S2024_Bis2a_Namekawa_Regulation_of_Gene_Expression_II
- Page ID
- 132383
\( \newcommand{\vecs}[1]{\overset { \scriptstyle \rightharpoonup} {\mathbf{#1}} } \)
\( \newcommand{\vecd}[1]{\overset{-\!-\!\rightharpoonup}{\vphantom{a}\smash {#1}}} \)
\( \newcommand{\id}{\mathrm{id}}\) \( \newcommand{\Span}{\mathrm{span}}\)
( \newcommand{\kernel}{\mathrm{null}\,}\) \( \newcommand{\range}{\mathrm{range}\,}\)
\( \newcommand{\RealPart}{\mathrm{Re}}\) \( \newcommand{\ImaginaryPart}{\mathrm{Im}}\)
\( \newcommand{\Argument}{\mathrm{Arg}}\) \( \newcommand{\norm}[1]{\| #1 \|}\)
\( \newcommand{\inner}[2]{\langle #1, #2 \rangle}\)
\( \newcommand{\Span}{\mathrm{span}}\)
\( \newcommand{\id}{\mathrm{id}}\)
\( \newcommand{\Span}{\mathrm{span}}\)
\( \newcommand{\kernel}{\mathrm{null}\,}\)
\( \newcommand{\range}{\mathrm{range}\,}\)
\( \newcommand{\RealPart}{\mathrm{Re}}\)
\( \newcommand{\ImaginaryPart}{\mathrm{Im}}\)
\( \newcommand{\Argument}{\mathrm{Arg}}\)
\( \newcommand{\norm}[1]{\| #1 \|}\)
\( \newcommand{\inner}[2]{\langle #1, #2 \rangle}\)
\( \newcommand{\Span}{\mathrm{span}}\) \( \newcommand{\AA}{\unicode[.8,0]{x212B}}\)
\( \newcommand{\vectorA}[1]{\vec{#1}} % arrow\)
\( \newcommand{\vectorAt}[1]{\vec{\text{#1}}} % arrow\)
\( \newcommand{\vectorB}[1]{\overset { \scriptstyle \rightharpoonup} {\mathbf{#1}} } \)
\( \newcommand{\vectorC}[1]{\textbf{#1}} \)
\( \newcommand{\vectorD}[1]{\overrightarrow{#1}} \)
\( \newcommand{\vectorDt}[1]{\overrightarrow{\text{#1}}} \)
\( \newcommand{\vectE}[1]{\overset{-\!-\!\rightharpoonup}{\vphantom{a}\smash{\mathbf {#1}}}} \)
\( \newcommand{\vecs}[1]{\overset { \scriptstyle \rightharpoonup} {\mathbf{#1}} } \)
\( \newcommand{\vecd}[1]{\overset{-\!-\!\rightharpoonup}{\vphantom{a}\smash {#1}}} \)
Eukaryotic Gene Regulation
Regulation overview
As previously noted, regulation is all about decision making. Gene regulation, as a general topic, relates to deciding about the functional expression of genetic material. Whether the final product is an RNA species or a protein, the production of the final expressed product requires processes that take multiple steps. We have spent some time discussing some of these steps (i.e. transcription and translation) and some mechanisms that nature uses for sensing cellular and environmental information to regulate the initiation of transcription.
When we discussed the concept of strong and weak promoters, we introduced the idea that regulating the amount (number of molecules) of a transcript produced from a promoter in some unit of time might also be important for function. This should not be entirely surprising. For a protein-coding gene, the more transcript produced, the greater potential there is to make more protein. This might be important when making a lot of a particular enzyme is key for survival. In other cases, the cell needs only a little of a specific protein and making too much would be a waste of cellular resources. Here, the cell may prefer low levels of transcription. Promoters of differing strengths can accommodate these varying needs. Regarding transcript number, we also briefly mentioned that synthesis is not the only way to regulate abundance. Degradation processes are also important to consider.
In this section, we add to these themes by focusing on eukaryotic regulatory processes. Specifically, we examine - and sometimes re-examine - the multiple steps required to express genetic material in eukaryotic organisms in the context of regulation. We want you not only to think about the processes but also to recognize that each step in the process of expression is also an opportunity to fine tune not only the abundance of a transcript or protein but also its functional state, form (or variant), and/or stability. Each of these additional factors may also be vitally important to consider for influencing the abundance of conditionally specific functional variants.
