22.4B: Biofilms and Disease
- Page ID
- 13568
Biofilms, complex colonies of bacteria acting as a unit in their release of toxins, are highly resistant to antibiotics and host defense.
- Give examples of the roles played by biofilms in human diseases
Key Points
- Once a biofilm infection is established, it is very difficult to eradicate because biofilms exhibit great resistance to most methods used to control microbial growth, including antibiotics.
- Biofilms are able to grow anywhere there is an optimal combination of moisture, nutrients, and a surface.
- Biofilms are responsible for diseases such as infections in patients and readily settle within wounds and burns; they can also easily colonize medical devices and other surfaces where sterility is vital for health.
Key Terms
- biofilm: a thin film of mucus created by and containing a colony of bacteria and other microorganisms
- nosocomial: contracted in a hospital, or arising from hospital treatment
Biofilms and Disease
Biofilms are complex colonies of bacteria (often containing several species) that exchange chemical signals to coordinate the release of toxins that will attack the host. Once established, they are very difficult to destroy as they are highly resistant to antimicrobial treatments and host defense. Biofilms form when microorganisms adhere to the surface of some object in a moist environment and begin to reproduce. They grow virtually everywhere in almost any environment where there is a combination of moisture, nutrients, and a surface. Biofilms are responsible for diseases such as infections in patients with cystic fibrosis, Legionnaires’ disease, and otitis media. They produce dental plaque and colonize catheters, prostheses, transcutaneous and orthopedic devices, contact lenses, and internal devices such as pacemakers. They also form in open wounds and burned tissue. In healthcare environments, biofilms grow on hemodialysis machines, mechanical ventilators, shunts, and other medical equipment. In fact, 65 percent of all infections acquired in the hospital (nosocomial infections) are attributed to biofilms. Biofilms are also related to diseases contracted from food because they colonize the surfaces of vegetable leaves and meat, as well as food-processing equipment that is not adequately cleaned.
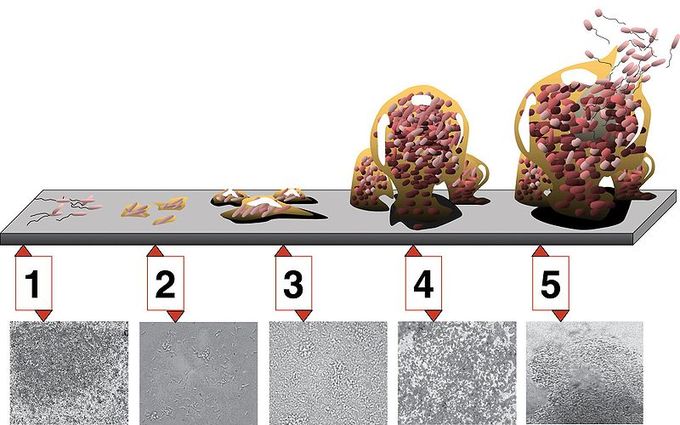
Biofilm infections develop gradually and often do not cause immediate symptoms. They are rarely resolved by host defense mechanisms. Once an infection by a biofilm is established, it is very difficult to eradicate because biofilms tend to be resistant to most of the methods used to control microbial growth, including antibiotics. Biofilms respond poorly or only temporarily to antibiotics. It has been said that they can resist up to 1,000 times the antibiotic concentrations used to kill the same bacteria when they are free-living or planktonic. An antibiotic dose that large would harm the patient; therefore, scientists are working on new ways to eradicate biofilms.


