2.21: Carbon - Organic Enantiomers
- Page ID
- 12679
- Give examples of enantiomers
Stereoisomers are a type of isomer where the order of the atoms in the two molecules is the same but their arrangement in space is different. The two main types of stereoisomerism are diastereomerism (including ‘cis-trans isomerism’) and optical isomerism (also known as ‘enantiomerism’ and ‘chirality’). Optical isomers are stereoisomers formed when asymmetric centers are present; for example, a carbon with four different groups bonded to it. Enantiomers are two optical isomers (i.e. isomers that are reflections of each other). Every stereocenter in one isomer has the opposite configuration in the other. They share the same chemical structure and chemical bonds, but differ in the three-dimensional placement of atoms so that they are mirror images, much as a person’s left and right hands are. Compounds that are enantiomers of each other have the same physical properties except for the direction in which they rotate polarized light and how they interact with different optical isomers of other compounds.
The amino acid alanine is example of an entantiomer. The two structures, D-alanine and L-alanine, are non-superimposable. In nature, only the L-forms of amino acids are used to make proteins. Some D forms of amino acids are seen in the cell walls of bacteria, but never in their proteins. Similarly, the D-form of glucose is the main product of photosynthesis and the L-form of the molecule is rarely seen in nature.
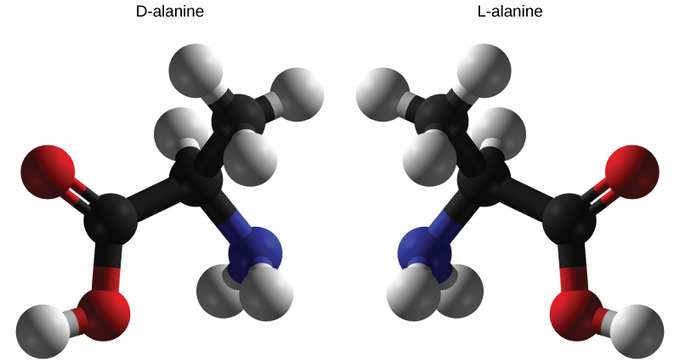
Organic compounds that contain a chiral carbon usually have two non-superposable structures. These two structures are mirror images of each other and are, thus, commonly called enantiomorphs; hence, this structural property is now commonly referred to as enantiomerism. Enantiopure compounds refer to samples having, within the limits of detection, molecules of only one chirality.
Enantiomers of each other often show different chemical reactions with other substances that are also enantiomers. Since many molecules in the bodies of living beings are enantiomers themselves, there is sometimes a marked difference in the effects of two enantiomers on living beings. In drugs, for example, often only one of a drug’s enantiomers is responsible for the desired physiologic effects, while the other enantiomer is less active, inactive, or sometimes even responsible for adverse effects. Owing to this discovery, drugs composed of only one enantiomer (“enantiopure”) can be developed to enhance the pharmacological efficacy and sometimes do away with some side effects.
Key Points
- Enantiomers are stereoisomers, a type of isomer where the order of the atoms in the two molecules is the same but their arrangement in space is different.
- Many molecules in the bodies of living beings are enantiomers; there is sometimes a large difference in the effects of two enantiomers on organisms.
- Enantiopure compounds refer to samples having, within the limits of detection, molecules of only one chirality.
- Compounds that are enantiomers of each other have the same physical properties except for the direction in which they rotate polarized light and how they interact with different optical isomers of other compounds.
Key Terms
- enantiomer: One of a pair of stereoisomers that is the mirror image of the other, but may not be superimposed on this other stereoisomer.
- chirality: The phenomenon in chemistry, physics, and mathematics in which objects are mirror images of each other, but are not identical.
- stereoisomer: one of a set of the isomers of a compound in which atoms are arranged differently about a chiral center; they exhibit optical activity


