2.8: Atoms, Isotopes, Ions, and Molecules - Ions and Ionic Bonds
- Page ID
- 12663
- Predict whether a given element will more likely form a cation or an anion
Ions and Ionic Bonds
Some atoms are more stable when they gain or lose an electron (or possibly two) and form ions. This results in a full outermost electron shell and makes them energetically more stable. Now, because the number of electrons does not equal the number of protons, each ion has a net charge. Cations are positive ions that are formed by losing electrons (as the number of protons is now greater than the number of electrons). Negative ions are formed by gaining electrons and are called anions (wherein there are more electrons than protons in a molecule ). Anions are designated by their elemental name being altered to end in “-ide”. For example, the anion of chlorine is called chloride, and the anion of sulfur is called sulfide.
This movement of electrons from one element to another is referred to as electron transfer. As illustrated, sodium (Na) only has one electron in its outer electron shell. It takes less energy for sodium to donate that one electron than it does to accept seven more electrons to fill the outer shell. When sodium loses an electron, it will have 11 protons, 11 neutrons, and only 10 electrons. This leaves it with an overall charge of +1 since there are now more protons than electrons. It is now referred to as a sodium ion. Chlorine (Cl) in its lowest energy state (called the ground state) has seven electrons in its outer shell. Again, it is more energy efficient for chlorine to gain one electron than to lose seven. Therefore, it tends to gain an electron to create an ion with 17 protons, 17 neutrons, and 18 electrons. This gives it a net charge of -1 since there are now more electrons than protons. It is now referred to as a chloride ion. In this example, sodium will donate its one electron to empty its shell, and chlorine will accept that electron to fill its shell. Both ions now satisfy the octet rule and have complete outer shells. These transactions can normally only take place simultaneously; in order for a sodium atom to lose an electron, it must be in the presence of a suitable recipient like a chlorine atom.
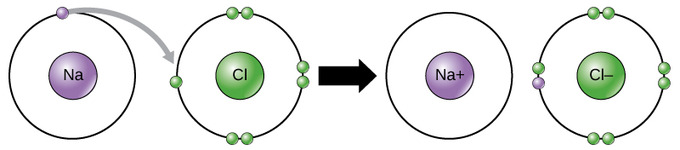
Ionic bonds are formed between ions with opposite charges. For instance, positively charged sodium ions and negatively charged chloride ions bond together to form sodium chloride, or table salt, a crystalline molecule with zero net charge. The attractive force holding the two atoms together is called the electromagnetic force and is responsible for the attraction between oppositely charged ions.
Certain salts are referred to in physiology as electrolytes (including sodium, potassium, and calcium). Electrolytes are ions necessary for nerve impulse conduction, muscle contractions, and water balance. Many sports drinks and dietary supplements provide these ions to replace those lost from the body via sweating during exercise.
Key Points
- Ions form from elements when they gain or lose an electron causing the number of protons to be unequal to the number of electrons, resulting in a net charge.
- If there are more electrons than protons (from an element gaining one or more electrons), the ion is negatively charged and called an anion.
- If there are more protons than electrons (via loss of electrons), the ion is positively charged and is called a cation.
- Ionic bonds result from the interaction between a positively charged cation and a negatively charged anion.
Key Terms
- ion: An atom, or group of atoms, bearing an electrical charge, such as the sodium and chlorine atoms in a salt solution.
- ionic bond: A strong chemical bond caused by the electrostatic attraction between two oppositely charged ions.


