favorite search engine and search independently for images.
- For carbohydrates try the following terms: “carbohydrate molecule examples”, “ploysaccharide molecule examples” “glycogen molecule examples”.
- For lipids try the following terms: “lipid molecule examples”, “polyketide molecule examples”, “sterol molecule examples”, "sphingolipids molecule example” and “glycerophospholipids molecule example”
- For nucleic acids try typing: “nucleic acid molecule examples” and “nucleotide molecule examples”
- For proteins type: “protein structure examples”.
Peruse the images. Look for similar patterns within groups and differences between groups. Most of all, get used to seeing and classifying how these molecules are represented.
This exercise is good practice for learning objective: “MS.6 Be able to classify common biomolecules as a lipid, protein, carbohydrate, or nucleic acid.”
5. Why: The diversity of lipid structures is huge - perhaps you got the feeling from exercise #4. Lipids, even similar looking ones can also play roles in quite different biological functions. Reinforcing the ability to recognize some key lipids and appreciating how structural similarities and differences can lead to a molecule having different functional roles is key.
How to practice: Go to Google Images and search for pictures of the following molecules: cholesterol (key molecule in mammalian membranes), testosterone (male sex hormone), aldosterone (hormone that regulates kidney function), and cortisol (hormone involved in glucose metabolism and several other functions). First note the different process and functions these molecules are associated with. Draw each them out next to one another. What are the key similarities? What are the differences?
This exercise is also good practice for learning objective: “MS.6 Be able to classify common biomolecules as a lipid, protein, carbohydrate, or nucleic acid.” The lipids above often fool people in classification exercises. Learn to recognize and classify structures like these too.
6. Why: You need to develop an intuitive feel for recognizing carbohydrate structures, their major “parts” and archetypical chemistry.
How to practice: The model to the right depicts the carbohydrate sucrose (table sugar). This molecule is a disaccharide (two sugars).
- Redraw this molecule in your sketchbook.
- Identify the glycosidic bond between the two components.
- Highlight the key chemical feature that make this molecule a carbohydrate.
- Draw this molecule’s potential interactions with water molecules.

This is practice for learning objective: “MS.24 Create simple illustrations of carbohydrates that include the major functional groups, the formation of glycosidic bonds, and the potential interactions of carbohydrates with water molecules or other biomolecules.”
7. Why: Glycerophospholipids are a core component of nearly all biological membranes. Elements of their structures and how they interact with water play critical roles in their functions. It is, therefore, important to identify and understand the elements of their structures. There’s nothing like actively creating models yourself to boost understanding and knowledge retention.
How to practice: The molecule below is classified as a glycerophospholipid. Redraw this molecule in your sketchbook and then circle/label all parts including: hydrophobic tails, the glycerol backbone, the phosphate group, and the R-group.
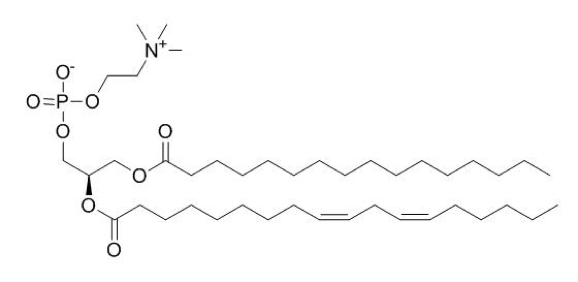
Circle the polar head group. Draw some water molecules around the glycerophospholipid. Then redraw a “cartoon” version that has less molecular detail but includes the key parts listed above. An example of such a cartoon is in the reading.
Looking at the molecule and “seeing” the parts is not nearly as good for your learning as making yourself redraw it is. This more active form of learning will engrain the image and ideas in your mind much more effectively.
This is practice for the learning objectives: “MS.22 Create a "parts-level" abstract sketch of a generic glycerophospholipid that includes: (a) lipid tails, (b) glycerol, (c) phosphate, and (d) a "decoration" (e.g. ethanolamine). This need not be an atom-by-atom structure but rather a cartoon depicting the order and types of bonds between structural elements.”; “MS.20 Draw how water molecules might interact with hydrophobic parts of molecules."
PRACTICE EXAM QUESTIONS
Question Q4.1
Q4.1 The molecule of cortisol below is an important hormone. In the blood, cortisol is not found free (on its own), but is mostly found circulating in solution bound to proteins called transcortin or albumin. Select the answer choice that best completes the explanation for this observation.

Proposed explanation: Cortisol is a (1)____________ molecule and is (2) ____________ soluble in water. The binding of cortisol to transcortin is most likely mediated by hydrogen bonds involving (3) ____________ and hydrophobic interactions involving (4)__________.
- (1) carbohydrate; (2) poorly; (3) glutamine; (4) tryptophan
- (1) lipid; (2) poorly; (3) glutamine; (4) tryptophan
- (1) carbohydrate; (2) highly; (3) tryptophan; (4) glutamine
- (1) lipid; (2) highly; (3) tryptophan; (4) glutamine
- (1) lipid; (2) poorly; (3) tryptophan; (4) glutamine