Structural differences between bacterial and eukaryotic cells influencing gene regulation
The defining hallmark of the eukaryotic cell is the nucleus, a double membrane that encloses the cell's hereditary material. In order to efficiently fits the organism's DNA into the confined space of the nucleus, the DNA is first packaged and organized by protein into a structure called chromatin. This packaging of the nuclear material reduces access to specific parts of the chromatin. Some elements of the DNA are so tightly packed that the transcriptional machinery cannot access regulatory sites like promoters. This means that one of the first sites of transcriptional regulation in eukaryotes must be the control access to the DNA itself. Chromatin proteins can be subject to enzymatic modification that can influence whether they bind tightly (limited transcriptional access) or more loosely (greater transcriptional access) to a segment of DNA . This process of modification - whichever direction is considered first - is reversible. Therefore, DNA can be dynamically sequestered and made available when the "time is right".
The regulation of gene expression in eukaryotes also involves some of the same additional fundamental mechanisms discussed in the module on bacterial regulation (i.e. the use of strong or weak promoters, transcription factors, terminators etc.) but the actual number of proteins involved is typically much greater in eukaryotes than bacteria or archaea.
The post-transcriptional enzymatic processing of RNA that occurs in the nucleus and the export of the mature mRNA to the cytosol are two additional difference between bacterial and eukaryotic gene regulation. We will consider this level of regulation in more detail below.
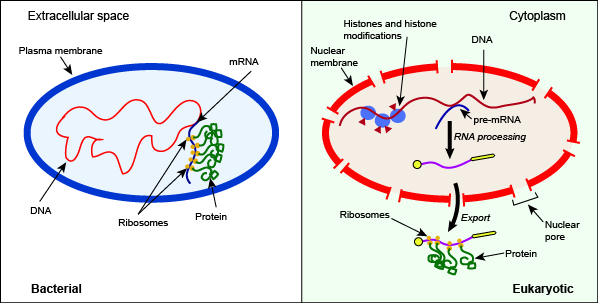
Depiction of some key differences between the processes of bacterial and eukaryotic gene expression. Note in this case the presence of histone and histone modifiers, the splicing of pre-mRNA, and the export of the mature RNA from the nucleus as key differentiators between the bacterial and eukaryotic systems. Attribution: Marc T. Facciotti (own work)
Chromatin structure and gene regulation
The DNA in eukaryotic cells is precisely wound, folded, and compacted into chromosomes so it will fit into the nucleus. The nucleus also organizes the DNA so key proteins can easily access specific segments of the chromosomes as needed. Areas of the chromosomes that are more tightly compacted will be harder for proteins to bind and therefore lead to reduced gene expression of genes encoded in those areas. Loosely compacted regions of the genome will be easier for proteins to access, thus increasing the likelihood that a gene will be transcribed. Discussed here are how cells regulate the density of DNA compaction.
The first level of organization, or packing, is the winding of DNA strands around histone proteins. Histones package and order DNA into structural units called nucleosomes, which can control the access of proteins to specific DNA regions. Under the electron microscope, this winding of DNA around histone proteins to form nucleosomes looks like small beads on a string. These beads (nucleosome complexes) can move along the string (DNA) to alter which areas of the DNA are accessible to transcriptional machinery. While nucleosomes can move to open the chromosome structure to expose a segment of DNA, they do so in a very controlled manner.
