10.3: Plant Nutrition
- Page ID
- 46192
\( \newcommand{\vecs}[1]{\overset { \scriptstyle \rightharpoonup} {\mathbf{#1}} } \)
\( \newcommand{\vecd}[1]{\overset{-\!-\!\rightharpoonup}{\vphantom{a}\smash {#1}}} \)
\( \newcommand{\id}{\mathrm{id}}\) \( \newcommand{\Span}{\mathrm{span}}\)
( \newcommand{\kernel}{\mathrm{null}\,}\) \( \newcommand{\range}{\mathrm{range}\,}\)
\( \newcommand{\RealPart}{\mathrm{Re}}\) \( \newcommand{\ImaginaryPart}{\mathrm{Im}}\)
\( \newcommand{\Argument}{\mathrm{Arg}}\) \( \newcommand{\norm}[1]{\| #1 \|}\)
\( \newcommand{\inner}[2]{\langle #1, #2 \rangle}\)
\( \newcommand{\Span}{\mathrm{span}}\)
\( \newcommand{\id}{\mathrm{id}}\)
\( \newcommand{\Span}{\mathrm{span}}\)
\( \newcommand{\kernel}{\mathrm{null}\,}\)
\( \newcommand{\range}{\mathrm{range}\,}\)
\( \newcommand{\RealPart}{\mathrm{Re}}\)
\( \newcommand{\ImaginaryPart}{\mathrm{Im}}\)
\( \newcommand{\Argument}{\mathrm{Arg}}\)
\( \newcommand{\norm}[1]{\| #1 \|}\)
\( \newcommand{\inner}[2]{\langle #1, #2 \rangle}\)
\( \newcommand{\Span}{\mathrm{span}}\) \( \newcommand{\AA}{\unicode[.8,0]{x212B}}\)
\( \newcommand{\vectorA}[1]{\vec{#1}} % arrow\)
\( \newcommand{\vectorAt}[1]{\vec{\text{#1}}} % arrow\)
\( \newcommand{\vectorB}[1]{\overset { \scriptstyle \rightharpoonup} {\mathbf{#1}} } \)
\( \newcommand{\vectorC}[1]{\textbf{#1}} \)
\( \newcommand{\vectorD}[1]{\overrightarrow{#1}} \)
\( \newcommand{\vectorDt}[1]{\overrightarrow{\text{#1}}} \)
\( \newcommand{\vectE}[1]{\overset{-\!-\!\rightharpoonup}{\vphantom{a}\smash{\mathbf {#1}}}} \)
\( \newcommand{\vecs}[1]{\overset { \scriptstyle \rightharpoonup} {\mathbf{#1}} } \)
\( \newcommand{\vecd}[1]{\overset{-\!-\!\rightharpoonup}{\vphantom{a}\smash {#1}}} \)
\(\newcommand{\avec}{\mathbf a}\) \(\newcommand{\bvec}{\mathbf b}\) \(\newcommand{\cvec}{\mathbf c}\) \(\newcommand{\dvec}{\mathbf d}\) \(\newcommand{\dtil}{\widetilde{\mathbf d}}\) \(\newcommand{\evec}{\mathbf e}\) \(\newcommand{\fvec}{\mathbf f}\) \(\newcommand{\nvec}{\mathbf n}\) \(\newcommand{\pvec}{\mathbf p}\) \(\newcommand{\qvec}{\mathbf q}\) \(\newcommand{\svec}{\mathbf s}\) \(\newcommand{\tvec}{\mathbf t}\) \(\newcommand{\uvec}{\mathbf u}\) \(\newcommand{\vvec}{\mathbf v}\) \(\newcommand{\wvec}{\mathbf w}\) \(\newcommand{\xvec}{\mathbf x}\) \(\newcommand{\yvec}{\mathbf y}\) \(\newcommand{\zvec}{\mathbf z}\) \(\newcommand{\rvec}{\mathbf r}\) \(\newcommand{\mvec}{\mathbf m}\) \(\newcommand{\zerovec}{\mathbf 0}\) \(\newcommand{\onevec}{\mathbf 1}\) \(\newcommand{\real}{\mathbb R}\) \(\newcommand{\twovec}[2]{\left[\begin{array}{r}#1 \\ #2 \end{array}\right]}\) \(\newcommand{\ctwovec}[2]{\left[\begin{array}{c}#1 \\ #2 \end{array}\right]}\) \(\newcommand{\threevec}[3]{\left[\begin{array}{r}#1 \\ #2 \\ #3 \end{array}\right]}\) \(\newcommand{\cthreevec}[3]{\left[\begin{array}{c}#1 \\ #2 \\ #3 \end{array}\right]}\) \(\newcommand{\fourvec}[4]{\left[\begin{array}{r}#1 \\ #2 \\ #3 \\ #4 \end{array}\right]}\) \(\newcommand{\cfourvec}[4]{\left[\begin{array}{c}#1 \\ #2 \\ #3 \\ #4 \end{array}\right]}\) \(\newcommand{\fivevec}[5]{\left[\begin{array}{r}#1 \\ #2 \\ #3 \\ #4 \\ #5 \\ \end{array}\right]}\) \(\newcommand{\cfivevec}[5]{\left[\begin{array}{c}#1 \\ #2 \\ #3 \\ #4 \\ #5 \\ \end{array}\right]}\) \(\newcommand{\mattwo}[4]{\left[\begin{array}{rr}#1 \amp #2 \\ #3 \amp #4 \\ \end{array}\right]}\) \(\newcommand{\laspan}[1]{\text{Span}\{#1\}}\) \(\newcommand{\bcal}{\cal B}\) \(\newcommand{\ccal}{\cal C}\) \(\newcommand{\scal}{\cal S}\) \(\newcommand{\wcal}{\cal W}\) \(\newcommand{\ecal}{\cal E}\) \(\newcommand{\coords}[2]{\left\{#1\right\}_{#2}}\) \(\newcommand{\gray}[1]{\color{gray}{#1}}\) \(\newcommand{\lgray}[1]{\color{lightgray}{#1}}\) \(\newcommand{\rank}{\operatorname{rank}}\) \(\newcommand{\row}{\text{Row}}\) \(\newcommand{\col}{\text{Col}}\) \(\renewcommand{\row}{\text{Row}}\) \(\newcommand{\nul}{\text{Nul}}\) \(\newcommand{\var}{\text{Var}}\) \(\newcommand{\corr}{\text{corr}}\) \(\newcommand{\len}[1]{\left|#1\right|}\) \(\newcommand{\bbar}{\overline{\bvec}}\) \(\newcommand{\bhat}{\widehat{\bvec}}\) \(\newcommand{\bperp}{\bvec^\perp}\) \(\newcommand{\xhat}{\widehat{\xvec}}\) \(\newcommand{\vhat}{\widehat{\vvec}}\) \(\newcommand{\uhat}{\widehat{\uvec}}\) \(\newcommand{\what}{\widehat{\wvec}}\) \(\newcommand{\Sighat}{\widehat{\Sigma}}\) \(\newcommand{\lt}{<}\) \(\newcommand{\gt}{>}\) \(\newcommand{\amp}{&}\) \(\definecolor{fillinmathshade}{gray}{0.9}\)- Discuss the common nutritional needs of plants
Plants obtain food in two different ways. Autotrophic plants can make their own food from inorganic raw materials, such as carbon dioxide and water, through photosynthesis in the presence of sunlight. Green plants are included in this group. Some plants, however, are heterotrophic: they are totally parasitic and lacking in chlorophyll. These plants, referred to as holo-parasitic plants, are unable to synthesize organic carbon and draw all of their nutrients from the host plant.
Plants may also enlist the help of microbial partners in nutrient acquisition. Particular species of bacteria and fungi have evolved along with certain plants to create a mutualistic symbiotic relationship with roots. This improves the nutrition of both the plant and the microbe. The formation of nodules in legume plants and mycorrhization can be considered among the nutritional adaptations of plants. However, these are not the only type of adaptations that we may find; many plants have other adaptations that allow them to thrive under specific conditions.
Nutritional Requirements
Plants are unique organisms that can absorb nutrients and water through their root system, as well as carbon dioxide from the atmosphere. Soil quality and climate are the major determinants of plant distribution and growth. The combination of soil nutrients, water, and carbon dioxide, along with sunlight, allows plants to grow.
