4.4: The Action Potential in Neurons
- Page ID
- 16110
\( \newcommand{\vecs}[1]{\overset { \scriptstyle \rightharpoonup} {\mathbf{#1}} } \)
\( \newcommand{\vecd}[1]{\overset{-\!-\!\rightharpoonup}{\vphantom{a}\smash {#1}}} \)
\( \newcommand{\id}{\mathrm{id}}\) \( \newcommand{\Span}{\mathrm{span}}\)
( \newcommand{\kernel}{\mathrm{null}\,}\) \( \newcommand{\range}{\mathrm{range}\,}\)
\( \newcommand{\RealPart}{\mathrm{Re}}\) \( \newcommand{\ImaginaryPart}{\mathrm{Im}}\)
\( \newcommand{\Argument}{\mathrm{Arg}}\) \( \newcommand{\norm}[1]{\| #1 \|}\)
\( \newcommand{\inner}[2]{\langle #1, #2 \rangle}\)
\( \newcommand{\Span}{\mathrm{span}}\)
\( \newcommand{\id}{\mathrm{id}}\)
\( \newcommand{\Span}{\mathrm{span}}\)
\( \newcommand{\kernel}{\mathrm{null}\,}\)
\( \newcommand{\range}{\mathrm{range}\,}\)
\( \newcommand{\RealPart}{\mathrm{Re}}\)
\( \newcommand{\ImaginaryPart}{\mathrm{Im}}\)
\( \newcommand{\Argument}{\mathrm{Arg}}\)
\( \newcommand{\norm}[1]{\| #1 \|}\)
\( \newcommand{\inner}[2]{\langle #1, #2 \rangle}\)
\( \newcommand{\Span}{\mathrm{span}}\) \( \newcommand{\AA}{\unicode[.8,0]{x212B}}\)
\( \newcommand{\vectorA}[1]{\vec{#1}} % arrow\)
\( \newcommand{\vectorAt}[1]{\vec{\text{#1}}} % arrow\)
\( \newcommand{\vectorB}[1]{\overset { \scriptstyle \rightharpoonup} {\mathbf{#1}} } \)
\( \newcommand{\vectorC}[1]{\textbf{#1}} \)
\( \newcommand{\vectorD}[1]{\overrightarrow{#1}} \)
\( \newcommand{\vectorDt}[1]{\overrightarrow{\text{#1}}} \)
\( \newcommand{\vectE}[1]{\overset{-\!-\!\rightharpoonup}{\vphantom{a}\smash{\mathbf {#1}}}} \)
\( \newcommand{\vecs}[1]{\overset { \scriptstyle \rightharpoonup} {\mathbf{#1}} } \)
\( \newcommand{\vecd}[1]{\overset{-\!-\!\rightharpoonup}{\vphantom{a}\smash {#1}}} \)
The transport of solutes in and out of cells is critical to life. However, in neurons, the movement of ions has another crucial function in metazoan animals: production of action potentials used for neurotransmission. This specialization allows for extremely rapid transmission of information across long distances. An example my mentor would use when teaching basic neuroscience to schoolchildren was a bipolar neuron that extends from the toe to the brain. For information from the toe to be useful, it must reach the brain for processing very quickly. This signal may have to travel several meters in a giraffe, far more in whales. No chemical signal can move that quickly, and despite the popular simplification and dramatic depictions on television, electricity does not flow through neurons as though they were copper wires.
A number of potent toxins have been used in the study of the AchR, including histrionicatoxin, an arrow poison extracted from a South American tree frog, Dendrobates histrionicus, curare (d-tubocurarine) isolated from certain plants, and a-bungarotoxin, which is isolated from the Taiwanese snake, the banded krait. All three act by competitive inhibition, blocking the opening of the AchR channel.
Acetylcholine spontaneously dissociates from AchR in about a millisecond, closing the channel. It could then be reactivated by the acetylcholine, but this is tightly regulated by the enzyme acetylcholinesterase (AchE), which is a GPI-anchored protein on the membrane of the target cell, and prevents overstimulation by rapid degradation of the acetylcholine. The military nerve gas sarin, which gained notoriety in a 1995 attack on the Tokyo subway system, works by inactivating AchE, thus causing over- stimulation of AchR.
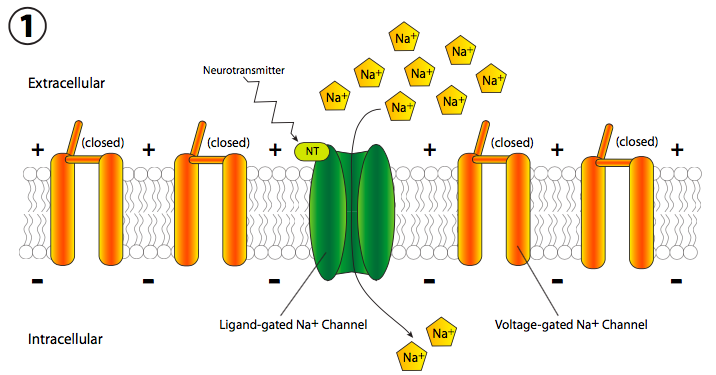
The reality does involve rapid changes to membrane potential. Since that is measured in volts and is a difference in electrical charge across the membrane, it is sometimes thought of colloquially as electrical transmission, but the mechanism is completely different from electrons flowing through a wire. Instead, there is a moving change in membrane potential due to sequential opening of ion channels along the length of the axon (the long thin part of the nerve cell). Generally, the signal starts with an excitatory chemical signal, or neurotransmitter (Figure \(\PageIndex{18}\)), that binds to a receptor on the neuron. This receptor may be linked to an ion channel or it may itself be a ligand-gated ion channel. In either case, the channel opens and Na+ comes into the cell.