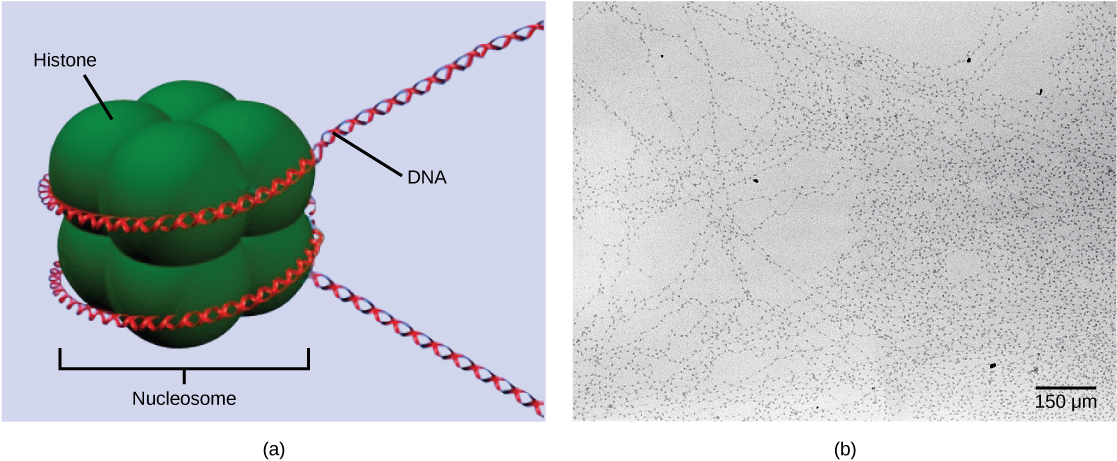
DNA folds around histone proteins to create (a) nucleosome complexes. These nucleosomes control the access of proteins to the underlying DNA. When viewed through an electron microscope (b), the nucleosomes look like beads on a string. (credit “micrograph”: modification of work by Chris Woodcock
Possible NB Discussion  Point
Point
In the later maturation phase of sperm cells, histones (containing high numbers of lysine amino acids) are replaced by protamines, which are small, nuclear proteins that are very rich in arginine amino acids. This process is said to be essential for sperm head condensation and DNA stabilization. Based on this information, what comparisons can you draw between protamines and histones? Why is it significant that there are high numbers of lysine and arginine in histones and protamines? For what reasons do you think protamines replace histones in sperm but not other cells?
Eukaryotic gene structure
Many eukaryotic genes, particularly those encoding protein products, are encoded on the genome discontinuously. The coding region is broken into pieces by intervening non-coding gene elements. We term the coding regions exons while the intervening non-coding elements are termed introns. The figure below depicts a generic eukaryotic gene.
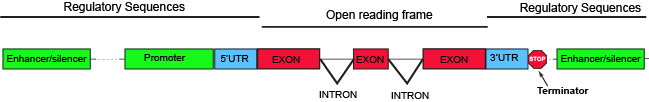
The parts of a typical discontinuous eukaryotic gene. Attribution: Marc T. Facciotti (own work)
Parts of a generic eukaryotic gene include familiar elements like a promoter and terminator. Between those two elements, the region encoding all the elements of the gene that have the potential to be translated (they have no stop codons), like in bacterial systems, is called the open reading frame (ORF). Enhancer and/or silencer elements are regions of the DNA that recruit regulatory proteins. These can be relatively close to the promoter, like in bacterial systems, or thousands of nucleotides away. Also present in many bacterial transcripts, 5' and 3' untranslated regions (UTRs) also exist. These regions of the gene encode segments of the transcript, which, as their names imply, are not translated and sit 5' and 3', respectively, to the ORF. The UTRs typically encode some regulatory elements critical for regulating transcription or steps of gene expression that occur post-transcriptionally.
Promoters
Genes are organized to make the control of gene expression easier. The promoter region is immediately upstream of the coding sequence. This region can be short (only a few nucleotides in length) or quite long (hundreds of nucleotides long). The longer the promoter, the more available space for proteins to bind. This also adds more control to the transcription process. The length of the promoter is gene-specific and can differ dramatically between genes. Consequently, the level of control of gene expression can also differ quite dramatically between genes. The purpose of the promoter is to bind transcription factors that control the initiation of transcription.
Within the promoter region, just upstream of the transcriptional start site, resides the TATA box. This box is simply a repeat of thymine and adenine dinucleotides (literally, TATA repeats). RNA polymerase binds to the transcription initiation complex, allowing transcription to occur. To initiate transcription, a transcription factor (TFIID) is the first to bind to the TATA box. Binding of TFIID recruits other transcription factors to the TATA box. Once this transcription initiation complex is assembled, RNA polymerase can bind to its upstream sequence. When bound along with the transcription factors, RNA polymerase is phosphorylated. This releases part of the protein from the DNA to activate the transcription initiation complex and places RNA polymerase in the correct orientation to begin transcription; DNA-bending protein brings the enhancer, which can be quite a distance from the gene, in contact with transcription factors and mediator proteins.
