6.1: Photosynthesis
- Page ID
- 16121
In one way or another, the energy of sugar and fat fuel molecules is derived from photosynthesis - the conversion of solar light energy into chemical bond energy, whether directly in photosynthetic plant cells and certain photosynthetic bacteria, or indirectly by the ingestion of those plants and bacteria. Photosynthesis is a simple idea: atmospheric carbon dioxide molecules are joined with water molecules to form sugars and oxygen:
\[\ce{CO2 + H2O ->[light] CH2O + O2}\]
The production of usable energy from sunlight and the fixation of atmospheric carbon dioxide are two separate sets of reactions. In plants, photosynthesis takes place only in cells containing chloroplasts. Chloroplasts are organelles with an evolutionary origin suspected to be similar to that of mitochondria, and like mitochondria, chloroplasts generate ATP and use a nicotinamide-based high-energy electron carrier. There are further similarities: they both have highly folded inner membranes, though in chloroplasts, there are three membranes in all, while mitochondria only have two. Finally, an electron transport chain is embedded in the thylakoid membrane of chloroplasts, functioning very similarly to electron transport in the mitochondria. In addition to the electron transport components and ATP synthase (structurally and functionally almost identical to mitochondrial ATP synthase), the thylakoid membrane is also rich in a set of molecules that are not found in the inner mitochondrial membrane: light-absorbing pigment molecules.
In plants, these pigment molecules fall into two classes: the chlorophylls and the carotenoids (Figure \(\PageIndex{1}\)) but only the chlorophylls can mediate photosynthesis. Photosynthetic bacteria do not contain chlorophyll, but do have carotenoid pigments that can carry out photosynthesis. Both are hydrophobic hydrocarbons that are held in place within the plane of the membrane by transmembrane proteins. Chlorophylls are easily recognizable by the very large Mg2+-containing porphyrin ring, while the carotenoids are long hydrocarbon chains that may or may not have small ring structures on the ends (e.g. β-carotene). While there is variation in the chlorophyll family, they all impart a green color to the leaf. Carotenoids, on the other hand have a much wider range of colors from yellows to reds. Both chlorophylls and carotenoids are able to absorb light energy of a particular wavelength/energy range and enter an unstable excited state. When the molecule returns to its ground state, the energy would be emitted as heat or light in an isolated situation. However within the context of the pigment arrays (antenna complex) in a living cell, most of the energy is shuttled to another pigment molecule of lower energy by resonance transfer. As described below, only one pair of chlorophyll molecules in an antenna complex will actually eject an electron as it drops from an excited state back to ground state. It is the transfer of that high-energy electron that powers photosynthesis.
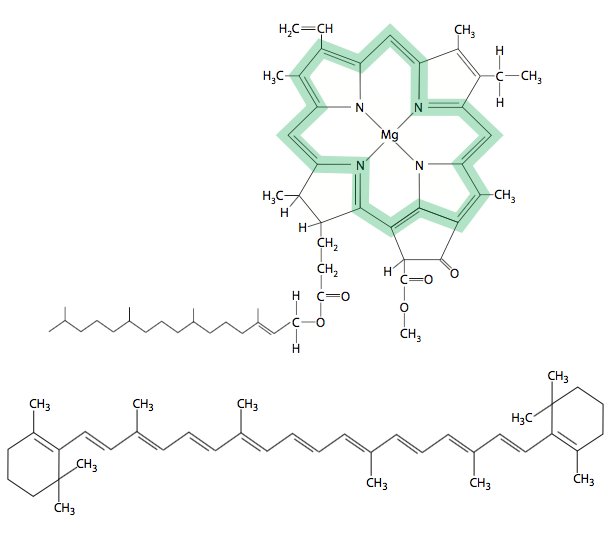
Chlorophyll molecules are made up of a phytol hydrocarbon tail that anchors the molecule within a membrane, and an electron- carrying porphyrin ring containing a magnesium cation. Note that the phytol tail is not drawn to scale with the porphyrin ring in Figure \(\PageIndex{1}\). Among different types of chlorophyll, the chemical groups attached to the ring may vary, and this variation is responsible for differences in the absorption spectrum from one type of chlorophyll to the next. For example, chlorophyll a has absorption peaks at approximately 430 and 662 nm, whereas chlorophyll b has peaks at 453 and 642 nm. The difference between the two is small: at C7, there is a —CH3 group on chlorophyll a, but a —CHO group on chlorophyll b. Presently, there are five known chlorophylls: chlorophyll a is found in all photosynthetic organisms, chlorophyll b is only found in plants, chlorophylls c1 and c2 are found in photosynthetic algae, and chlorophyll d is found in cyanobacteria.