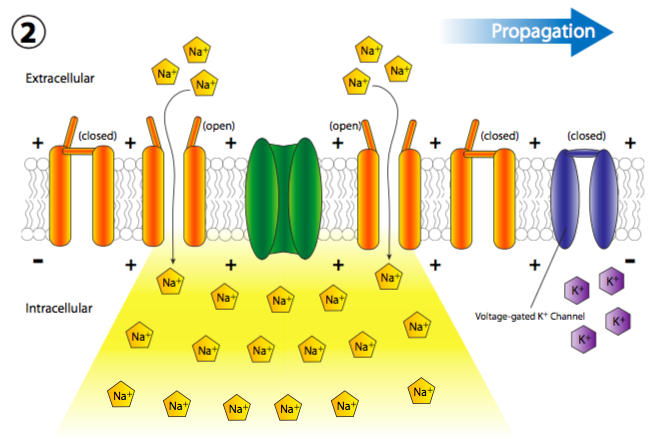
The sudden influx of positive charges transiently and locally depolarizes the membrane (more + charges along the inside of the membrane than outside). The depolarization of the membrane near the channel causes all nearby voltage-gated Na+ channels to transiently open (Figure \(\PageIndex{19}\)), thus allowing a new rush of Na+ ions into the cell that can then depolarize another small section of membrane. Although technically, channels open up on either side of the receptor, normally, the receptor is located on the main cell body, or soma, while most of the ion channels are lined up along the axon. Therefore, there is a de facto directionality to the propagation of the depolarization.
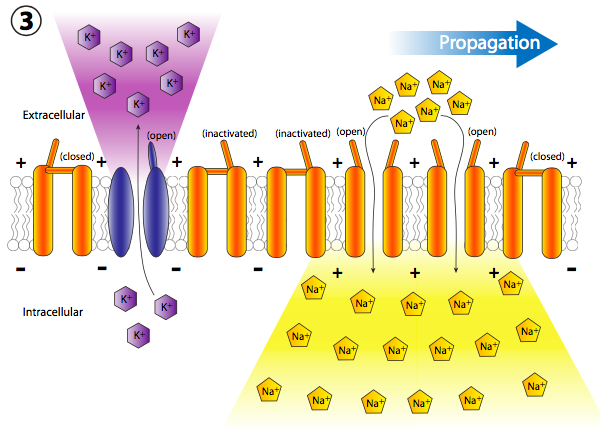
More voltage gated Na+ channels are opened up quickly after their neighboring channels have opened and depolarized their section of the membrane, leading to a long chain reaction of voltage-gated depolarizations all the way down the axon (Figure \(\PageIndex{20}\)). Once the signal is propagating down the axon, there are two questions that should come into your head. First, neurons are not disposable cells, so how are all these opened channels reset and the membrane repolarized for the next action potential? And second, why wouldn’t the depolarization wave become bidirectional since each little depolarization area spreads out in both directions from the opened Na+ gate?
In fact, the answers to both questions are closely related, and has to do with the gating mechanism of voltage-gated ion channels (Figure \(\PageIndex{21}\)). Voltage-gated ion channels have two gates: one that opens in response to an increase in membrane potential, and one that closes the channel after a short period of time. This means that there are three potential states for most voltage-gated ion channels: closed, open, and inactivated.
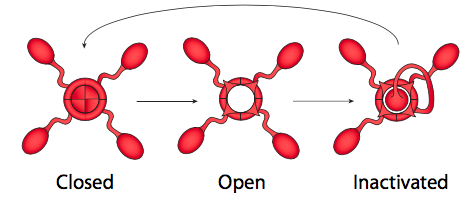
The inactivated state occurs when the second gate (more like a plug, really) closes, because this is voltage-insensitive. The plug eventually comes out of the pore and the voltage-sensitive gate is set back in place to the closed state. Voltage-sensitive K+ channels are tetramers, as shown here, and each subunit carries a potential inactivating domain. The choice of which domain appears to be random. Voltage-sensitive Na+ channels, on the other hand, also have four transmembrane regions, but they are all part of a single protein, and the channel only has one inactivation gate/domain. However, the three-state gating mechanism is still the same.
Back to the action potential: each new depolarization opens the next adjacent set of voltage gated Na+ channels, and so on. In a neuronal axon, which is where action potentials occur, the movement of the depolarizations happens very quickly and unidirectionally. It happens quickly because the axon is a very long thin projection of the cell, so the volume is small and therefore the influx of Na+ can quickly depolarize a long section of membrane. It happens unidirectionally because the previously opened voltage-sensitive Na+ channels go into a refractory inactivated state that cannot be reopened immediately.