In addition to the general transcription factors, other transcription factors can bind to the promoter to regulate gene transcription. These transcription factors bind to the promoters of a specific set of genes. They are not general transcription factors that bind to every promoter complex, but are recruited to a specific sequence on the promoter of a specific gene. There are hundreds of transcription factors in a cell that each bind specifically to a particular DNA sequence motif. When transcription factors bind to the promoter just upstream of the encoded gene, they are referred to as cis-acting elements because they are on the same chromosome, just next to the gene. The region that a particular transcription factor binds to is called the transcription factor binding site. Transcription factors respond to environmental stimuli that cause the proteins to find their binding sites and initiate transcription of the gene that is needed.
Enhancers
In some eukaryotic genes, there are regions that help increase or enhance transcription. These regions, called enhancers, are not necessarily close to the genes they enhance. They can be located upstream of a gene, within the coding region of the gene, downstream of a gene, or may be thousands of nucleotides away.
Enhancer regions are binding sequences, or sites, for transcription factors. When a DNA-bending protein binds to an enhancer, the shape of the DNA changes. This shape change allows the interaction between the activators bound to the enhancers and the transcription factors bound to the promoter region and the RNA polymerase to occur. Whereas DNA is generally depicted as a straight line in two dimensions, it is actually a three-dimensional object. Therefore, a nucleotide sequence thousands of nucleotides away can fold over and interact with a specific promoter.
 Figure 16.4𝐵.1
Figure 16.4𝐵.1Figure: Enhancers: An enhancer is a DNA sequence that promotes transcription. Each enhancer is made up of short DNA sequences called distal control elements. Activators bound to the distal control elements interact with mediator proteins and transcription factors.
RNA processing
The RNA species resulting from the transcription of these genes are also discontinuous and must therefore be processed before exiting the nucleus to be translated or used in the cytosol as mature RNAs. In eukaryotic systems this includes RNA splicing, 5' capping, 3' end cleavage and polyadenylation. This series of steps is a complex molecular process that must occur within the closed confines of the nucleus. Each one of these steps provides an opportunity for regulating the abundance of exported transcripts and the functional forms that these transcripts will take. While these would be topics for more advanced courses, think about how to frame some of the following topics as subproblems of the Design Challenge of genetic regulation. If nothing else, begin to appreciate the highly orchestrated molecular dance that must occur to express a gene and how this is a stunning bit of evolutionary engineering.
RNA Stability and microRNAs
After processing of mRNAs, microRNAs influence the stability of the RNA in the cell. The microRNAs, or miRNAs, are short RNA molecules that are only 21–24 nucleotides in length. The miRNAs are transcribed in the nucleus as longer pre-miRNAs. These pre-miRNAs are subsequently chopped into mature miRNAs by a protein called dicer. These mature miRNAs recognize a specific sequence of a target RNA through complementary base pairing. miRNAs, however, also associate with a ribonucleoprotein complex called the RNA-induced silencing complex (RISC). RISC binds a target mRNA, along with the miRNA, to degrade the target mRNA. Together, miRNAs and the RISC complex rapidly destroy the RNA molecule. As one might expect, the transcription of pre-miRNAs and their subsequent processing is also tightly regulated.
Nuclear export
Fully processed, mature transcripts, must be exported through the nucleus. Not surprisingly this process involves the coordination of a mature RNA species to which are bound many accessory proteins - some of which have been intimately involved in the modifications discussed above - and a protein complex called the nuclear pore complex (NPC). Transport through the NPC allows flow of proteins and RNA species to move in both directions and is mediated by a number of proteins. This process can be used to selectively regulate the transport of various transcripts depending on which proteins associate with the transcript in question. This means that not all transcripts are treated equally by the NPC - depending on modification state and the proteins that have associated with a specific species of RNA it can be moved either more or less efficiently across the nuclear membrane. Since the rate of movement across the pore will influence the abundance of mature transcript that is exported into the cytosol for translation export control is another example of a step in the process of gene regulation that can be modulated. In addition, recent research has implicated interactions between the NPC and transcription factors in the regulation of transcription initiation, likely through some mechanism whereby the transcription factors tether themselves to the nuclear pores. This last example demonstrates how interconnected the regulation of gene expression is across the multiple steps of this complex process.