Carotenoids have two functions. As noted in the primary text at left, they can participate in energy transfer in toward the reaction center chlorophylls. They are also a protectant molecule, preventing reaction center auto-oxidation. Carotenoids can be highly efficient free radical scavengers due to the conjugation of alternating single-double carbon bond structures.
Photosynthesis can be divided into two mechanisms: the light reactions, which use light energy to excite the electrons of certain chlorophylls, and participate in the electron transport chain to generate ATP and NADPH, and the dark reactions, which use that ATP and NADPH to fix carbon from CO2 into organic molecules (carbohydrates). As the name implies, the light reactions require light energy to excite the chlorophyll and begin electron transport. Dark reactions, however, do not require darkness. They are technically light-independent, but in some plants, the dark reactions run better in the light for reasons to be discussed.
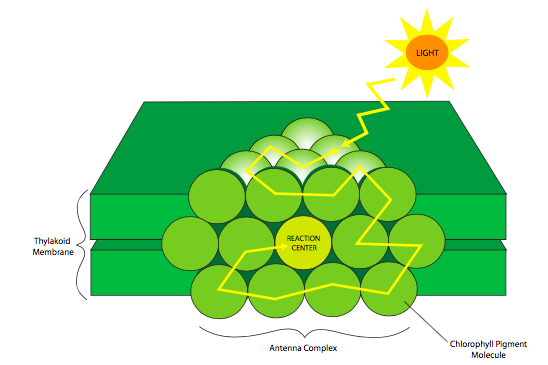
The light reactions are intimately tied to the anatomy of the thylakoid membrane; specifically, the arrangement of light-absorbing pigment molecules in antenna complexes, also called light-harvesting complexes (sometimes abbreviated LHC, not to be confused with the Large Hadron Collider). These pigments are held by proteins in ordered three-dimensional groups so that the pigments that absorb the highest energy light are toward the periphery, and the lowest-energy-absorbing chlorophylls are in the center (Figure \(\PageIndex{2}\)). Sunlight is composed of a broad range of wavelengths, some of which are transiently absorbed by the pigments. After a pigment molecule absorbs a photon, the energy is released and passed on to a pigment tuned to a slightly lower energy level (longer wavelength), and from there to an even lower-energy pigment, and so on until it reaches the reaction center chlorophylls. In this way, energy from a wide range of light wavelengths/energies can all contribute to the ATP and NADPH production by the light reactions. The antenna complex is crucial because it allows the use of a greater portion of the solar light spectrum. And, as a tightly-packed three-dimensional array, photons that pass by one pigment molecule may well hit another one on its way through the array. All of these characteristics combine to increase the efficiency of light use for photosynthesis. The reaction center chlorophylls (P680 for photosystem II, P700 for photosystem I) are the only chlorophylls that actually send excited electrons into the electron transport chain. The other chlorophylls and pigments only act to transfer the energy to the reaction center.
When excited, the reaction center chlorophyll of photosystem II (Figure \(\PageIndex{3}\)) begins the process of electron transport. This chlorophyll is part of a protein complex that also includes a Mn-based oxygen-evolving complex (OEC), pheophytin, and a docking site for plastoquinone. Although the chlorophyll electron is the one excited by the solar energy, the origin of the electrons to keep the chlorophyll replenished comes from the splitting (oxidation) of water to O2 and 4 H+.
