3.2: Carbon
- Page ID
- 35668
What you’ll learn to do: Discuss why it is said that life is carbon-based and the bonding properties of carbon
You’ve probably heard the term carbon-based life thrown about in scientific conversations before. But what exactly does the term mean? Possibly the quickest answer to this question is simply that all living things are reliant on molecules that include carbon. There are no living things on our planet that do not have carbon (however, there are nonliving things made up of carbon as well: e.g, diamonds and, well, carbon itself).
In this outcome we’ll learn about the importance of carbon to every living thing on earth and its unique properties that make it particularly suited for its role in living things.
Discuss why it is said that life is carbon-based and the bonding properties of carbon.
Carbon
Living things are carbon-based because carbon plays such a prominent role in the chemistry of living things. This means that carbon atoms, bonded to other carbon atoms or other elements, form the fundamental components of many, if not most, of the molecules found uniquely in living things. Other elements play important roles in biological molecules, but carbon certainly qualifies as the “foundation” element for molecules in living things. It is the bonding properties of carbon atoms that are responsible for its important role.
Carbon Bonding
The four covalent bonding positions of the carbon atom can give rise to a wide diversity of compounds with many functions, accounting for the importance of carbon in living things.
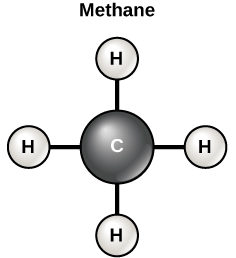
Carbon contains four electrons in its outer shell. Therefore, it can form four covalent bonds with other atoms or molecules. The simplest organic carbon molecule is methane (CH4), in which four hydrogen atoms bind to a carbon atom (Figure 1).
However, structures that are more complex are made using carbon. Any of the hydrogen atoms can be replaced with another carbon atom covalently bonded to the first carbon atom. In this way, long and branching chains of carbon compounds can be made (Figure 2a). The carbon atoms may bond with atoms of other elements, such as nitrogen, oxygen, and phosphorus (Figure 2b). The molecules may also form rings, which themselves can link with other rings (Figure 2c). This diversity of molecular forms accounts for the diversity of functions of the biological macromolecules and is based to a large degree on the ability of carbon to form multiple bonds with itself and other atoms.

Check Your Understanding
Answer the question(s) below to see how well you understand the topics covered in the previous section. This short quiz does not count toward your grade in the class, and you can retake it an unlimited number of times.
Use this quiz to check your understanding and decide whether to (1) study the previous section further or (2) move on to the next section.
Contributors and Attributions
- Introduction to Carbon. Provided by: Lumen Learning. License: CC BY: Attribution
- Concepts of Biology. Provided by: OpenStax CNX. Located at: http://cnx.org/contents/b3c1e1d2-839c-42b0-a314-e119a8aafbdd@9.25. License: CC BY: Attribution. License Terms: Download for free at http://cnx.org/contents/b3c1e1d2-839...9a8aafbdd@9.25
- Biology. Provided by: OpenStax CNX. Located at: http://cnx.org/contents/185cbf87-c72e-48f5-b51e-f14f21b5eabd@10.8. License: CC BY: Attribution. License Terms: Download for free at http://cnx.org/contents/185cbf87-c72...f21b5eabd@10.8