We know many additional details of the processes described above to some level of detail, but many more questions remain to be answered. For the sake of Bis2a it is sufficient to form a model of the steps that occur in the production of a mature transcript in eukaryotic organisms. We have painted a picture with very broad strokes, trying to present a scene that reflect what happens generally in all eukaryotes. Besides learning the key differentiating features of eukaryotic gene regulation, we would also like for Bis2a students to think of each of these steps as an opportunity for Nature to regulate gene expression in some way and to rationalize how deficiencies or changes in these pathways - potentially introduced through mutation - might influence gene expression.
While we did not explicitly bring up the Design Challenge or Energy Story here these formalisms are equally adept at helping you to make some sense of what is being described. We encourage you to try making an Energy Story for various processes. We also encourage you to use the Design Challenge rubric to reexamine the stories above: identify problems that need solving; hypothesize potential solutions and criteria for success. Use there formalisms to dig deeper and ask new questions/identify new problems or things that you don't know about the processes is what experts do. Chances are that doing this suggested exercise will lead you to identify a direction of research that someone has already pursued (you'll feel pretty smart about that!). Alternatively, you may raise some brand new question that no one has thought of yet.
Control of Protein Abundance
After a mRNA has been transported to the cytoplasm, it is translated into protein. Control of this process is largely dependent on the RNA molecule. As previously discussed, the stability of the RNA will have a large impact on its translation into a protein. As the stability changes, the amount of time that it is available for translation also changes.
Like transcription, translation is controlled by proteins complexes of proteins and nucleic acids that must associate to initiate the process. In translation, one of the first complexes that must assembles to start the process is referred to as the initiation complex. The first protein to bind to the mRNA that helps initiate translation is called eukaryotic initiation factor-2 (eIF-2). Activity of the eIF-2 protein is controlled by multiple factors. The first is whether or not it is bound to a molecule of GTP. When the eIF-2 is bound to GTP it is considered to be in an active form. The eIF-2 protein bound to GTP can bind to the small 40S ribosomal subunit. When bound, the eIF-2/40S ribosome complex, bringing with it the mRNA to be translated, also recruits the methionine initiator tRNA associates. At this point, when the initiator complex is assembled, the GTP is hydrolyzed into GDP creating an "inactive form of eIF-2 that is released, along with the inorganic phosphate, from the complex. This step, in turn, allows the large 60S ribosomal subunit to bind and to begin translating the RNA. The binding of eIF-2 to the RNA further controlled by protein phosphorylation. When eIF-2 is phosphorylated, it undergoes a conformational change and cannot bind to GTP thus inhibiting the initiation complex from forming - translation is therefore inhibited (see the figure below). In the dephosphorylated state eIF-2 can bind GTP and allow the assembly of the translation initiation complex as described above. The ability of the cell therefore to tune the assembly of the translation invitation complex via a reversible chemical modification (phosphorylation) to a regulatory protein is another example of how Nature has taken advantage of even this seemingly simple step to tuned gene expression.
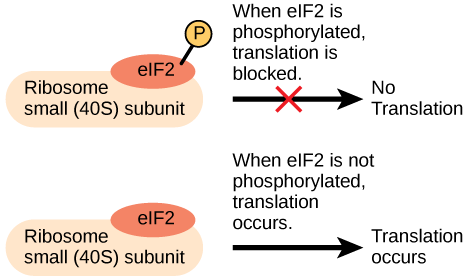
An increase in phosphorylation levels of eIF-2 has been observed in patients with neurodegenerative diseases such as Alzheimer’s, Parkinson’s, and Huntington’s. What impact do you think this might have on protein synthesis?
In conclusion, we see that gene regulation is complex and that it can be modulated at each step in the process of expressing a functional gene product. Moreover, the regulatory elements that happen at each step can act to influence other regulatory steps both earlier and later in the process of gene expression (i.e. the process of chemically altering a transcription factor can influence the regulation of its own transcription many steps earlier in the process). These complex sets of interactions form what are known as gene regulatory networks. Understanding the structure and dynamics of these networks is critical for understanding how different cells function, the basis for numerous diseases, developmental processes, and how cells make decisions about how to react to the many factors that are in constant flux both inside and outside.
Link to external resources
DNA and chromatin regulation | Biomolecules | MCAT | Khan Academy
https://www.youtube.com/watch?v=OqRt723t33o
Gene Regulation in Eukaryotes
https://www.youtube.com/watch?v=fabBvmzGBYw