The OEC, or oxygen-evolving complex (also WOC, water oxidizing complex) is a metalloenzyme with a Mn4OXCa catalytic cluster, where X is the number of m-oxo-bridges connecting the metal atoms, with surrounding amino acids, especially crucial tyrosines, also playing a role in the coordination sphere of the active site. The overall complex undergoes a series of 4 oxidation state changes as the P680 chlorophylls are excited by the light energy and transfer electrons, but at present it is not known what the exact oxidation state of any given Mn atom is through this series of state changes. The crucial reaction is the formation of the O-O bond to form O2. There are two proposed models for this mechanism. One is that the O-O bond is formed when the OEC has reached its fully oxidized state, and an oxygen in a m-oxo-bridge radical state interacts with a water molecule. The other proposed mechanism is that the O-O bond forms earlier as a complexed peroxide held by the OEC center.
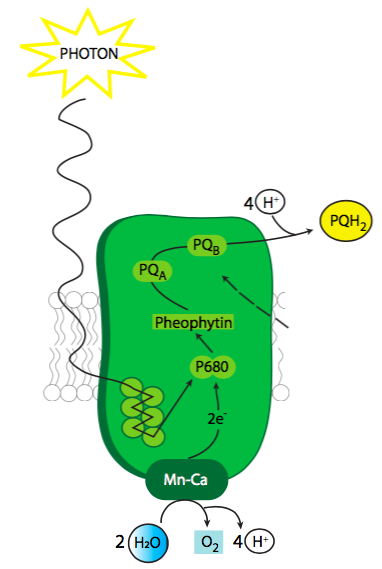
The question of how a cell could generate the energy needed to split water was long a thorny issue because water is an exceptionally stable molecule. The current model suggests that the energy comes from an extremely strong oxidizer in the form of P680+. After P680 is energized by light, an excited electron has enough energy to break away from the chlorophyll and jumps to pheophytin. Pheophytin becomes Pheo- temporarily, and the charge separation in the complex between P680+ and Pheo- helps to enhance the oxidative power of P680+. That extraordinarily strong attraction for electrons is what allows the P680 chlorophyll to tear them away from H2O and split the water. In fact, P680+ is one of the strongest biological oxidizers known. Since four electrons must be taken to fully oxidize two water molecules and generate molecular oxygen, four photoexcitation events are needed. While the exact mechanism is still to be elucidated, it appears that the OEC helps to stabilize the water molecule during this process as well as holding onto each electron as it comes off.
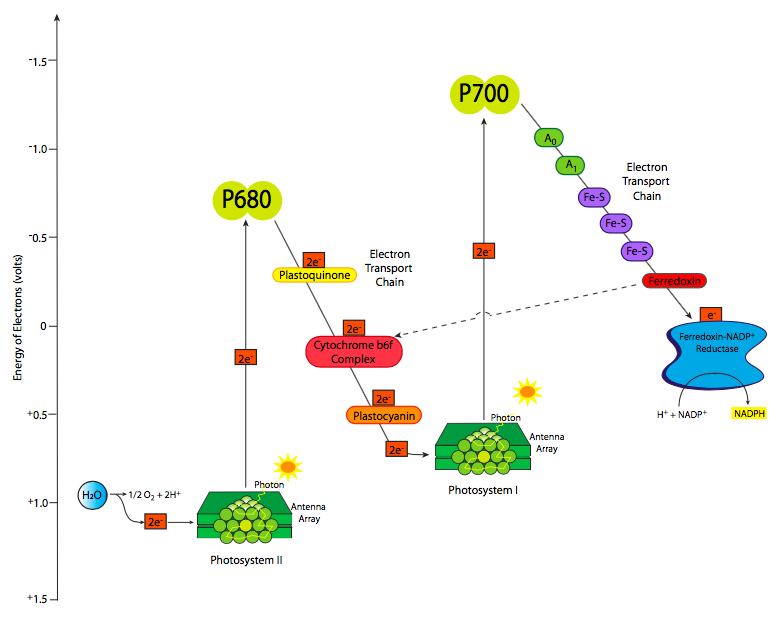
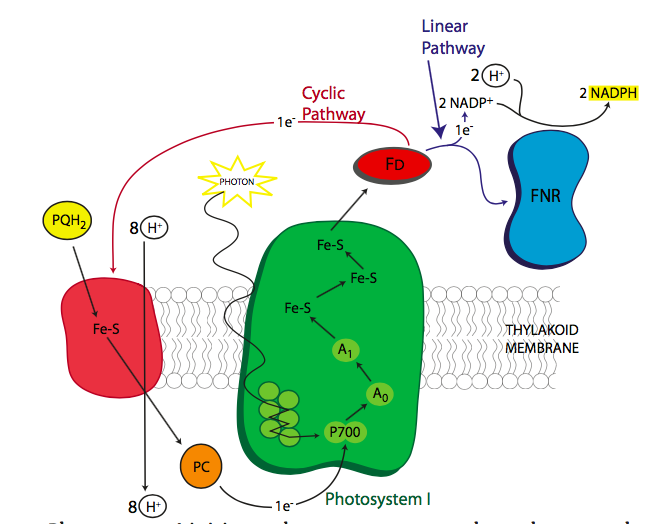