Thus when the adjacent wave of depolarization hits, the previously opened channels do not open again, and the opening of more voltage-gated channels continues unidirectionally away from the start.
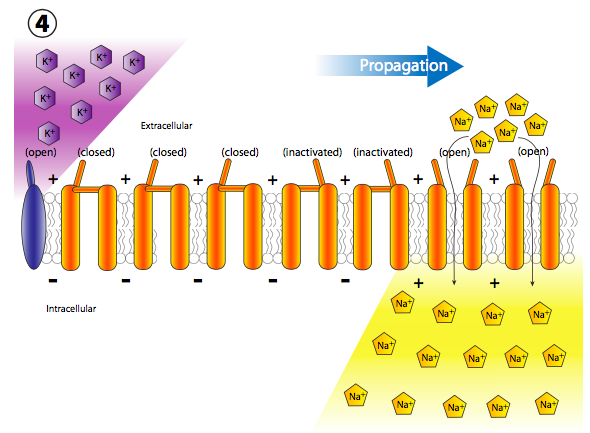
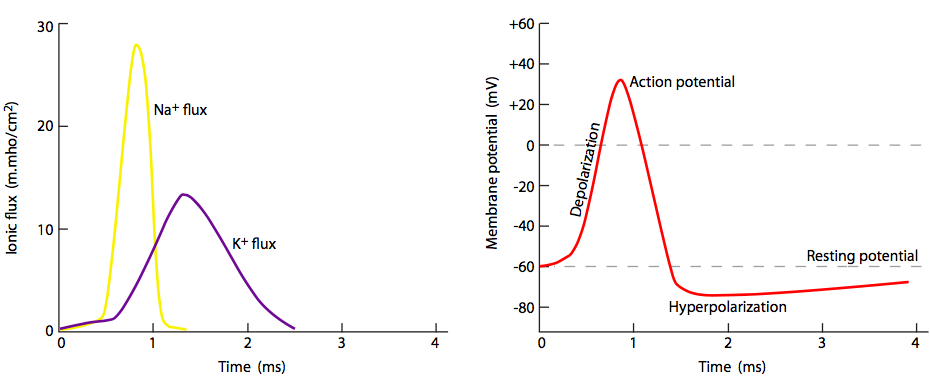
But wait, there’s more! Just because the Na+ channels are locked closed doesn’t mean that the membrane goes back to normal. What repolarizes the membrane behind the action potential? The voltage-gated potassium channels, which are interspersed among the Na+ channels (note purple ions and channels in figs. 20, 22). They open more slowly than the sodium channels (Figure \(\PageIndex{23}\)) and thus reach peak flux shortly after the peak flux of Na+. Thus, the recovery phase, or repolarization back towards the resting potential, is helped by both cessation of the inward Na+ movement and continued outward K+ movement.
Since all action potentials behave the same way from the standpoint of changes in membrane potential, the difference between stronger and weaker nerve signals is in the frequency/rate of action potential firing, not in magnitude of the ion flux.
Since neuronal transmission of information is critical to many systems, it is not surprising that a variety of organisms have evolved toxins that can paralyze or kill an attacker or prey. Interestingly, the primary target of the toxins is the voltage-gated Na+ channel, and not the K+ channel.
The toxin most widely used in research is tetrodotoxin (TTX), which is derived from the skin and certain organs of the puffer fish (Fugu rubripes). This fish is considered a delicacy (primarily in Japan), and even when prepared by expert and specially-trained chefs, the small amount of residual TTX in the flesh causes a tingling numbing of the tongue as neural excitation is blocked. Interestingly (but unrelated to ion channels) fugu has a highly compressed genome - where most other eukaryotes have large swaths of non-coding regions, fugu has little, and its genes are very close together. In fact, it has almost as many predicted genes as the human genome in only 1/8th the DNA!
Saxitoxin (STX) is another poison that acts on the voltage-gated Na+ channel. It is produced by the plankton that cause “red tide” (dino agellates of the genus Alexandrium, Gymnodium, and Pyrodinium) and is concentrated by filter-feeders such as mussels, oysters, and other shell fish. Interestingly, STX was once investigated by the US military as a nerve agent for chemical warfare, under the designation TZ. Both STX and TTX bind to the external surface of the closed voltage-gated Na+ channel near the mouth of the pore, preventing opening, and thereby blocking Na+ flow.
Batrachotoxin, on the other hand, binds to voltage-gated Na+ channels when they are open rather than closed, so there is a massive continuous influx of Na+. A product of another South American tree frog, Phyllobates aurotaenia, this toxin is used as a hunting poison by aboriginal tribes in the area, and is the most potent venom currently known. With a lethal dose of less than 2 mg/kg, batrachotoxin is roughly ten times more deadly than TTX. However, to put this in perspective, botulinum toxin, used pharmacologically as Botox® and the cause of botulism, is well over a hundred times more potent (though acting through a completely different mechanism - prevention of neurotransmitter release).