The Chemical Composition of Plants
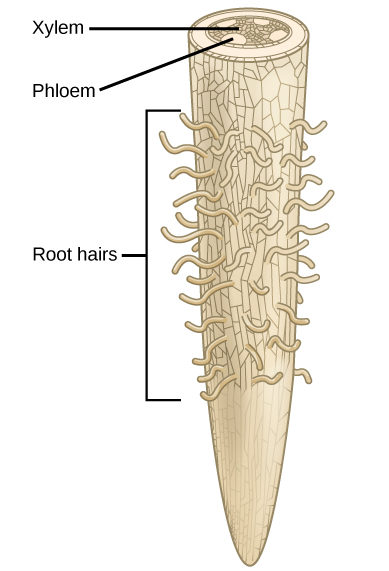
Since plants require nutrients in the form of elements such as carbon and potassium, it is important to understand the chemical composition of plants. The majority of volume in a plant cell is water; it typically comprises 80 to 90 percent of the plant’s total weight. Soil is the water source for land plants, and can be an abundant source of water, even if it appears dry. Plant roots absorb water from the soil through root hairs and transport it up to the leaves through the xylem. As water vapor is lost from the leaves, the process of transpiration and the polarity of water molecules (which enables them to form hydrogen bonds) draws more water from the roots up through the plant to the leaves (Figure 1). Plants need water to support cell structure, for metabolic functions, to carry nutrients, and for photosynthesis.
Plant cells need essential substances, collectively called nutrients, to sustain life. Plant nutrients may be composed of either organic or inorganic compounds. An organic compound is a chemical compound that contains carbon, such as carbon dioxide obtained from the atmosphere. Carbon that was obtained from atmospheric CO2 composes the majority of the dry mass within most plants. An inorganic compound does not contain carbon and is not part of, or produced by, a living organism. Inorganic substances, which form the majority of the soil solution, are commonly called minerals: those required by plants include nitrogen (N) and potassium (K) for structure and regulation.
Essential Nutrients
Plants require only light, water and about 20 elements to support all their biochemical needs: these 20 elements are called essential nutrients (Table 1). For an element to be regarded as essential, three criteria are required: 1) a plant cannot complete its life cycle without the element; 2) no other element can perform the function of the element; and 3) the element is directly involved in plant nutrition.
| Table 1. Essential Elements for Plant Growth | |
|---|---|
| Macronutrients | Micronutrients |
| Carbon (C) | Iron (Fe) |
| Hydrogen (H) | Manganese (Mn) |
| Oxygen (O) | Boron (B) |
| Nitrogen (N) | Molybdenum (Mo) |
| Phosphorus (P) | Copper (Cu) |
| Potassium (K) | Zinc (Zn) |
| Calcium (Ca) | Chlorine (Cl) |
| Magnesium (Mg) | Nickel (Ni) |
| Sulfur (S) | Cobalt (Co) |
| Sodium (Na) | |
| Silicon (Si) | |
Macronutrients and Micronutrients
The essential elements can be divided into two groups: macronutrients and micronutrients. Nutrients that plants require in larger amounts are called macronutrients. About half of the essential elements are considered macronutrients: carbon, hydrogen, oxygen, nitrogen, phosphorus, potassium, calcium, magnesium and sulfur. The first of these macronutrients, carbon (C), is required to form carbohydrates, proteins, nucleic acids, and many other compounds; it is therefore present in all macromolecules. On average, the dry weight (excluding water) of a cell is 50 percent carbon. As shown in Figure 2, carbon is a key part of plant biomolecules.

The next most abundant element in plant cells is nitrogen (N); it is part of proteins and nucleic acids. Nitrogen is also used in the synthesis of some vitamins. Hydrogen and oxygen are macronutrients that are part of many organic compounds, and also form water. Oxygen is necessary for cellular respiration; plants use oxygen to store energy in the form of ATP. Phosphorus (P), another macromolecule, is necessary to synthesize nucleic acids and phospholipids. As part of ATP, phosphorus enables food energy to be converted into chemical energy through oxidative phosphorylation. Likewise, light energy is converted into chemical energy during photophosphorylation in photosynthesis, and into chemical energy to be extracted during respiration. Sulfur is part of certain amino acids, such as cysteine and methionine, and is present in several coenzymes. Sulfur also plays a role in photosynthesis as part of the electron transport chain, where hydrogen gradients play a key role in the conversion of light energy into ATP. Potassium (K) is important because of its role in regulating stomatal opening and closing. As the openings for gas exchange, stomata help maintain a healthy water balance; a potassium ion pump supports this process.
Magnesium (Mg) and calcium (Ca) are also important macronutrients. The role of calcium is twofold: to regulate nutrient transport, and to support many enzyme functions. Magnesium is important to the photosynthetic process. These minerals, along with the micronutrients, which are described below, also contribute to the plant’s ionic balance.
In addition to macronutrients, organisms require various elements in small amounts. These micronutrients, or trace elements, are present in very small quantities. They include boron (B), chlorine (Cl), manganese (Mn), iron (Fe), zinc (Zn), copper (Cu), molybdenum (Mo), nickel (Ni), silicon (Si), and sodium (Na).