The excited electrons, moving from the OEC to P680+ to pheophytin, next move to the lipid-soluble carrier, plastoquinone. The similarity of the name with the mitochondrial carrier ubiquinone is not a coincidence. They function similarly, and as the plastoquinone takes on the electrons, it also takes on protons from the stromal side of the thylakoid membrane. The PQ moves within the membrane from pheophytin to cytochrome b6f. As the electrons are transferred to cytochrome b6f, the protons are then dropped off on the lumenal side of the membrane, increasing their concentration in the chloroplast lumen, and building a proton gradient to power ATP synthase. Cytochrome b6f passes the electrons on to plastocyanin, an aqueous-phase carrier, which shuttles the electrons to the P700 reaction center chlorophyll of photosystem I. However, after all the transfers, the energy level of the electrons is now fairly low (Figure \(\PageIndex{4}\)) and unable to power the upcoming reactions. Since it is now on a reaction center chlorophyll, the obvious answer is to re-energize it with a bit of sunlight. This raises the electron energy sufficiently to reduce ferredoxin. Now things get a little complicated.
This part of photosynthesis can take one of two directions, the linear pathway, which generates both NADPH and ATP, and the cyclic pathway which mostly generates ATP. Most of the time, the linear pathway is taken, with the electrons on ferredoxin transferred via ferredoxin-NADPH reductase (FNR) onto NADPH. However, sometimes the cell requires significantly more ATP than NADPH, in which case, the electrons from ferredoxin are transferred back to plastoquinone via ferredoxin-plastoquinone reductase. This acts just as described above, and pumps more protons across the membrane to power the ATP synthase. ATP synthesis goes up and NADPH synthesis goes down.
The ATP and NADPH generated by the chloroplast are almost exclusively used by the chloroplast itself (and not distributed to the rest of the cell) to power the dark reactions, which are energetically expensive. In fact, when the light reactions are not running due to darkness, some plant cells have mechanisms to prevent the dark reactions from using the limited resources of cellular, non-chloroplastic, respiration. The simplest method of such limitation is the pH sensitivity of rubisco (ribulose bis-phosphate carboxylase), at least in C3 plants (see below). Rubisco has a very sharp pH optimum at about pH 8.0, so while the light reactions are running and the protons are being pumped, the pH rises to about 8 and rubisco works, but in the dark, the pH drops back to its basal level close to 7.0, inhibiting rubisco activity.