Deficiencies in any of these nutrients—particularly the macronutrients—can adversely affect plant growth (Figure 3). Depending on the specific nutrient, a lack can cause stunted growth, slow growth, or chlorosis (yellowing of the leaves). Extreme deficiencies may result in leaves showing signs of cell death.
Visit this website to participate in an interactive experiment on plant nutrient deficiencies. You can adjust the amounts of N, P, K, Ca, Mg, and Fe that plants receive . . . and see what happens.Hydroponics is a method of growing plants in a water-nutrient solution instead of soil. Since its advent, hydroponics has developed into a growing process that researchers often use. Scientists who are interested in studying plant nutrient deficiencies can use hydroponics to study the effects of different nutrient combinations under strictly controlled conditions. Hydroponics has also developed as a way to grow flowers, vegetables, and other crops in greenhouse environments. You might find hydroponically grown produce at your local grocery store. Today, many lettuces and tomatoes in your market have been hydroponically grown.
Plants can absorb inorganic nutrients and water through their root system, and carbon dioxide from the environment. The combination of organic compounds, along with water, carbon dioxide, and sunlight, produce the energy that allows plants to grow. Inorganic compounds form the majority of the soil solution. Plants access water though the soil. Water is absorbed by the plant root, transports nutrients throughout the plant, and maintains the structure of the plant. Essential elements are indispensable elements for plant growth. They are divided into macronutrients and micronutrients. The macronutrients plants require are carbon, nitrogen, hydrogen, oxygen, phosphorus, potassium, calcium, magnesium, and sulfur. Important micronutrients include iron, manganese, boron, molybdenum, copper, zinc, chlorine, nickel, cobalt, silicon and sodium.
Autotrophic Plants
Nitrogen Fixation: Root and Bacteria Interactions
Nitrogen is an important macronutrient because it is part of nucleic acids and proteins. Atmospheric nitrogen, which is the diatomic molecule N2, or dinitrogen, is the largest pool of nitrogen in terrestrial ecosystems. However, plants cannot take advantage of this nitrogen because they do not have the necessary enzymes to convert it into biologically useful forms. However, nitrogen can be “fixed,” which means that it can be converted to ammonia (NH3) through biological, physical, or chemical processes. Biological nitrogen fixation (BNF) is the conversion of atmospheric nitrogen (N2) into ammonia (NH3), exclusively carried out by prokaryotes such as soil bacteria or cyanobacteria. Biological processes contribute 65 percent of the nitrogen used in agriculture.
The most important source of BNF is the symbiotic interaction between soil bacteria and legume plants, including many crops important to humans (Figure 4). The NH3 resulting from fixation can be transported into plant tissue and incorporated into amino acids, which are then made into plant proteins. Some legume seeds, such as soybeans and peanuts, contain high levels of protein, and serve among the most important agricultural sources of protein in the world.

Farmers often rotate corn (a cereal crop) and soy beans (a legume), planting a field with each crop in alternate seasons. What advantage might this crop rotation confer?
[practice-area rows=”2″][/practice-area]
[reveal-answer q=”890921″]Show Answer[/reveal-answer]
[hidden-answer a=”890921″]Soybeans are able to fix nitrogen in their roots, which are not harvested at the end of the growing season. The belowground nitrogen can be used in the next season by the corn.[/hidden-answer]
Soil bacteria, collectively called rhizobia, symbiotically interact with legume roots to form specialized structures called nodules, in which nitrogen fixation takes place. This process entails the reduction of atmospheric nitrogen to ammonia, by means of the enzyme nitrogenase. Therefore, using rhizobia is a natural and environmentally friendly way to fertilize plants, as opposed to chemical fertilization that uses a nonrenewable resource, such as natural gas. Through symbiotic nitrogen fixation, the plant benefits from using an endless source of nitrogen from the atmosphere. The process simultaneously contributes to soil fertility because the plant root system leaves behind some of the biologically available nitrogen. As in any symbiosis, both organisms benefit from the interaction: the plant obtains ammonia, and bacteria obtain carbon compounds generated through photosynthesis, as well as a protected niche in which to grow (Figure 5).

Mycorrhizae: The Symbiotic Relationship between Fungi and Roots
A nutrient depletion zone can develop when there is rapid soil solution uptake, low nutrient concentration, low diffusion rate, or low soil moisture. These conditions are very common; therefore, most plants rely on fungi to facilitate the uptake of minerals from the soil. Fungi form symbiotic associations called mycorrhizae with plant roots, in which the fungi actually are integrated into the physical structure of the root. The fungi colonize the living root tissue during active plant growth.

Through mycorrhization, the plant obtains mainly phosphate and other minerals, such as zinc and copper, from the soil. The fungus obtains nutrients, such as sugars, from the plant root (Figure 6). Mycorrhizae help increase the surface area of the plant root system because hyphae, which are narrow, can spread beyond the nutrient depletion zone. Hyphae can grow into small soil pores that allow access to phosphorus that would otherwise be unavailable to the plant. The beneficial effect on the plant is best observed in poor soils. The benefit to fungi is that they can obtain up to 20 percent of the total carbon accessed by plants. Mycorrhizae functions as a physical barrier to pathogens. It also provides an induction of generalized host defense mechanisms, and sometimes involves production of antibiotic compounds by the fungi.
There are two types of mycorrhizae: ectomycorrhizae and endomycorrhizae. Ectomycorrhizae form an extensive dense sheath around the roots, called a mantle. Hyphae from the fungi extend from the mantle into the soil, which increases the surface area for water and mineral absorption. This type of mycorrhizae is found in forest trees, especially conifers, birches, and oaks. Endomycorrhizae, also called arbuscular mycorrhizae, do not form a dense sheath over the root. Instead, the fungal mycelium is embedded within the root tissue. Endomycorrhizae are found in the roots of more than 80 percent of terrestrial plants.
Heterotrophic Plants
Some plants cannot produce their own food and must obtain their nutrition from outside sources—these plants are heterotrophic. This may occur with plants that are parasitic or saprophytic. Some plants are mutualistic symbionts, epiphytes, or insectivorous.
Plant Parasites
A parasitic plant depends on its host for survival. Some parasitic plants have no leaves. An example of this is the dodder (Figure 7a), which has a weak, cylindrical stem that coils around the host and forms suckers. From these suckers, cells invade the host stem and grow to connect with the vascular bundles of the host. The parasitic plant obtains water and nutrients through these connections. The plant is a total parasite (a holoparasite) because it is completely dependent on its host. Other parasitic plants (hemiparasites) are fully photosynthetic and only use the host for water and minerals. There are about 4,100 species of parasitic plants.
Saprophytes
A saprophyte is a plant that does not have chlorophyll and gets its food from dead matter, similar to bacteria and fungi (note that fungi are often called saprophytes, which is incorrect, because fungi are not plants). Plants like these use enzymes to convert organic food materials into simpler forms from which they can absorb nutrients (Figure 7b). Most saprophytes do not directly digest dead matter: instead, they parasitize fungi that digest dead matter, or are mycorrhizal, ultimately obtaining photosynthate from a fungus that derived photosynthate from its host. Saprophytic plants are uncommon; only a few species are described.

Symbionts
A symbiont is a plant in a symbiotic relationship, with special adaptations such as mycorrhizae or nodule formation. Fungi also form symbiotic associations with cyanobacteria and green algae (called lichens). Lichens can sometimes be seen as colorful growths on the surface of rocks and trees (Figure 8a). The algal partner (phycobiont) makes food autotrophically, some of which it shares with the fungus; the fungal partner (mycobiont) absorbs water and minerals from the environment, which are made available to the green alga. If one partner was separated from the other, they would both die.
Epiphytes
An epiphyte is a plant that grows on other plants, but is not dependent upon the other plant for nutrition (Figure 8b). Epiphytes have two types of roots: clinging aerial roots, which absorb nutrients from humus that accumulates in the crevices of trees; and aerial roots, which absorb moisture from the atmosphere.

Insectivorous Plants

An insectivorous plant has specialized leaves to attract and digest insects. The Venus flytrap is popularly known for its insectivorous mode of nutrition, and has leaves that work as traps (Figure 9).
The minerals it obtains from prey compensate for those lacking in the boggy (low pH) soil of its native North Carolina coastal plains. There are three sensitive hairs in the center of each half of each leaf. The edges of each leaf are covered with long spines. Nectar secreted by the plant attracts flies to the leaf. When a fly touches the sensory hairs, the leaf immediately closes. Next, fluids and enzymes break down the prey and minerals are absorbed by the leaf. Since this plant is popular in the horticultural trade, it is threatened in its original habitat.
Contributors and Attributions
- Introduction to Plant Nutrition. Authored by: Shelli Carter and Lumen Learning. Provided by: Lumen Learning. License: CC BY: Attribution
- Biology. Provided by: OpenStax CNX. Located at: http://cnx.org/contents/185cbf87-c72e-48f5-b51e-f14f21b5eabd@10.8. License: CC BY: Attribution. License Terms: Download for free at http://cnx.org/contents/185cbf87-c72...f21b5eabd@10.8

